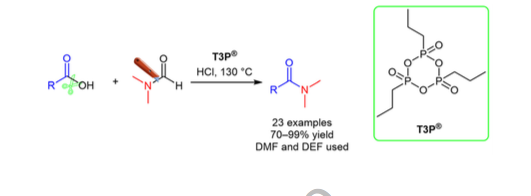
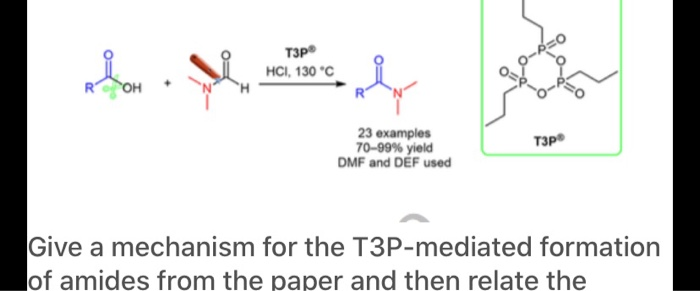
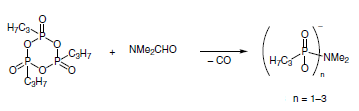General and Scalable Amide Bond Formation with Epimerization-Prone Substrates Using T3P and Pyridine

Formamide catalyzed activation of carboxylic acids - versatile and cost-efficient amidation and esterification. - Abstract - Europe PMC

Plausible mechanism of the T3P-DMSO mediated synthesis of title compounds. | Download Scientific Diagram
![PDF] Propanephosphonic acid anhydride (T3P). A benign reagent for diverse applications inclusive of large-scale synthesis | Semantic Scholar PDF] Propanephosphonic acid anhydride (T3P). A benign reagent for diverse applications inclusive of large-scale synthesis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e8bbf01e57f2f244366708ff0fb00545bb6ceab7/22-Figure18-1.png)
PDF] Propanephosphonic acid anhydride (T3P). A benign reagent for diverse applications inclusive of large-scale synthesis | Semantic Scholar

Study on the propylphosphonic anhydride (T3P®) mediated Ugi-type three-component reaction. Efficient synthesis of an α-amino amide library - ScienceDirect

An efficient catalytic method for the Friedländer annulation mediated by peptide coupling agent propylphosphonic anhydride (T3P®) - ScienceDirect
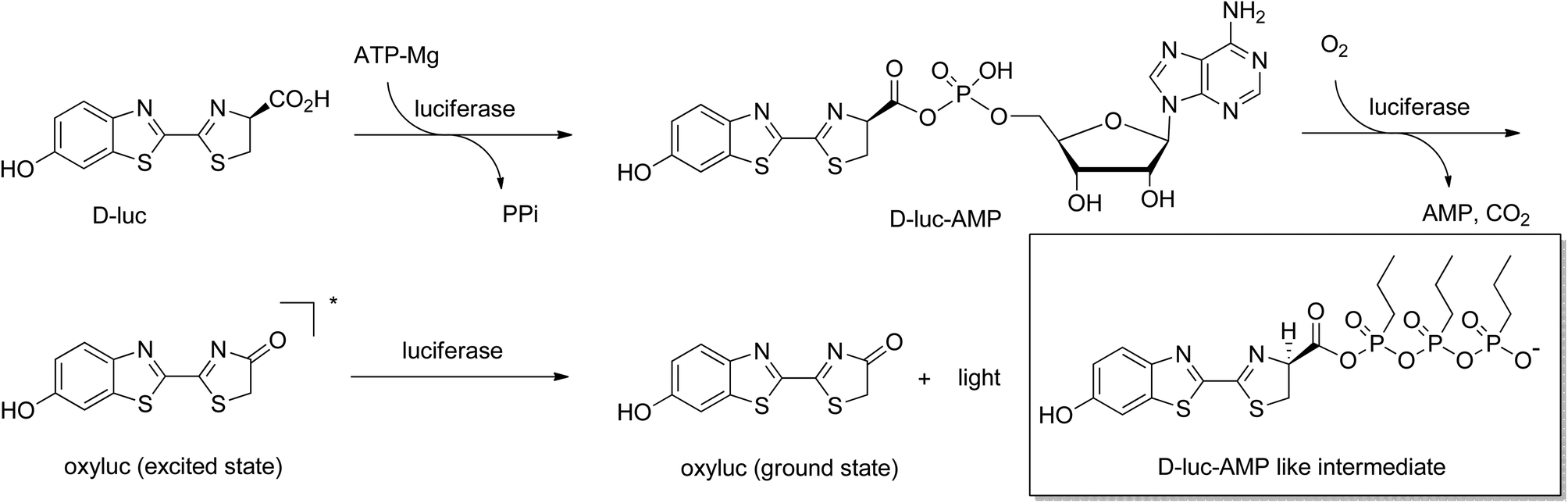
A firefly inspired one-pot chemiluminescence system using n -propylphosphonic anhydride (T3P) - Photochemical & Photobiological Sciences (RSC Publishing) DOI:10.1039/C4PP00250D
![PDF] Propanephosphonic acid anhydride (T3P). A benign reagent for diverse applications inclusive of large-scale synthesis | Semantic Scholar PDF] Propanephosphonic acid anhydride (T3P). A benign reagent for diverse applications inclusive of large-scale synthesis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e8bbf01e57f2f244366708ff0fb00545bb6ceab7/3-Figure2-1.png)
PDF] Propanephosphonic acid anhydride (T3P). A benign reagent for diverse applications inclusive of large-scale synthesis | Semantic Scholar

