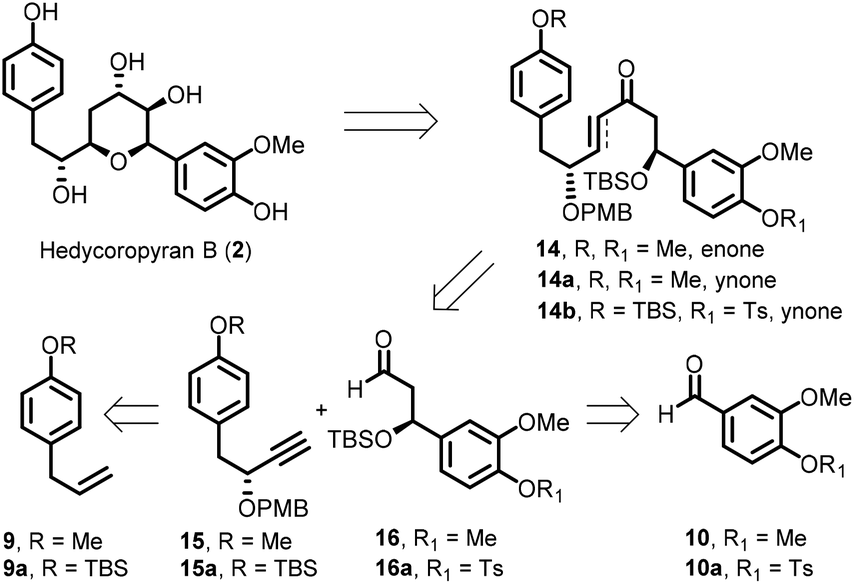
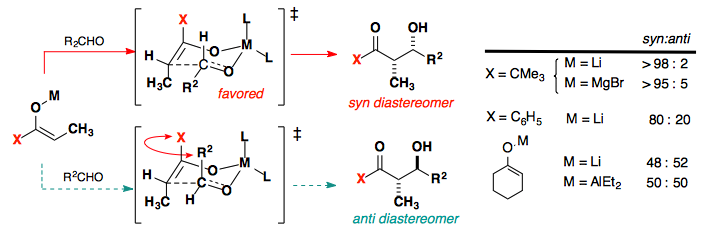
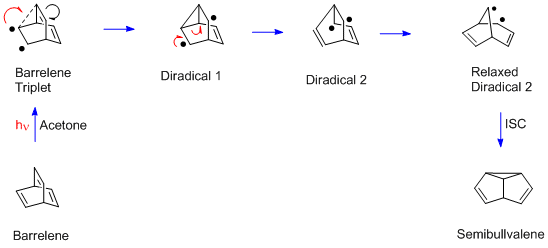
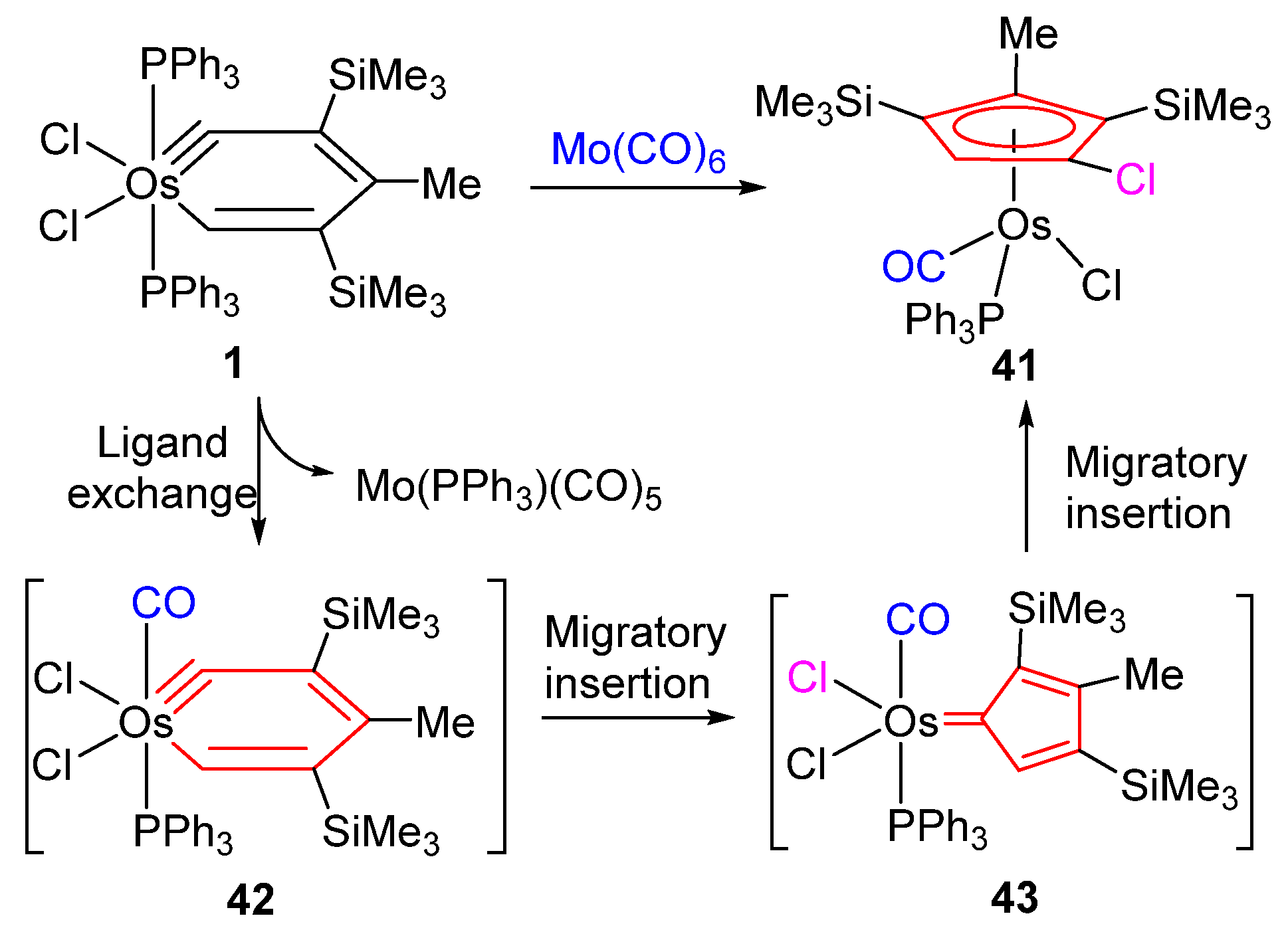
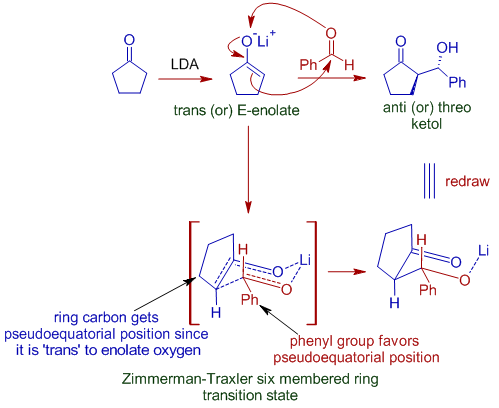
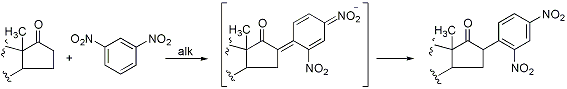
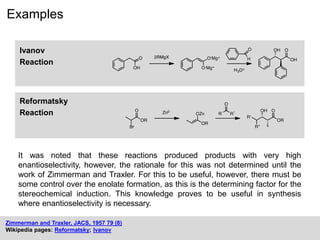
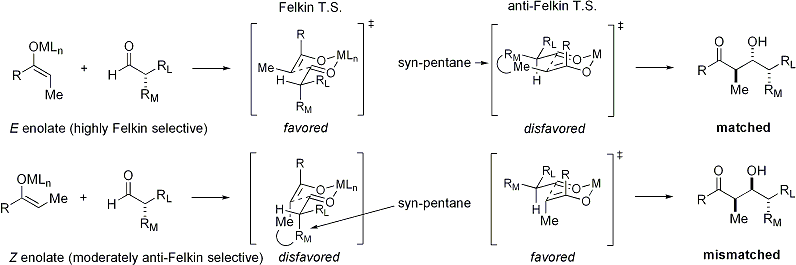
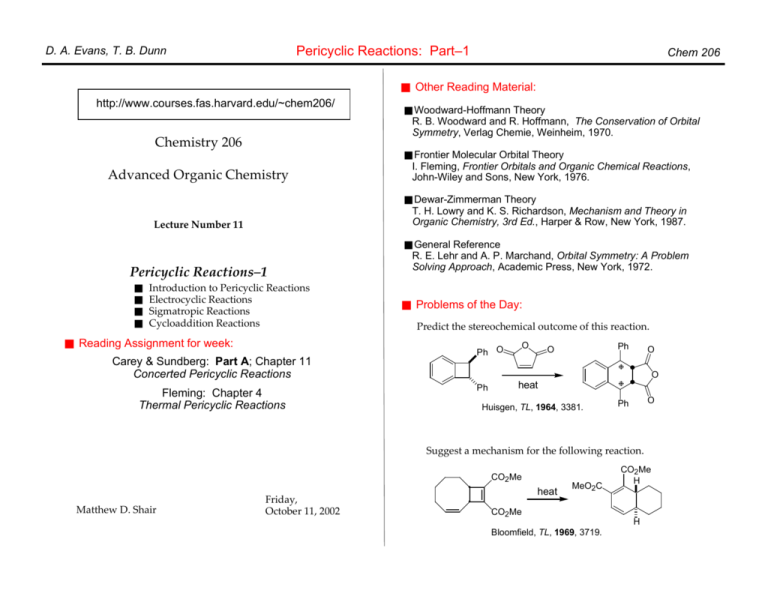
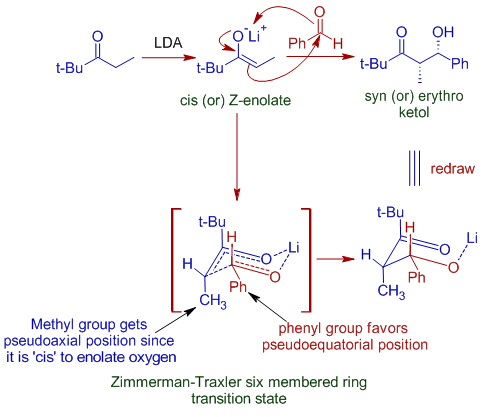
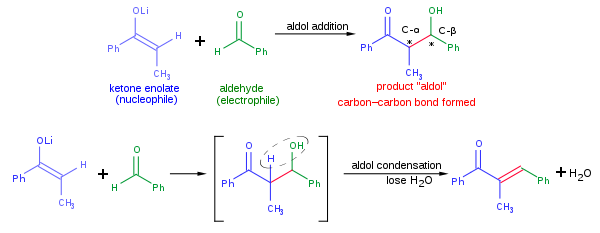
