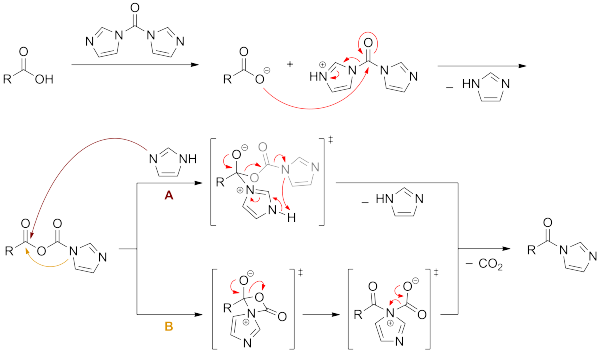
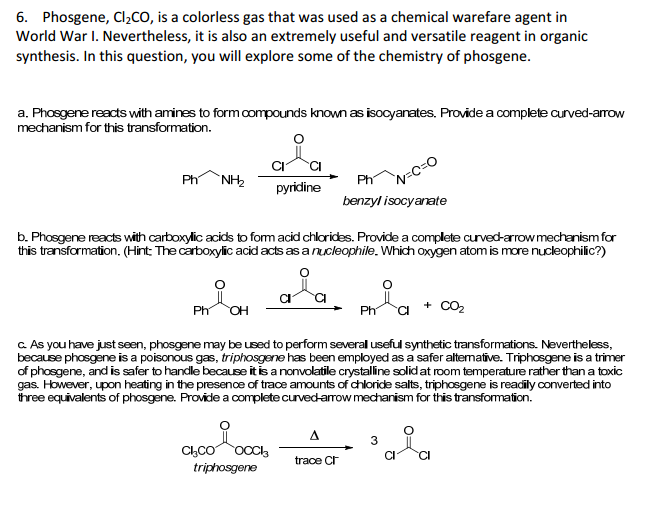
A REVIEW AND METHODS TO HANDLE PHOSGENE, TRIPHOSGENE SAFELY DURING DRUG SYNTHESIS | New Drug Approvals

Simple, rapid, and highly sensitive detection of diphosgene and triphosgene by spectrophotometric methods. - Abstract - Europe PMC

Transposition of a triphosgene-based process for pharmaceutical development: from mg·h-1 to kg·h-1 of an unsymmetrical urea
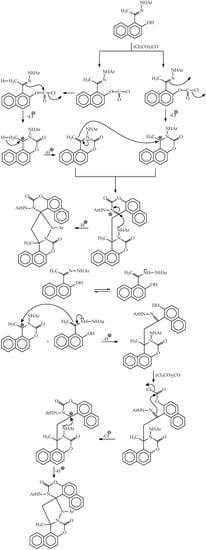
Molecules | Free Full-Text | Cyclization of Hydrazones of 2-Acetyl-1-naphthol and 1-Acetyl-2-naphthol with Triphosgene. Synthesis of Spiro Naphthoxazine Dimers | HTML

WO2000002898A1 - Processes for coupling amino acids using bis-(trichloromethyl) carbonate - Google Patents

Transposition of a triphosgene-based process for pharmaceutical development: from mg·h-1 to kg·h-1 of an unsymmetrical urea
![Thermally induced formal [3+2] cyclization of ortho-aminoaryl-tethered alkylidenecyclopropanes: facile synthesis of furoquinoline and thienoquinoline derivatives - Chemical Communications (RSC Publishing) Thermally induced formal [3+2] cyclization of ortho-aminoaryl-tethered alkylidenecyclopropanes: facile synthesis of furoquinoline and thienoquinoline derivatives - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/image/article/2016/cc/c5cc09218c/c5cc09218c-s4_hi-res.gif)
Thermally induced formal [3+2] cyclization of ortho-aminoaryl-tethered alkylidenecyclopropanes: facile synthesis of furoquinoline and thienoquinoline derivatives - Chemical Communications (RSC Publishing)
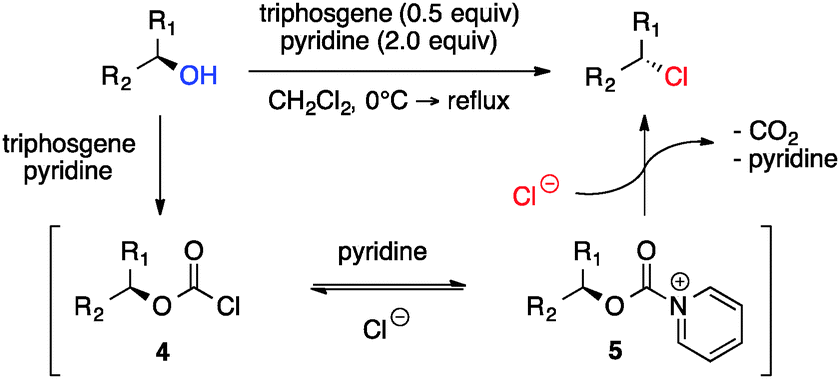
Triphosgene–pyridine mediated stereoselective chlorination of acyclic aliphatic 1,3-diols - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC06365E
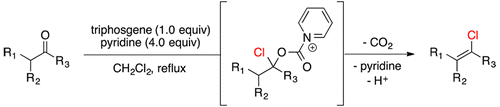
Synthesis of Vinyl Chlorides via Triphosgene–Pyridine Activation of Ketones,The Journal of Organic Chemistry - X-MOL