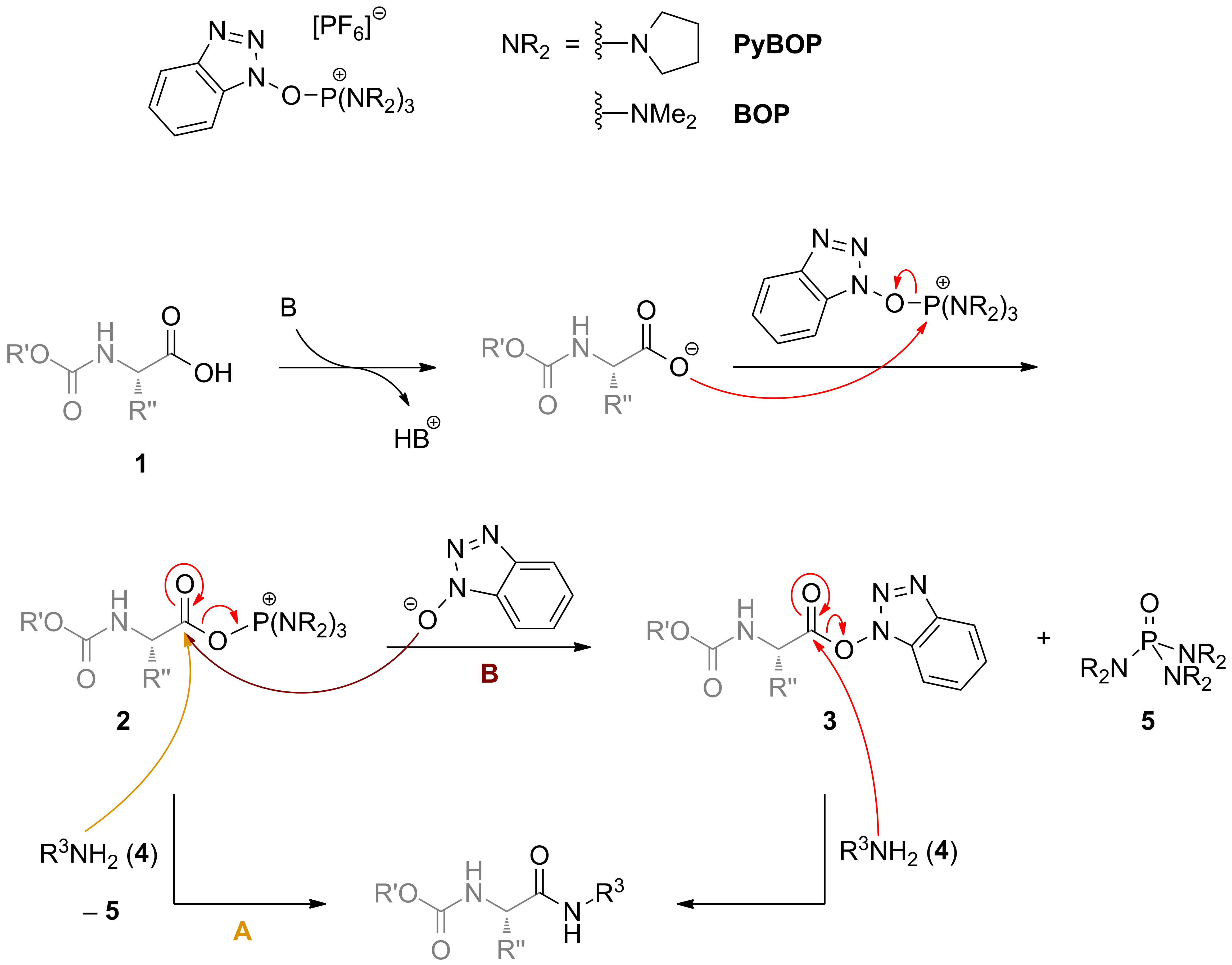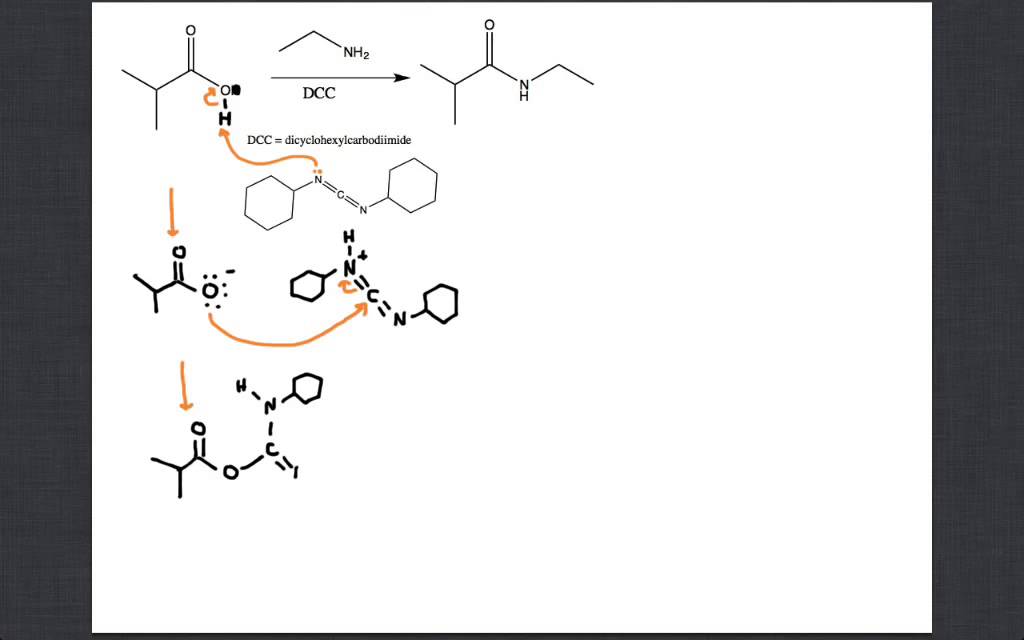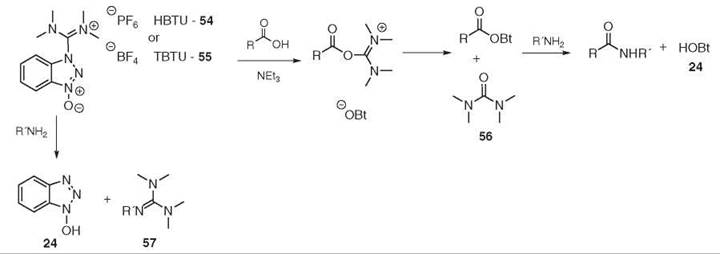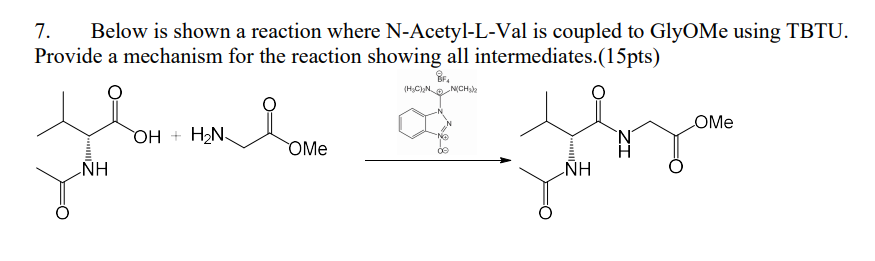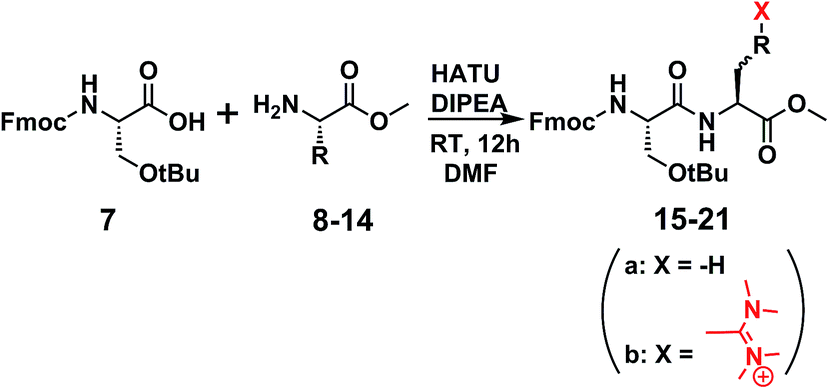
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates - RSC Advances (RSC Publishing) DOI:10.1039/C7RA06655D
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates
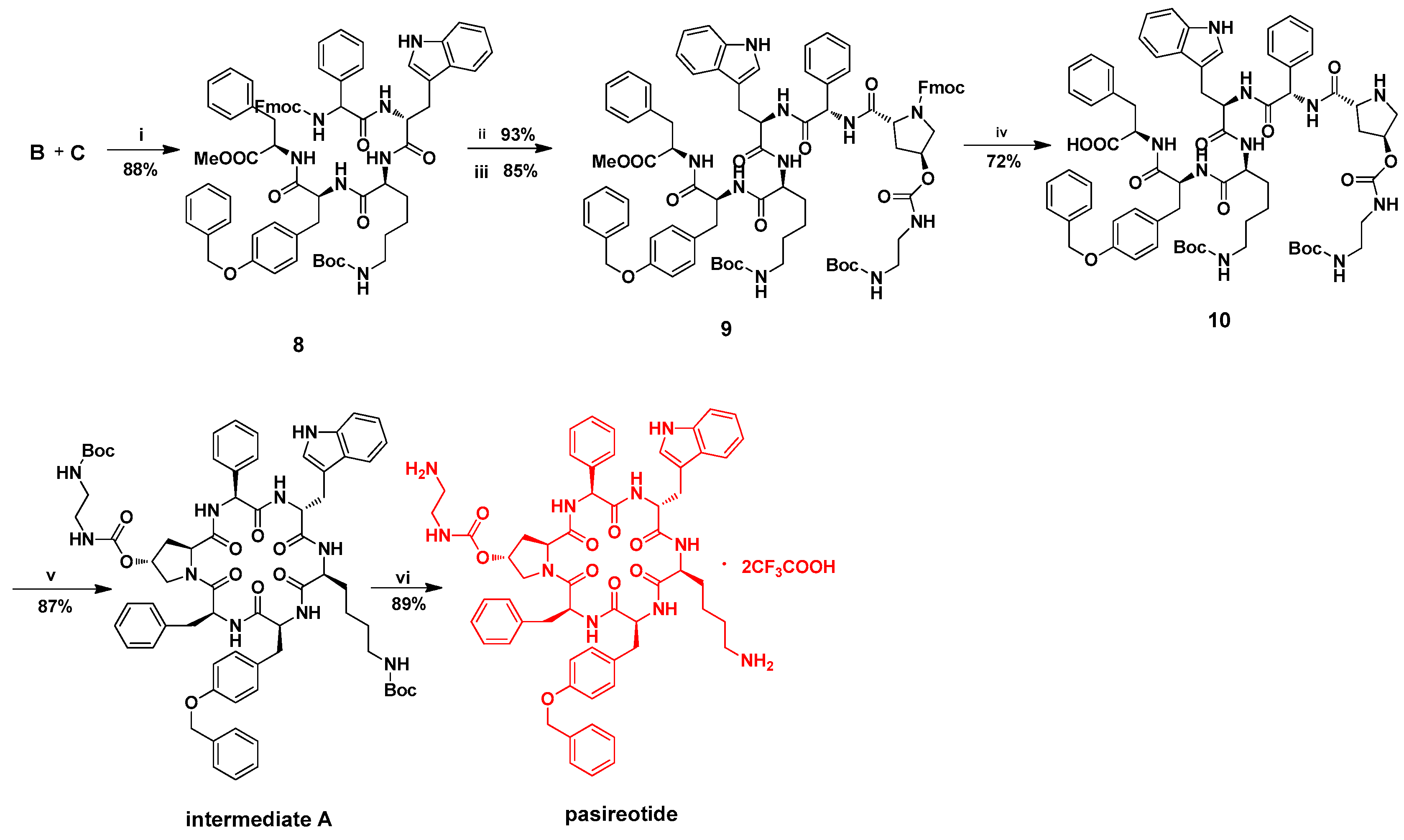
Molecules | Free Full-Text | A Practical and Total Synthesis of Pasireotide: Synthesis of Cyclic Hexapeptide via a Three-Component Condensation | HTML

Solid-Phase Peptide Synthesis of Dipeptide (Histidine-β-Alanine) as a Chelating Agent by Using Trityl Chloride Resin, for Removal of Al3+, Cu2+, Hg2+ and Pb2+: Experimental and Theoretical Study
DEPBT as an efficient coupling reagent for amide bond formation with remarkable resistance to racemization<link href='#fn
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates

DEPBT as an efficient coupling reagent for amide bond formation with remarkable resistance to racemization - Ye - 2005 - Peptide Science - Wiley Online Library

Scheme 2 | An efficient 2-(1 H -benzotriazole-1-yl)-1,1,3,3-tetramethyluronium tetrafluoroborate (TBTU)-mediated synthesis of 5-(trifluoromethyl)- N -alkyl-1-(3-phenylisoquinoline-1-yl)-1 H -pyrazole-4-carboxamides | SpringerLink