The Huisgen Reaction: Milestones of the 1,3‐Dipolar Cycloaddition - Breugst - 2020 - Angewandte Chemie International Edition - Wiley Online Library

The reaction of organic hydroperoxides with aromatic nitro compounds: First examples of a Vicarious Nucleophilic Hydroxylation - Mattersteig - 1990 - Journal für Praktische Chemie - Wiley Online Library
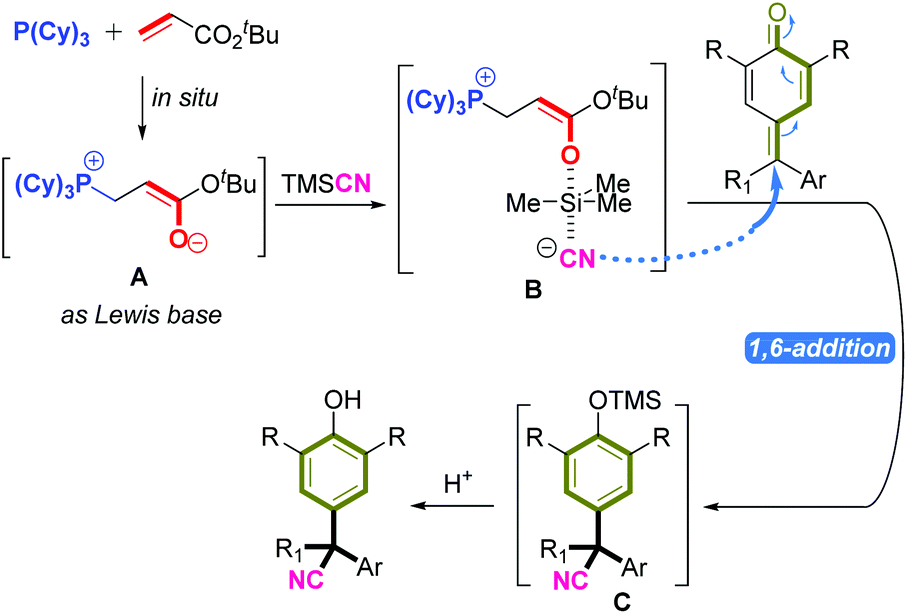