Plausible mechanism of the T3P-DMSO mediated synthesis of title compounds. | Download Scientific Diagram

T3P®-DMSO mediated one pot cascade protocol for the synthesis of 4-thiazolidinones from alcohols - ScienceDirect

Propylphosphonic anhydride (T3P®): an efficient reagent for the one-pot synthesis of 1,2,4-oxadiazoles, 1,3,4-oxadiazoles, and 1,3,4-thiadiazoles - ScienceDirect
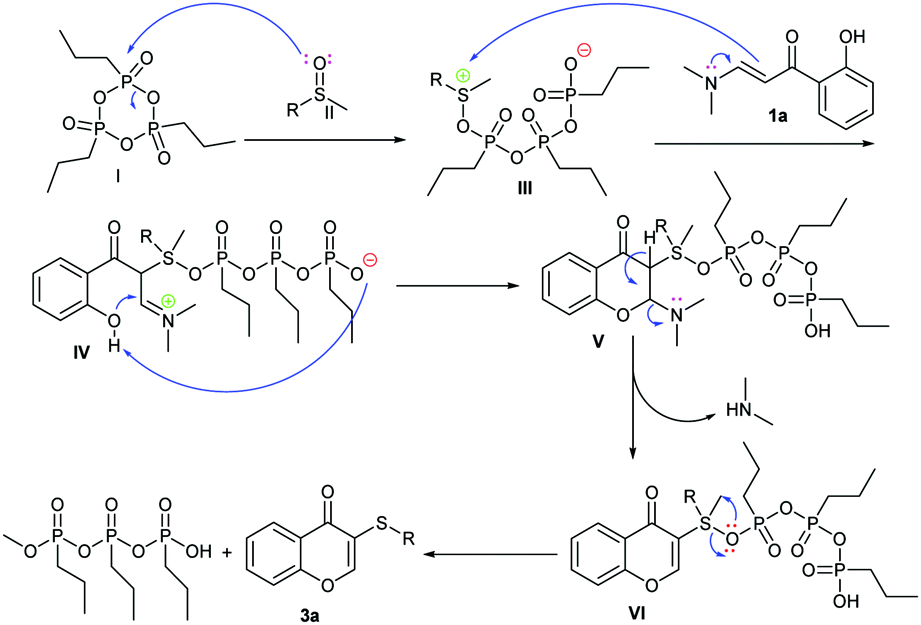
T3P® mediated domino C(sp 2 )–H sulfenylation/annulation of enaminones and methylsulfinyls for the synthesis of chromone thioether derivatives - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ05554H

