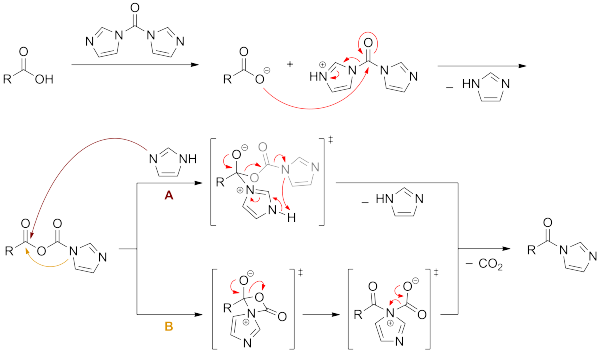
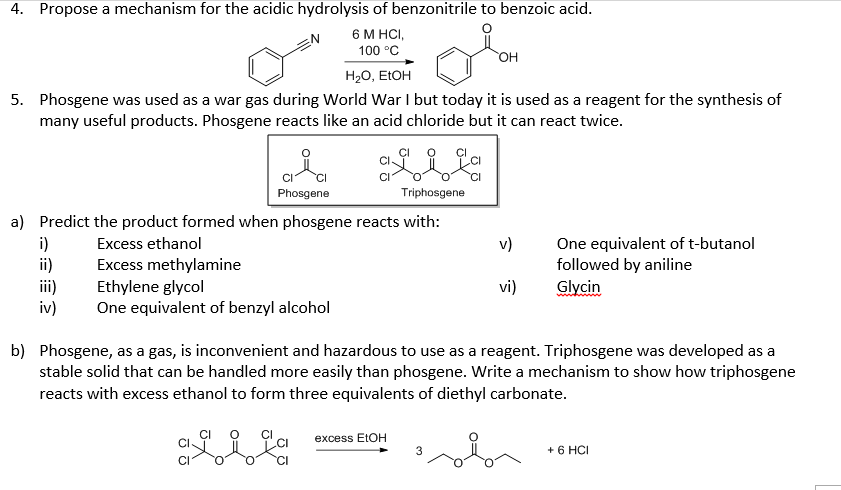
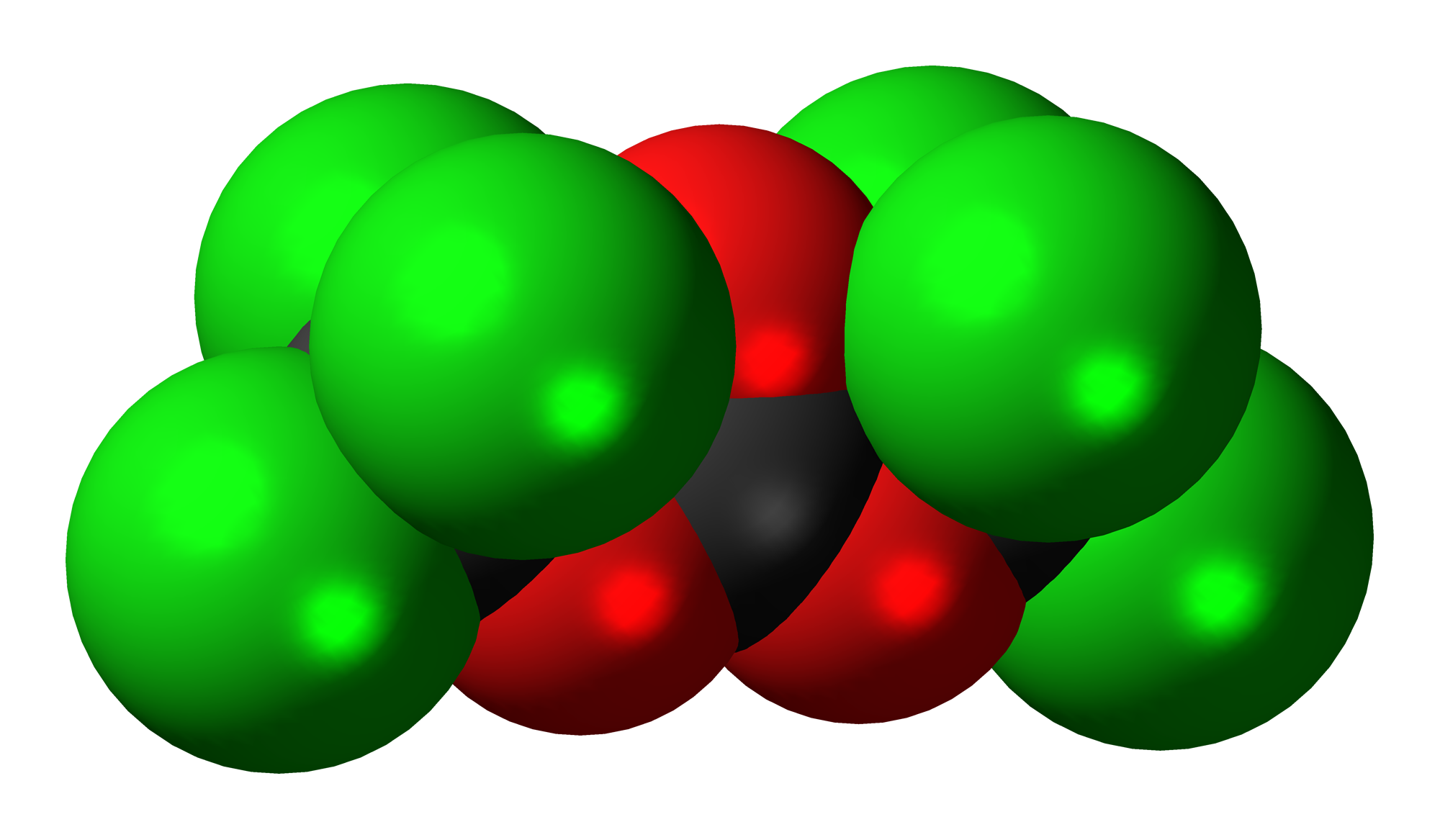
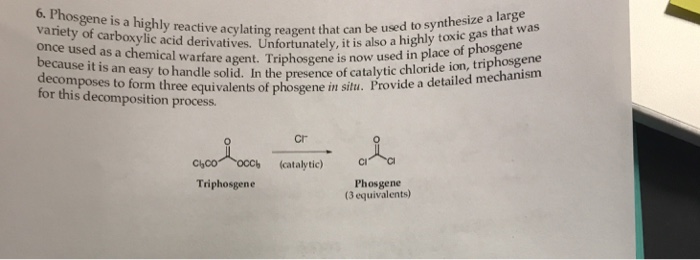
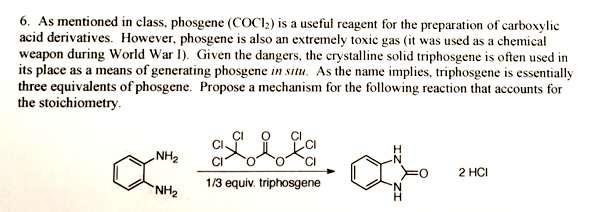
Transposition of a triphosgene-based process for pharmaceutical development: from mg·h-1 to kg·h-1 of an unsymmetrical urea

SciELO - Brasil - New aminoporphyrins bearing urea derivative substituents: synthesis, characterization, antibacterial and antifungal activity New aminoporphyrins bearing urea derivative substituents: synthesis, characterization, antibacterial and ...

