![PDF] tert-Butyl nitrite mediated nitrogen transfer reactions: synthesis of benzotriazoles and azides at room temperature. | Semantic Scholar PDF] tert-Butyl nitrite mediated nitrogen transfer reactions: synthesis of benzotriazoles and azides at room temperature. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/1ccc8dbf3ffb4ace8b7cc02a374671c6d75a1021/5-Table4-1.png)
PDF] tert-Butyl nitrite mediated nitrogen transfer reactions: synthesis of benzotriazoles and azides at room temperature. | Semantic Scholar

tert‐Butyl Nitrite (TBN), a Multitasking Reagent in Organic Synthesis - Dahiya - 2019 - Chemistry – An Asian Journal - Wiley Online Library
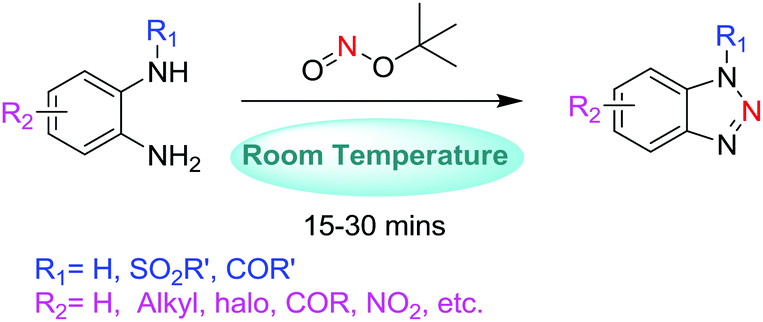
tert -Butyl nitrite mediated nitrogen transfer reactions: synthesis of benzotriazoles and azides at room temperature - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB01950A
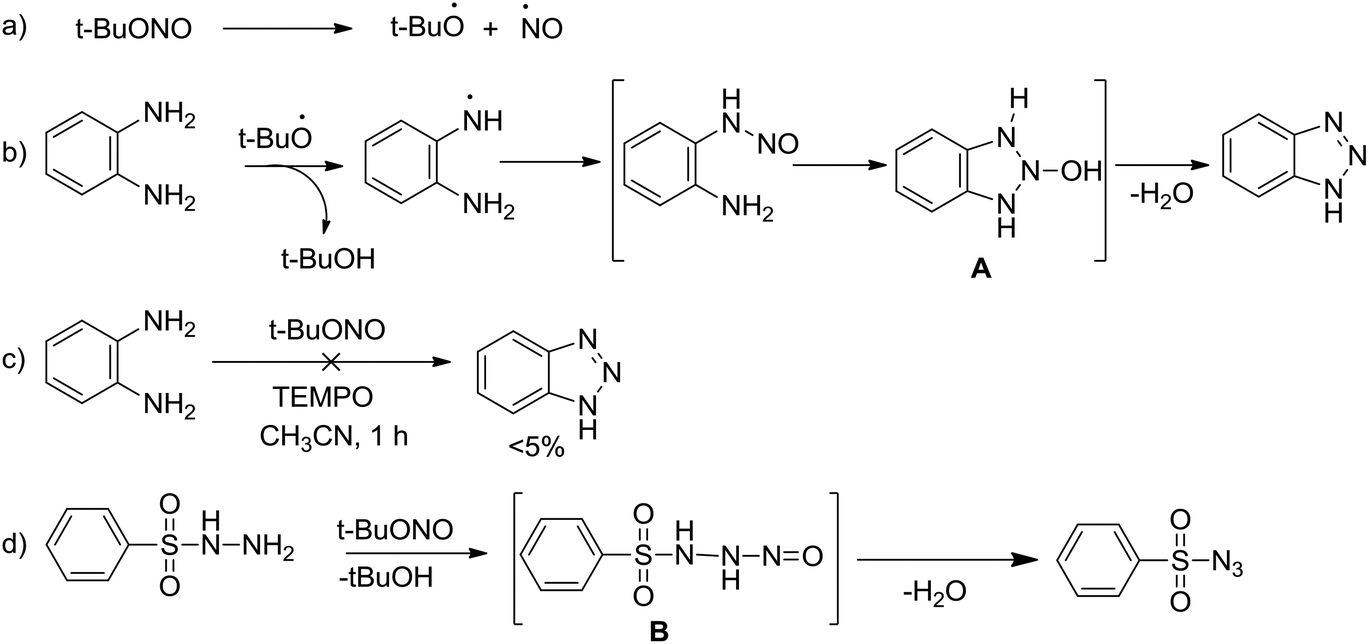
tert -Butyl nitrite mediated nitrogen transfer reactions: synthesis of benzotriazoles and azides at room temperature - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB01950A

tert‐Butyl Nitrite (TBN), a Multitasking Reagent in Organic Synthesis - Dahiya - 2019 - Chemistry – An Asian Journal - Wiley Online Library

tert‐Butyl Nitrite (TBN), a Multitasking Reagent in Organic Synthesis - Dahiya - 2019 - Chemistry – An Asian Journal - Wiley Online Library

