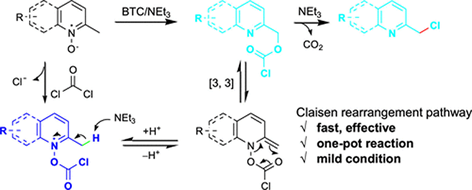
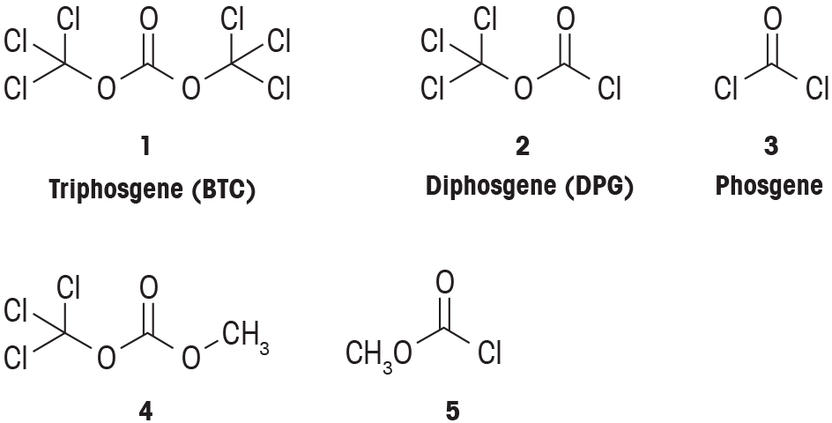
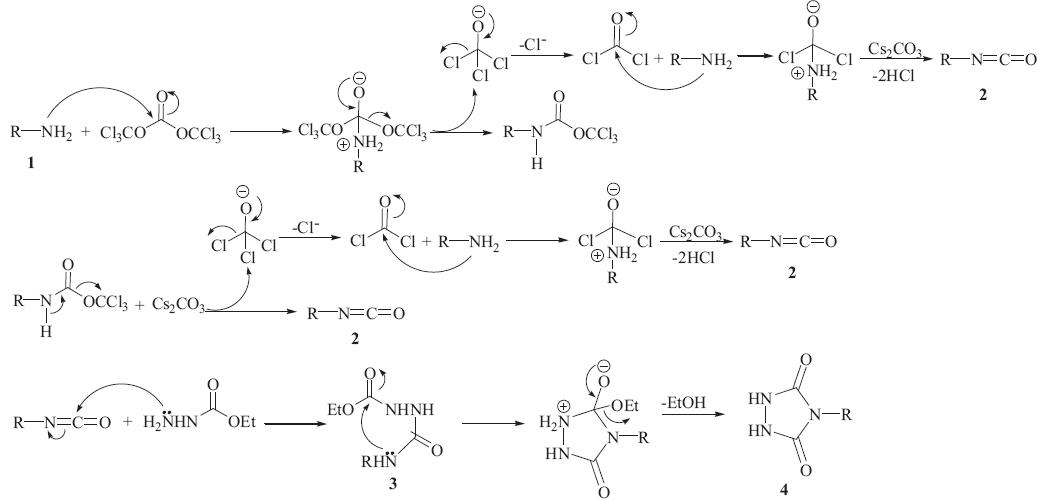
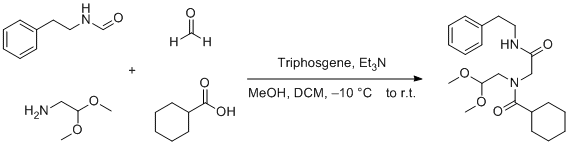
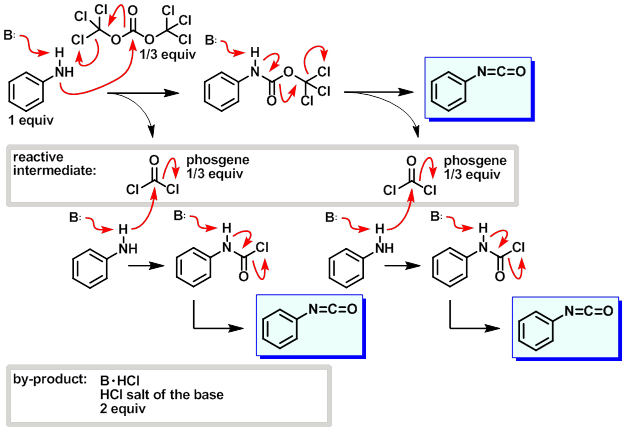
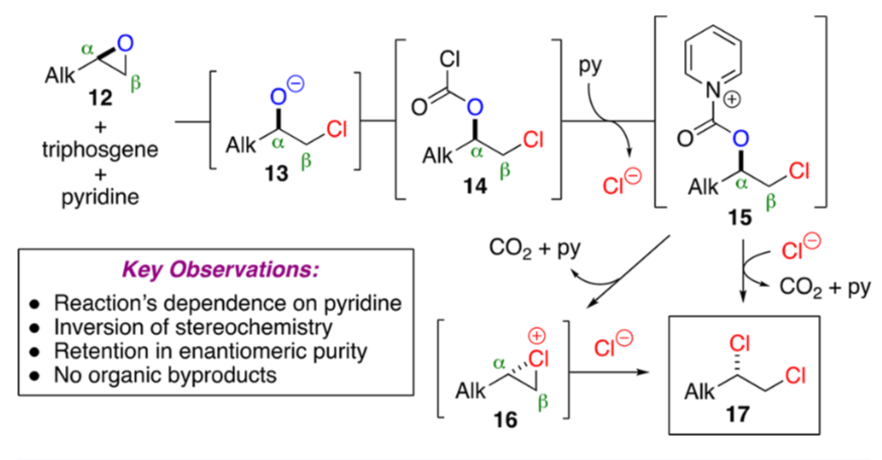
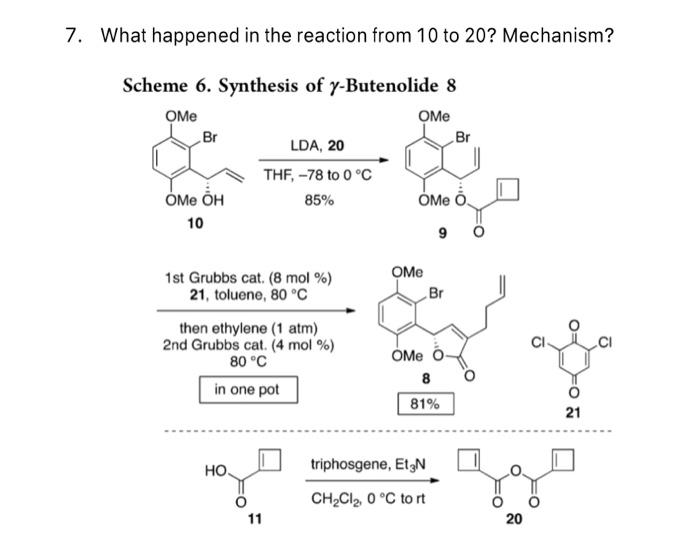
PDF) Conversion of Bis(trichloromethyl) Carbonate to Phosgene and Reactivity of Triphosgene, Diphosgene, and Phosgene with Methanol 1

