Suppression of Side Reactions During Final Deprotection Employing a Strong Acid in Boc Chemistry: Regeneration of Methionyl Residues from Their Sulfonium Salts | Semantic Scholar

SciELO - Brasil - An Efficient and Chemoselective Deprotection of Aryl <i>tert</i>-Butyldimethylsilyl (TBDMS) Ethers by NaCN An Efficient and Chemoselective Deprotection of Aryl <i>tert</i>-Butyldimethylsilyl (TBDMS) Ethers by NaCN

Deprotection of N-Boc Groups under Continuous-Flow High-Temperature Conditions | The Journal of Organic Chemistry

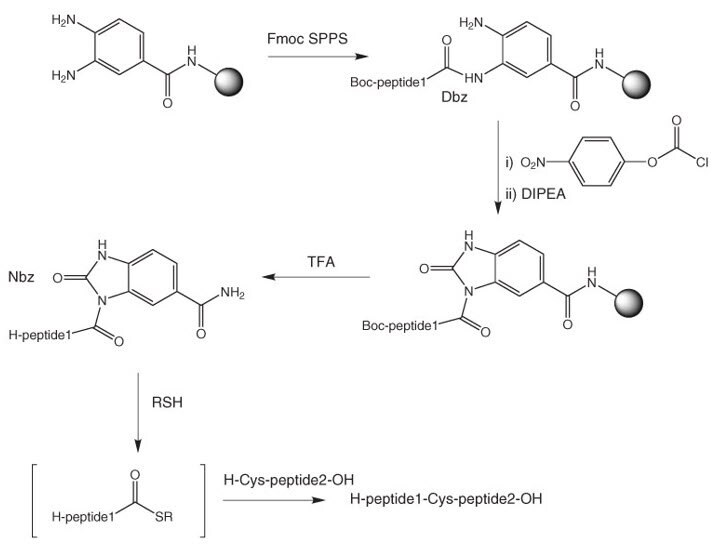
Further Development of New Deprotection Chemistry for Cysteine and Selenocysteine Side Chain Protecting Groups | SpringerLink

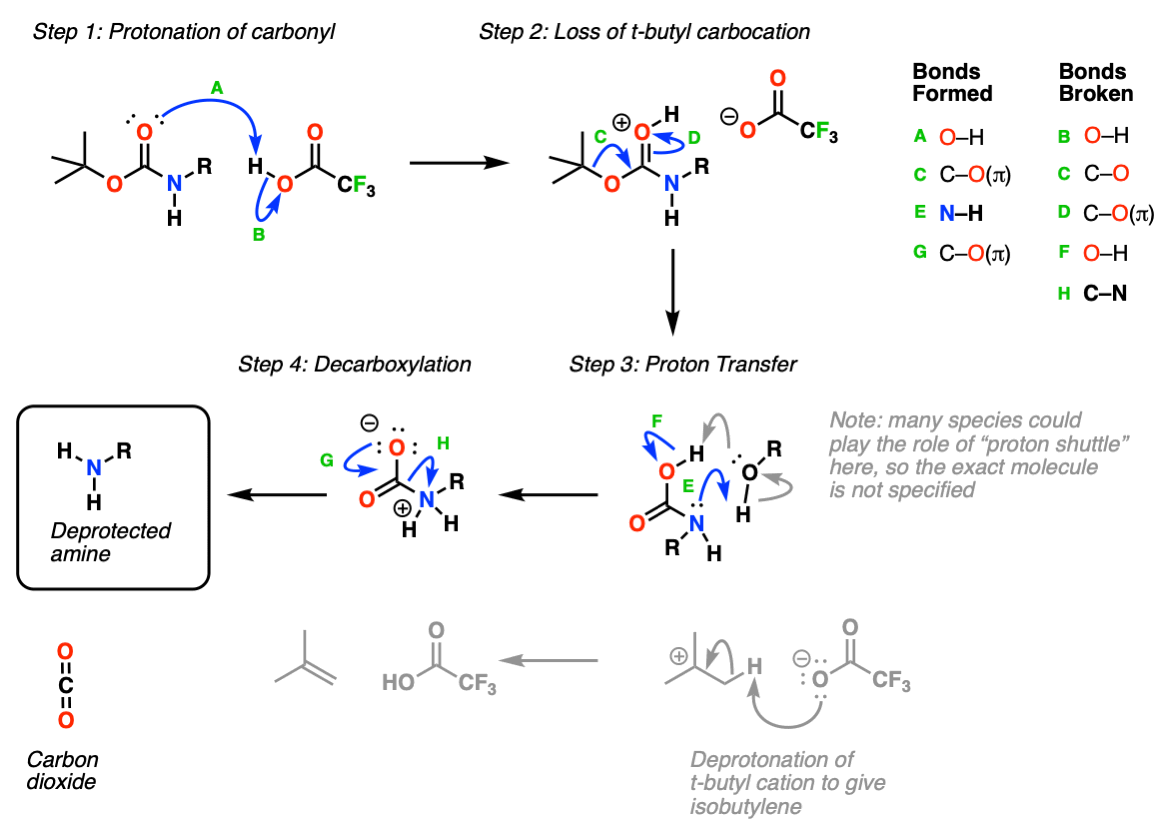
organic chemistry - What happens to the t-butyl cation in the TFA deprotection of a t-butyl ester? - Chemistry Stack Exchange