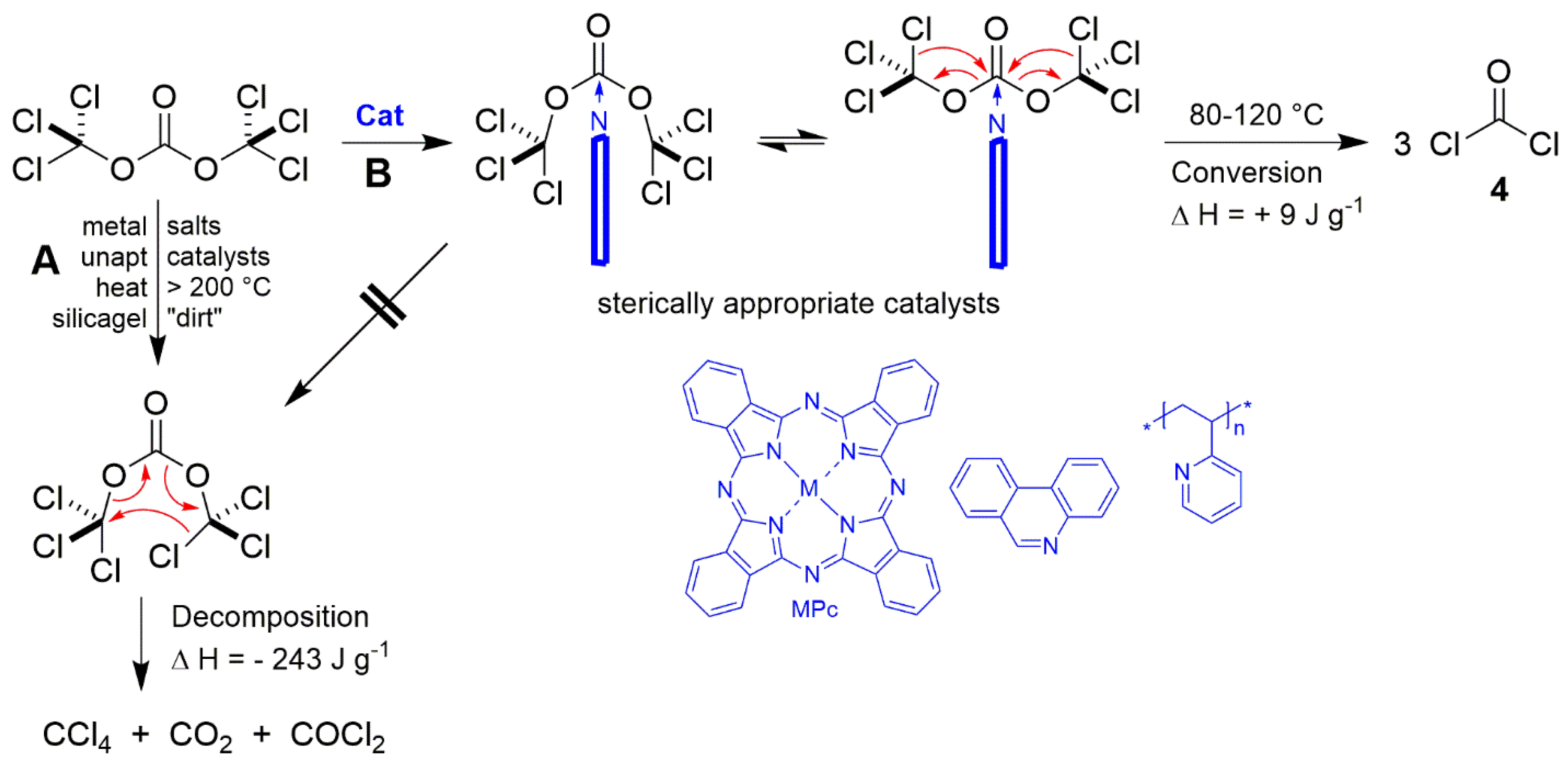
PDF) Conversion of Bis(trichloromethyl) Carbonate to Phosgene and Reactivity of Triphosgene, Diphosgene, and Phosgene with Methanol 1
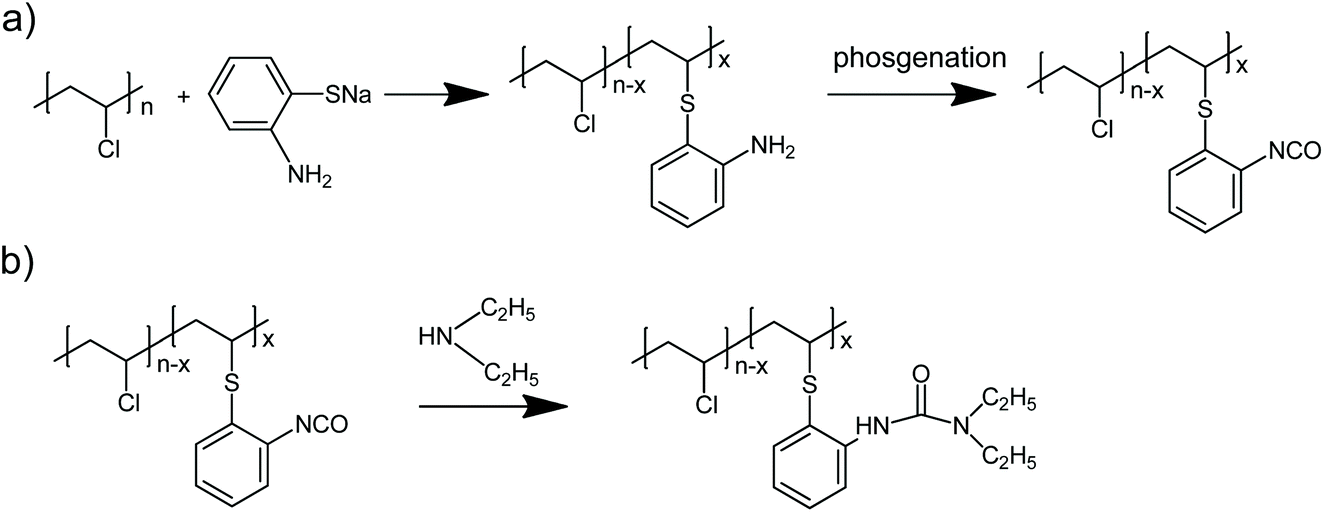
General approach to prepare polymers bearing pendant isocyanate groups - Polymer Chemistry (RSC Publishing) DOI:10.1039/D0PY00989J
Molecules | Free Full-Text | Synergy Effects in the Chemical Synthesis and Extensions of