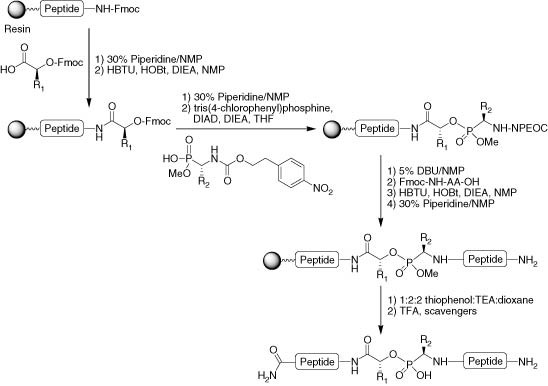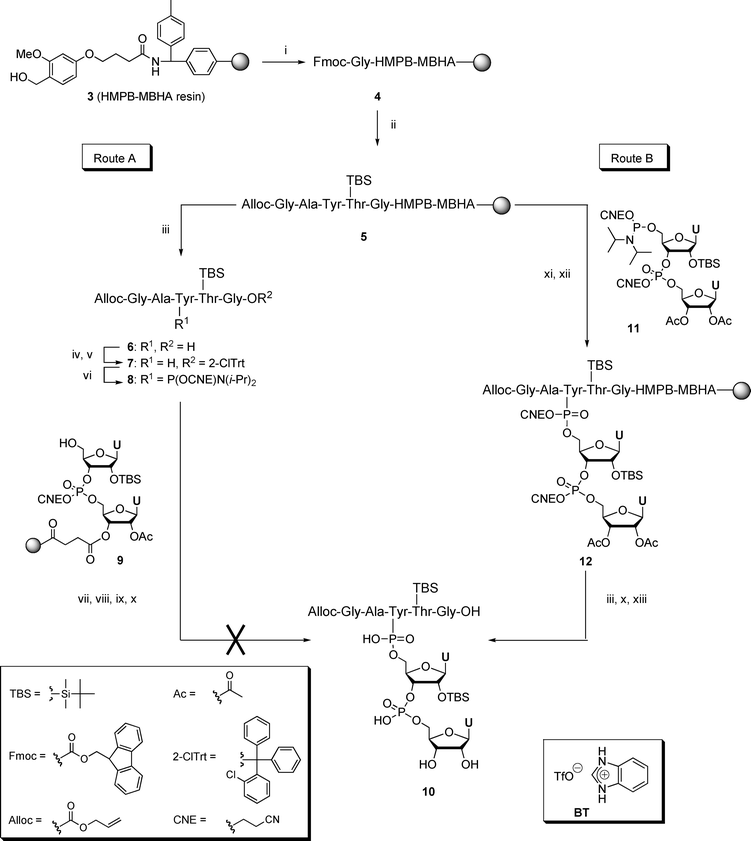
Chemical synthesis of picornaviral protein primers of RNA replication - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B608544J

Advances in Fmoc solid‐phase peptide synthesis - Behrendt - 2016 - Journal of Peptide Science - Wiley Online Library

Solid-Phase Peptide Synthesis of Dipeptide (Histidine-β-Alanine) as a Chelating Agent by Using Trityl Chloride Resin, for Removal of Al3+, Cu2+, Hg2+ and Pb2+: Experimental and Theoretical Study
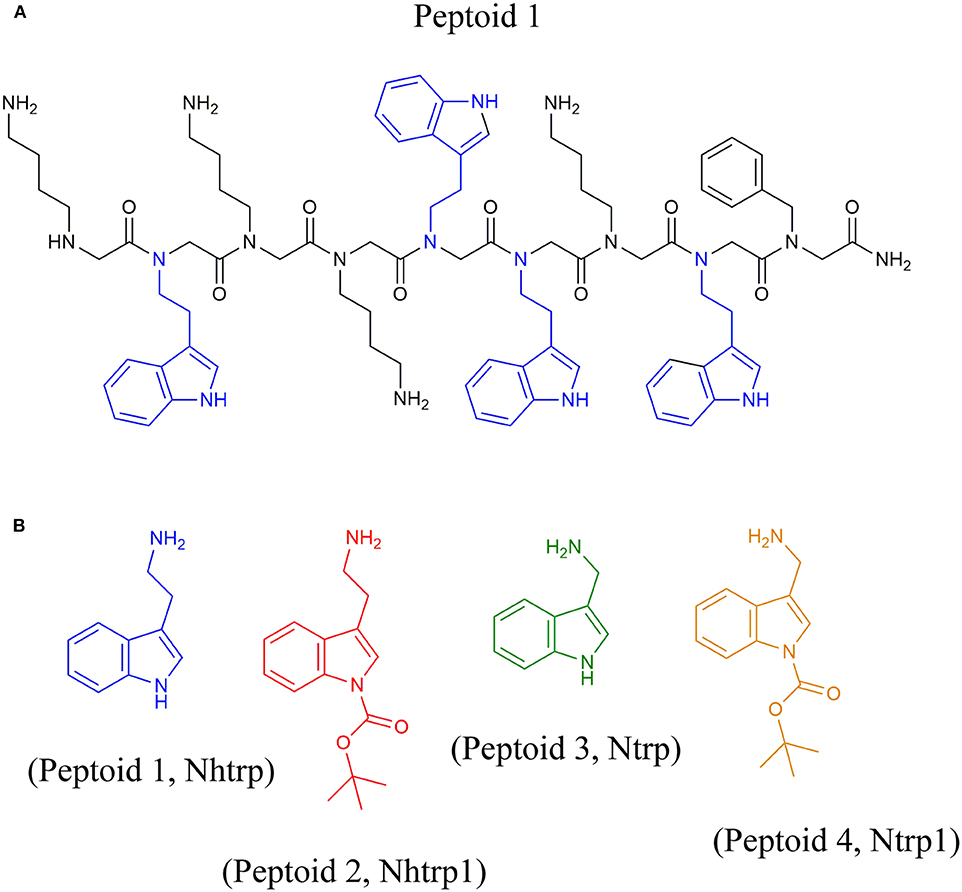
Frontiers | Synthesis of Peptoids Containing Multiple Nhtrp and Ntrp Residues: A Comparative Study of Resin, Cleavage Conditions and Submonomer Protection | Chemistry