T3P®‐Mediated One‐Pot Synthesis of Bis(α‐aminophosphonates) - Milen - 2014 - Heteroatom Chemistry - Wiley Online Library

Propylphosphonic anhydride (T3P®): an efficient reagent for the one-pot synthesis of 1,2,4-oxadiazoles, 1,3,4-oxadiazoles, and 1,3,4-thiadiazoles - ScienceDirect
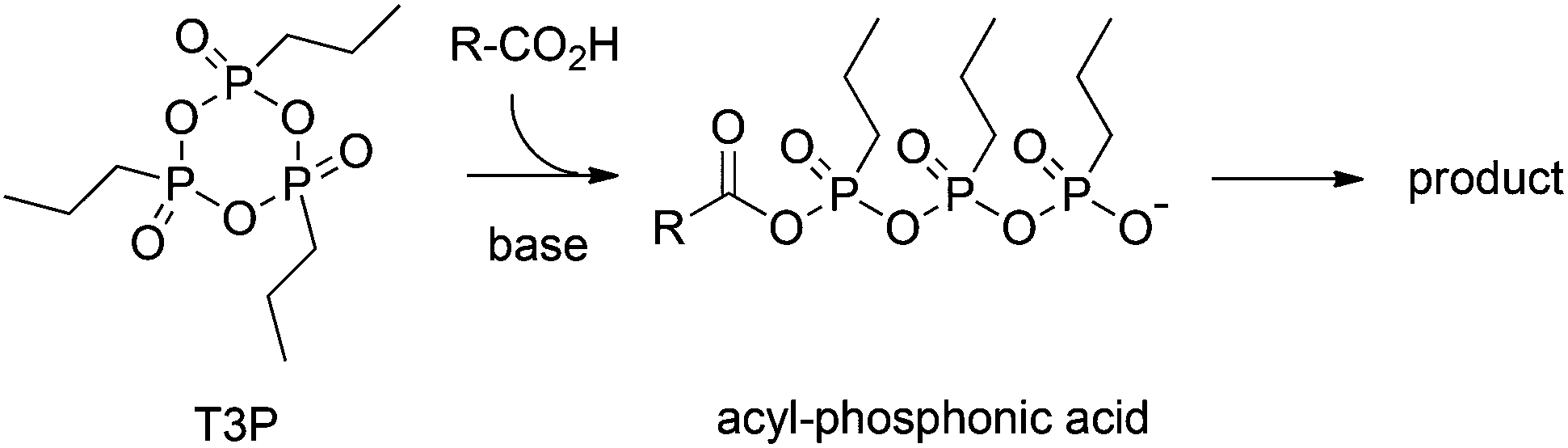
A firefly inspired one-pot chemiluminescence system using n -propylphosphonic anhydride (T3P) - Photochemical & Photobiological Sciences (RSC Publishing) DOI:10.1039/C4PP00250D

A Convenient Microwave‐Assisted Propylphosphonic Anhydride (T3P®) Mediated One‐Pot Pyrazolone Synthesis - Desroses - 2013 - European Journal of Organic Chemistry - Wiley Online Library

Theoretical Calculations on the Mechanism of the T3P<sup>®</sup>-Promoted Esterification and Amidation of Phosphinic Acids | Bentham Science
![Propylphosphonic anhydride (T3P®)-mediated three-component synthesis of hexahydrodibenzo[b,e][1,4]diazepin-1-one derivatives | SpringerLink Propylphosphonic anhydride (T3P®)-mediated three-component synthesis of hexahydrodibenzo[b,e][1,4]diazepin-1-one derivatives | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs10593-020-02650-3/MediaObjects/10593_2020_2650_Sch1_HTML.jpg)
Propylphosphonic anhydride (T3P®)-mediated three-component synthesis of hexahydrodibenzo[b,e][1,4]diazepin-1-one derivatives | SpringerLink