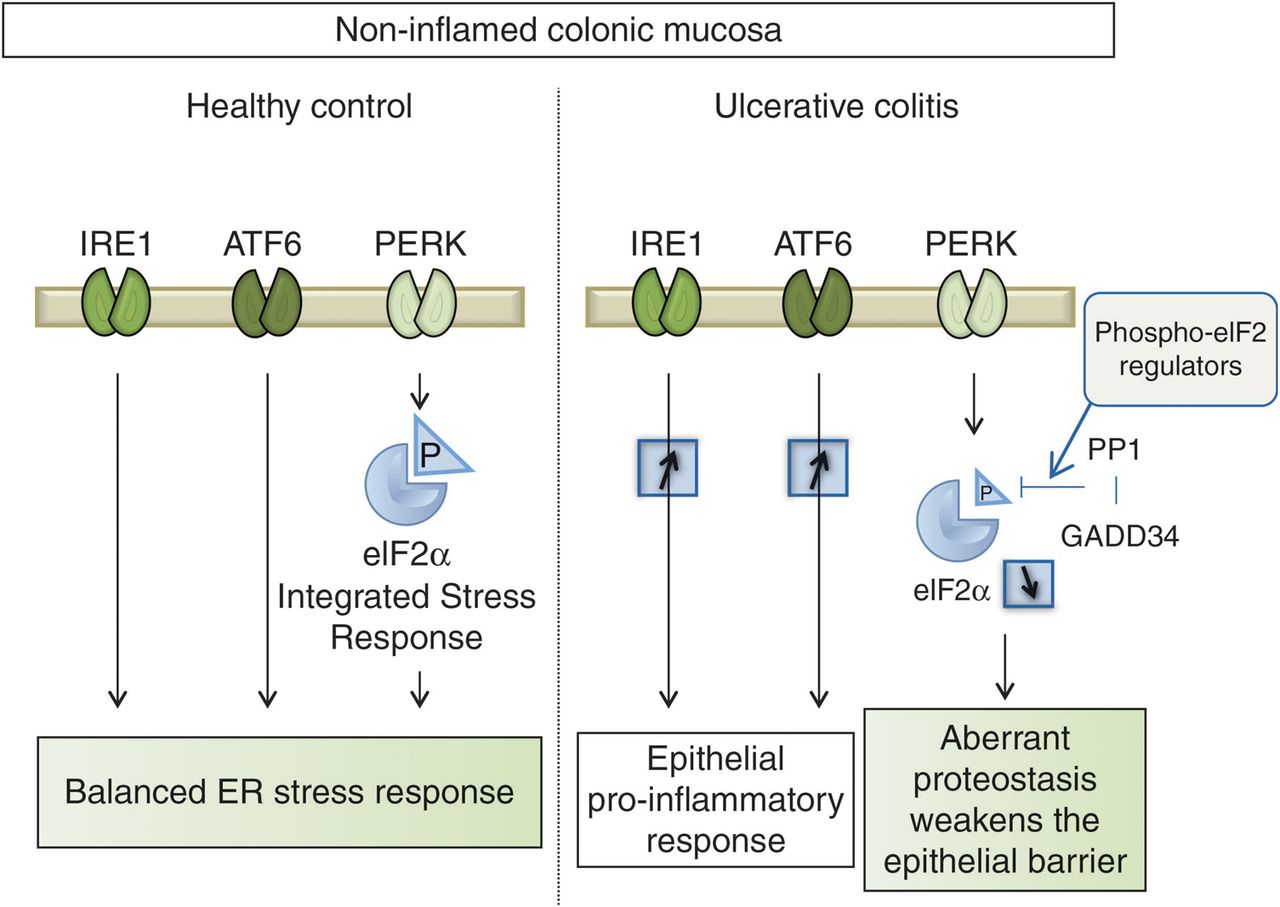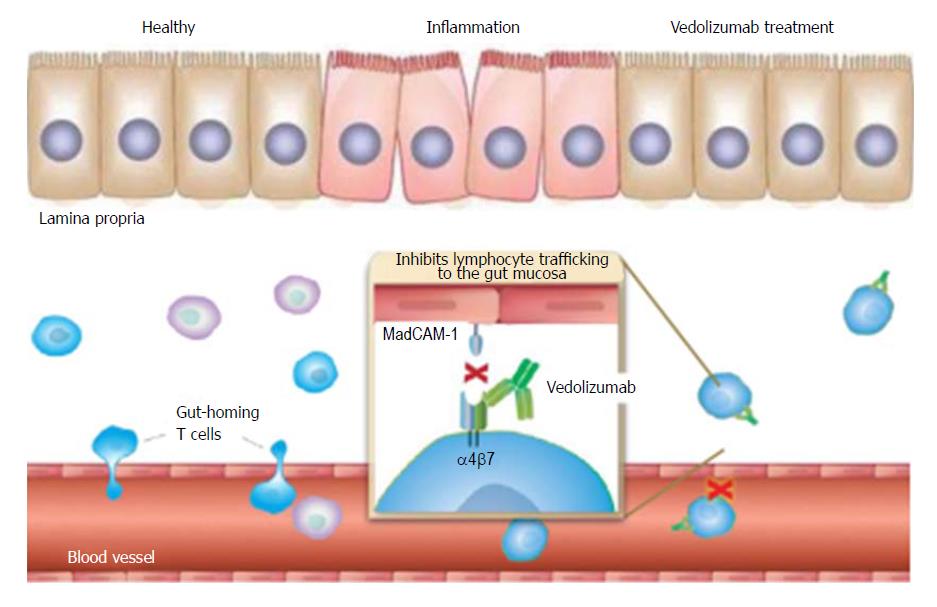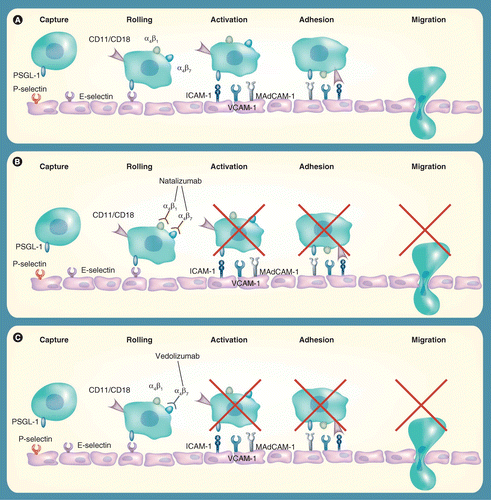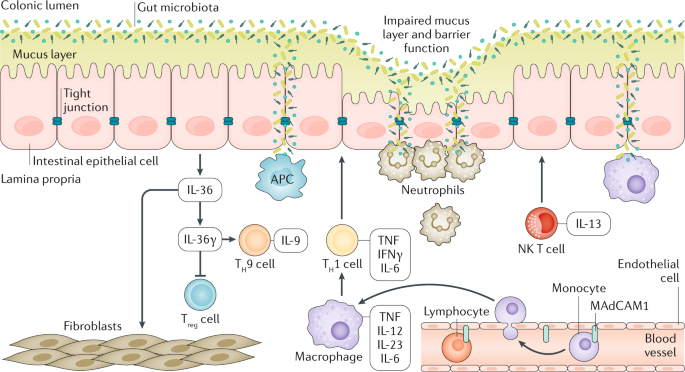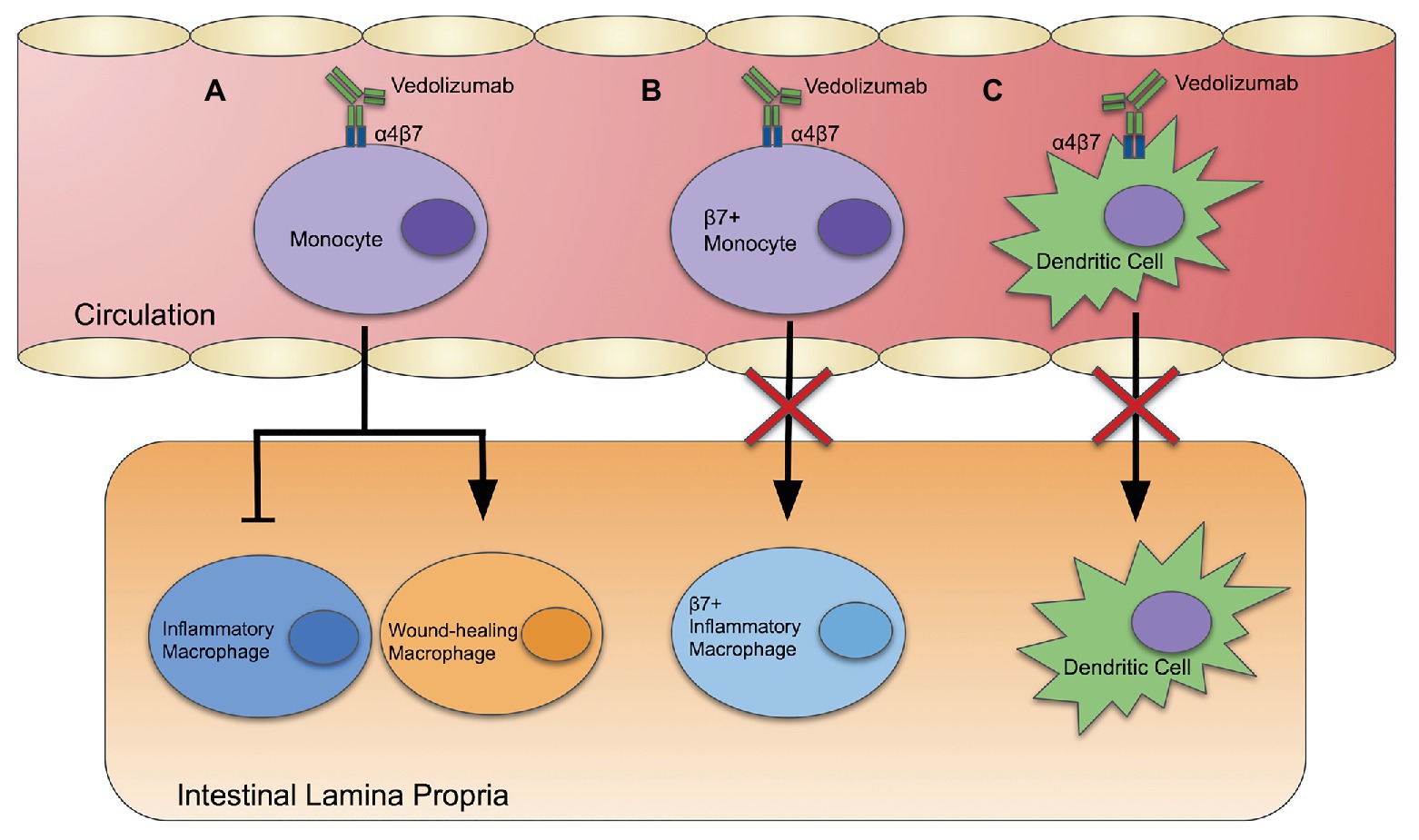
Frontiers | Vedolizumab: Potential Mechanisms of Action for Reducing Pathological Inflammation in Inflammatory Bowel Diseases | Cell and Developmental Biology