Reduction of cysteine‐S‐protecting groups by triisopropylsilane - Ste.Marie - 2018 - Journal of Peptide Science - Wiley Online Library

The renascence of continuous-flow peptide synthesis – an abridged account of solid and solution-based approaches - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB02759A

1,4-Benzenedimethanethiol (1,4-BDMT) as a scavenger for greener peptide resin cleavages - RSC Advances (RSC Publishing)
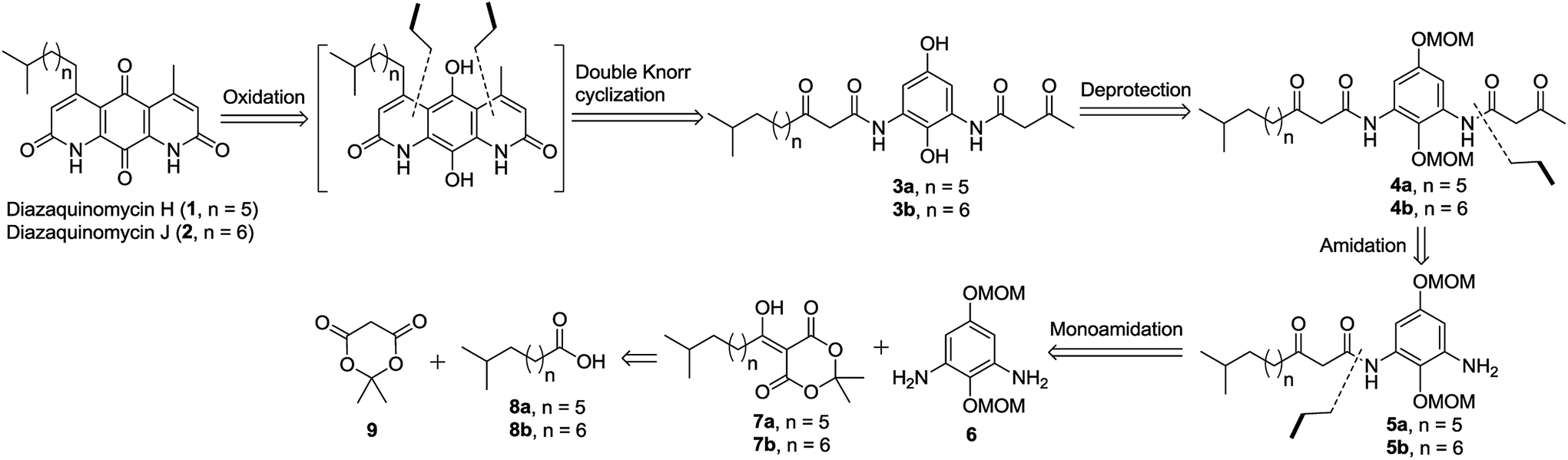
Total synthesis of diazaquinomycins H and J using double Knorr cyclization in the presence of triisopropylsilane - RSC Advances (RSC Publishing) DOI:10.1039/C8RA09792E
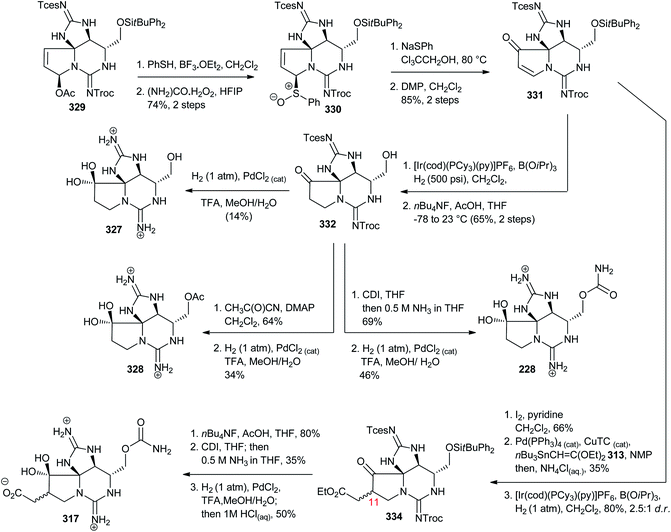
The chemistry and biology of guanidine secondary metabolites - Natural Product Reports (RSC Publishing) DOI:10.1039/D0NP00051E

An important side reaction using the thiol, 3,6‐dioxa‐1,8‐octanedithiol (DODT), in 9‐fluorenylmethoxycarbonyl‐based solid phase peptide synthesis - Harris - 2014 - Journal of Peptide Science - Wiley Online Library