Multicomponent Reactions (MCRs)—The Low Energy Way to Ultra-Short Syntheses of Tailor-Made Molecules | HTML

A REVIEW AND METHODS TO HANDLE PHOSGENE, TRIPHOSGENE SAFELY DURING DRUG SYNTHESIS | New Drug Approvals

EP1017623B1 - Method and device for preparing phosgene from diphosgene and/or triphosgene - Google Patents

Sensitive and Selective Detection of Phosgene, Diphosgene, and Triphosgene by a 3,4‐Diaminonaphthalimide in Solutions and the Gas Phase - Wang - 2018 - Chemistry – A European Journal - Wiley Online Library
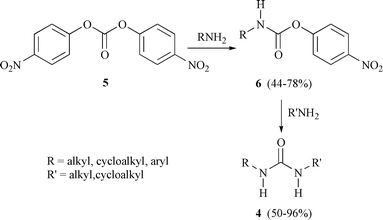
Selected syntheses of ureas through phosgene substitutes - Green Chemistry (RSC Publishing) DOI:10.1039/B002127J
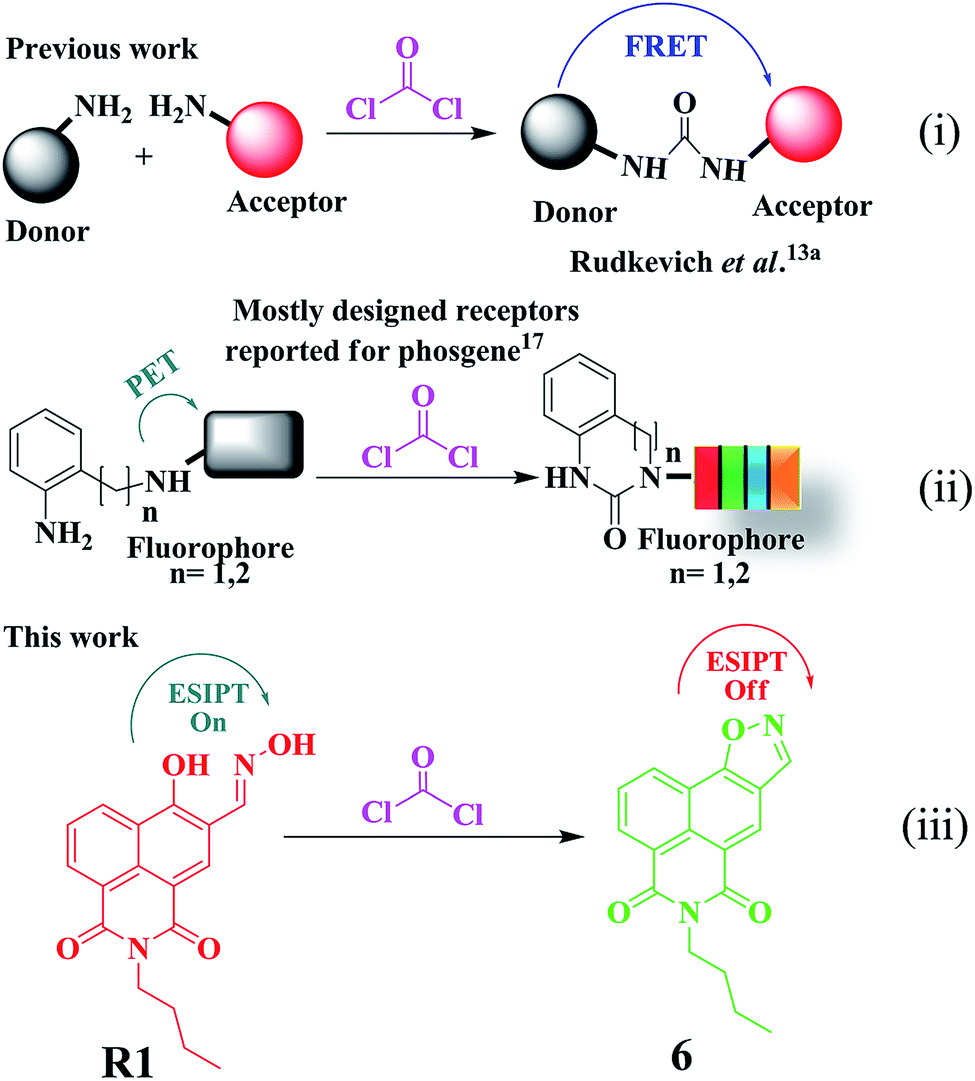
Ratiometric chemodosimeter: an organic-nanofiber platform for sensing lethal phosgene gas - Journal of Materials Chemistry A (RSC Publishing) DOI:10.1039/C8TA10481F

Polymers | Free Full-Text | Strategies to Fabricate Polypeptide-Based Structures via Ring-Opening Polymerization of N-Carboxyanhydrides | HTML