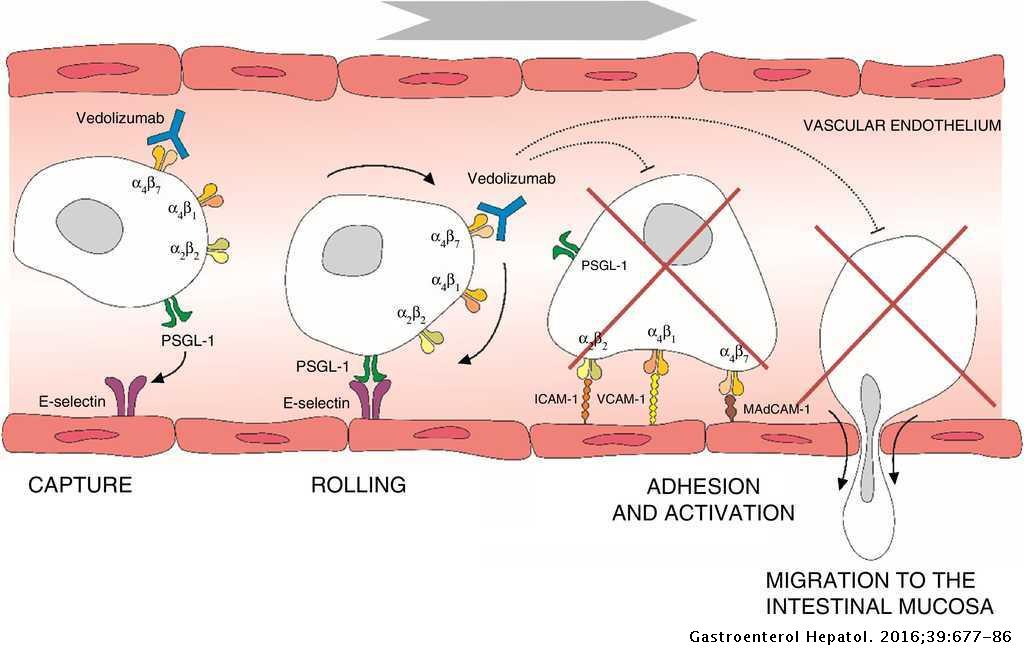
Efficacy and safety of vedolizumab in the treatment of ulcerative colitis | Gastroenterología y Hepatología (English Edition)
View of IMPORTANCE OF NANOCARRIERS AND PROBIOTICS IN THE TREATMENT OF ULCERATIVE COLITIS | International Journal of Applied Pharmaceutics