In situ phosphonium-containing Lewis base-catalyzed 1,6-cyanation reaction: a facile way to obtain α-diaryl and α-triaryl acetonitriles - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO01501J

Reaction mechanism of TaqAM leading to transglycosylation or hydrolysis. | Download Scientific Diagram
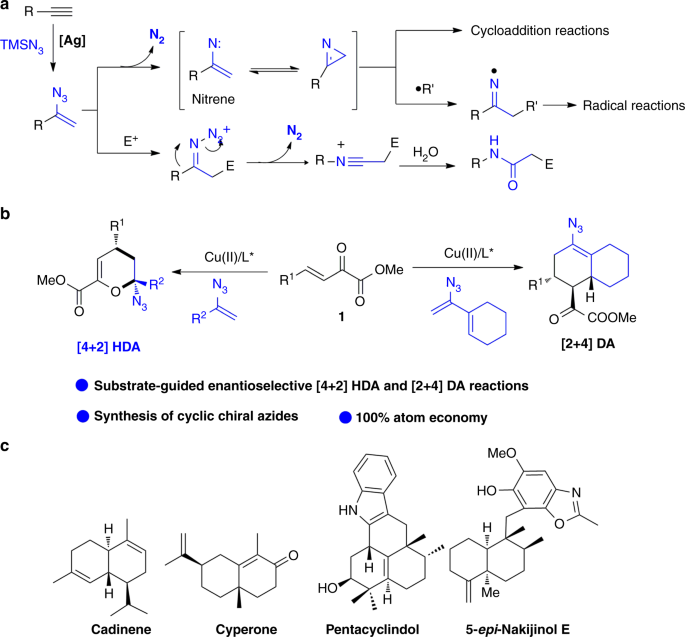
Divergent synthesis of chiral cyclic azides via asymmetric cycloaddition reactions of vinyl azides | Nature Communications