A REVIEW AND METHODS TO HANDLE PHOSGENE, TRIPHOSGENE SAFELY DURING DRUG SYNTHESIS | New Drug Approvals

Triphosgene and DMAP as Mild Reagents for Chemoselective Dehydration of Tertiary Alcohols | Organic Letters

Transposition of a triphosgene-based process for pharmaceutical development: from mg·h-1 to kg·h-1 of an unsymmetrical urea
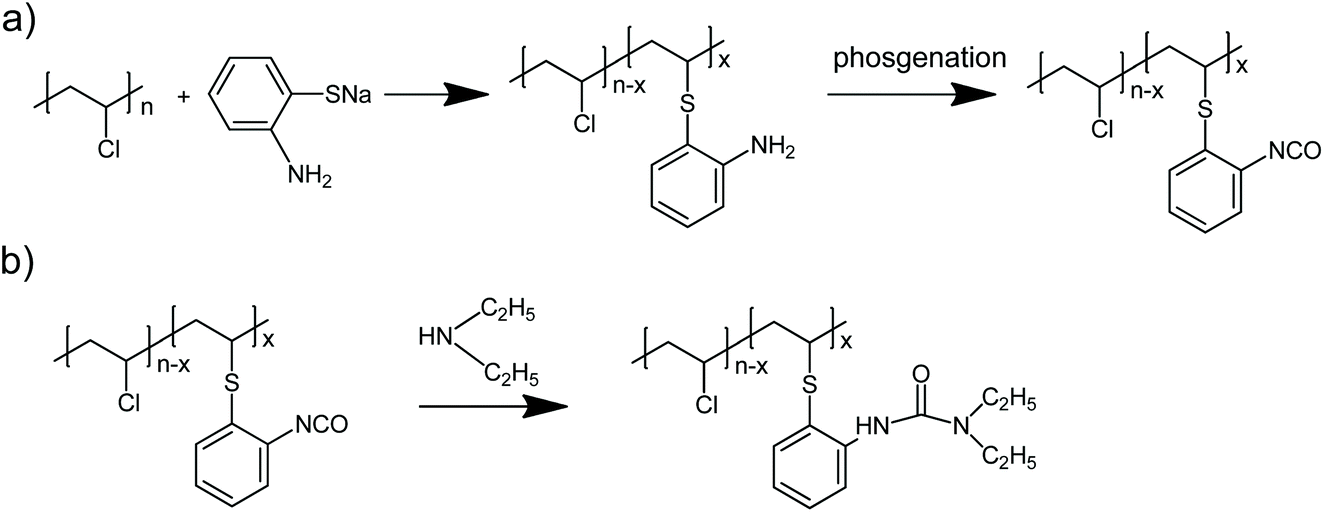
General approach to prepare polymers bearing pendant isocyanate groups - Polymer Chemistry (RSC Publishing) DOI:10.1039/D0PY00989J

Sensitive and Visual Detection of Phosgene by a TICT‐Based BODIPY Dye with 8‐(o‐Hydroxy)aniline as the Active Site - Fu - 2021 - Chemistry – A European Journal - Wiley Online Library

A REVIEW AND METHODS TO HANDLE PHOSGENE, TRIPHOSGENE SAFELY DURING DRUG SYNTHESIS | New Drug Approvals

Rapid and Mild Lactamization Using Highly Electrophilic Triphosgene in a Microflow Reactor - Fuse - 2021 - Chemistry – A European Journal - Wiley Online Library

Transposition of a triphosgene-based process for pharmaceutical development: from mg·h-1 to kg·h-1 of an unsymmetrical urea