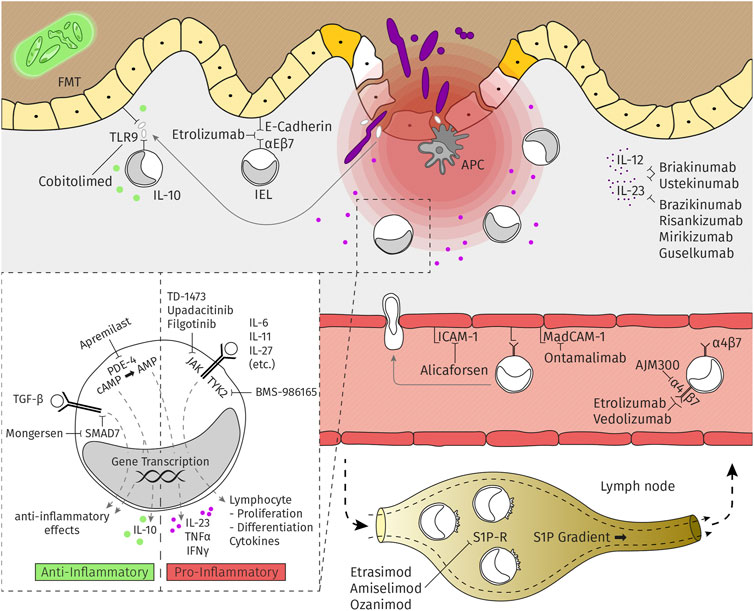
Frontiers | An Update for Pharmacologists on New Treatment Options for Inflammatory Bowel Disease: The Clinicians' Perspective | Pharmacology

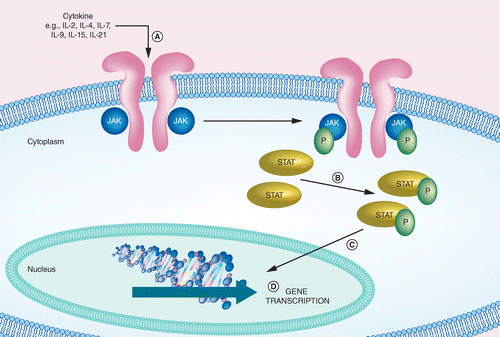
Upadacitinib for Crohn's Disease and Ulcerative Colitis Treatment: Hitting the Selective JAKpot - Gastroenterology