Mild and efficient TBAI-catalyzed synthesis of 1,2,3-benzotriazine-4-(3H)-ones from tert-butyl nitrite and 2-aminobenzamides under acid-free conditions - ScienceDirect
![Tert-butyl nitrite : a metal-free radical initiator for aerobic cleavage of benzylic C[double bond, length as m-dash]C bonds in compressed carbon dio ... - Green Chemistry (RSC Publishing) DOI:10.1039/C0GC00676A Tert-butyl nitrite : a metal-free radical initiator for aerobic cleavage of benzylic C[double bond, length as m-dash]C bonds in compressed carbon dio ... - Green Chemistry (RSC Publishing) DOI:10.1039/C0GC00676A](https://pubs.rsc.org/image/article/2011/GC/c0gc00676a/c0gc00676a-s3.gif)
Tert-butyl nitrite : a metal-free radical initiator for aerobic cleavage of benzylic C[double bond, length as m-dash]C bonds in compressed carbon dio ... - Green Chemistry (RSC Publishing) DOI:10.1039/C0GC00676A
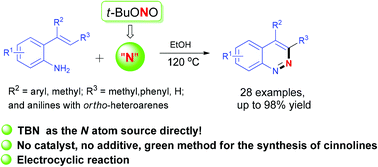
tert-Butyl nitrite (TBN) as the N atom source for the synthesis of substituted cinnolines with 2-vinylanilines and a relevant mechanism was studied - Organic & Biomolecular Chemistry (RSC Publishing)

tert‐Butyl Nitrite (TBN), a Multitasking Reagent in Organic Synthesis - Dahiya - 2019 - Chemistry – An Asian Journal - Wiley Online Library
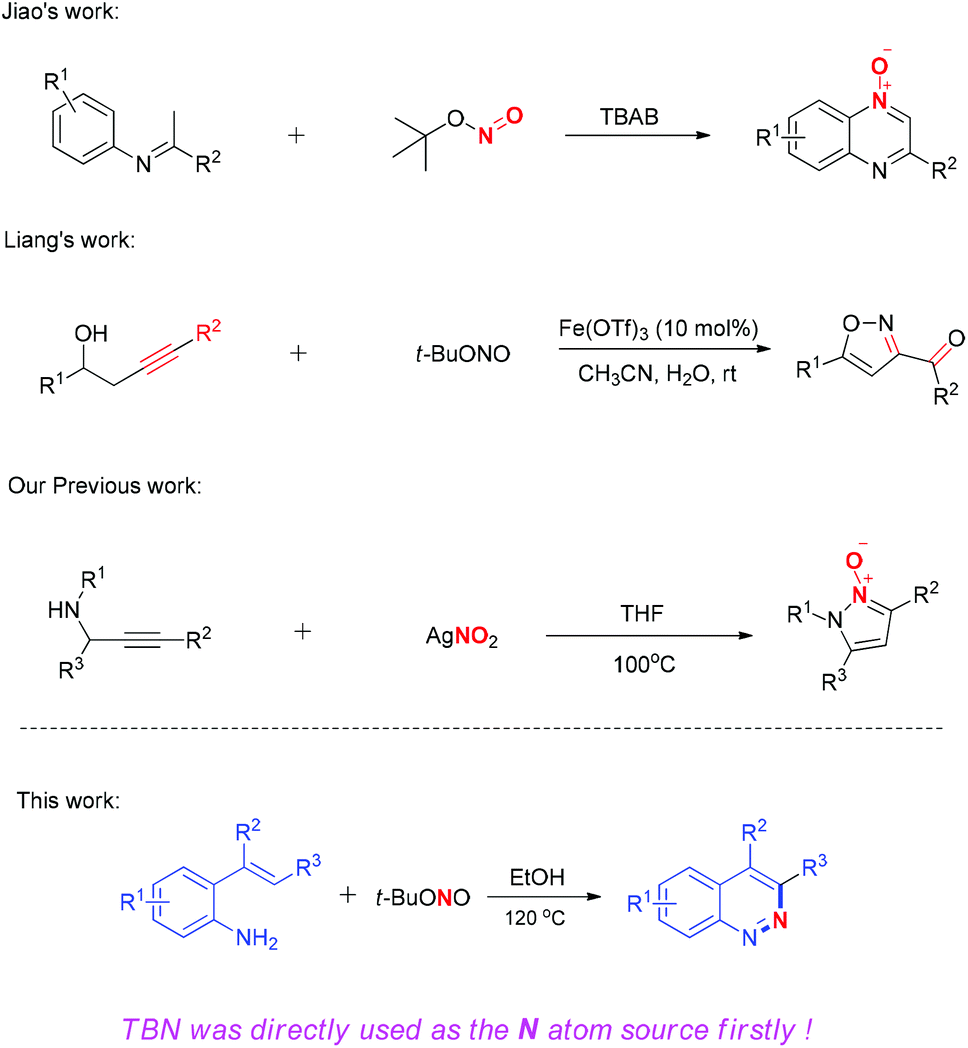
tert -Butyl nitrite (TBN) as the N atom source for the synthesis of substituted cinnolines with 2-vinylanilines and a relevant mechanism was studied ... - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB01553D

tert-Butyl nitrite induced radical dimerization of primary thioamides and selenoamides at room temperature - ScienceDirect

tert -Butyl nitrite-mediated vicinal sulfoximation of alkenes with sulfinic acids: a highly efficient approach toward α-sulfonyl ketoximes - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C6QO00535G