Small molecule oral targeted therapies in ulcerative colitis - The Lancet Gastroenterology & Hepatology

An Overview of Novel and Emerging Therapies for Inflammatory Bowel Disease - European Medical Journal

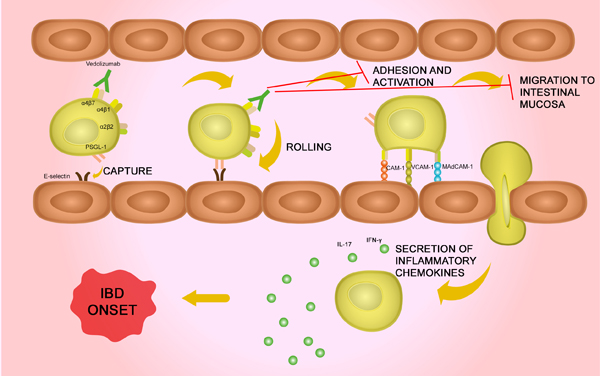
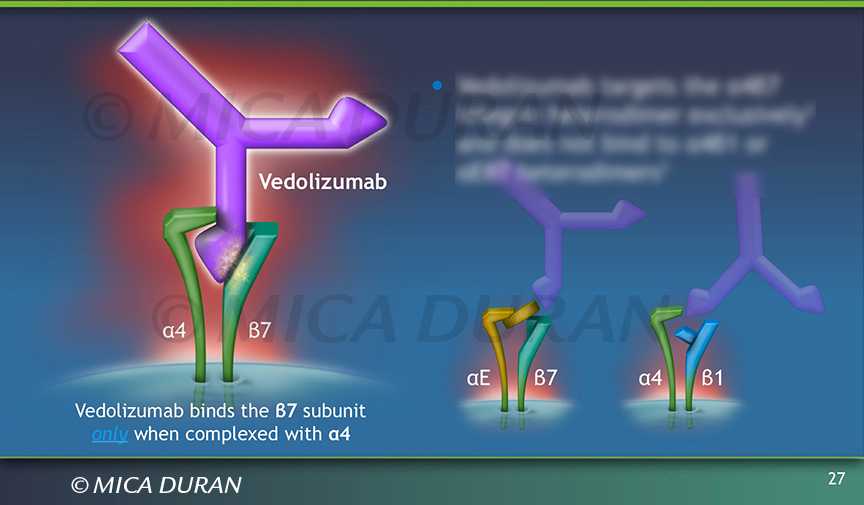
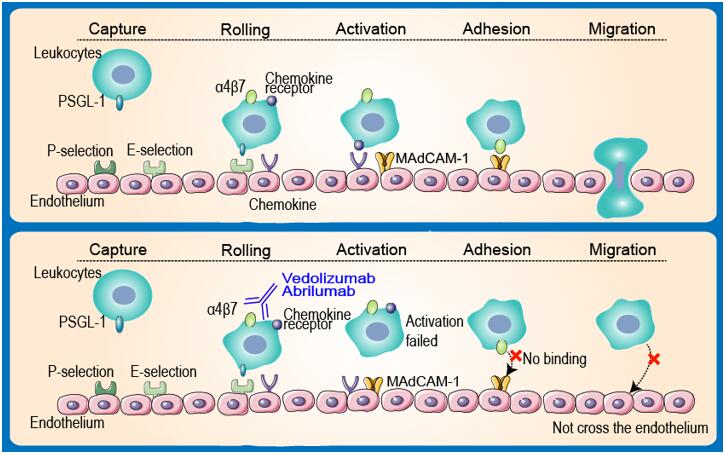
Mechanism of vedolizumab. This image is modified from their original... | Download Scientific Diagram

Takeda Submits Marketing Authorisation Application for Vedolizumab in Moderately to Severely Active Ulcerative Colitis and Crohn's Disease in the European Union | New Drug Approvals