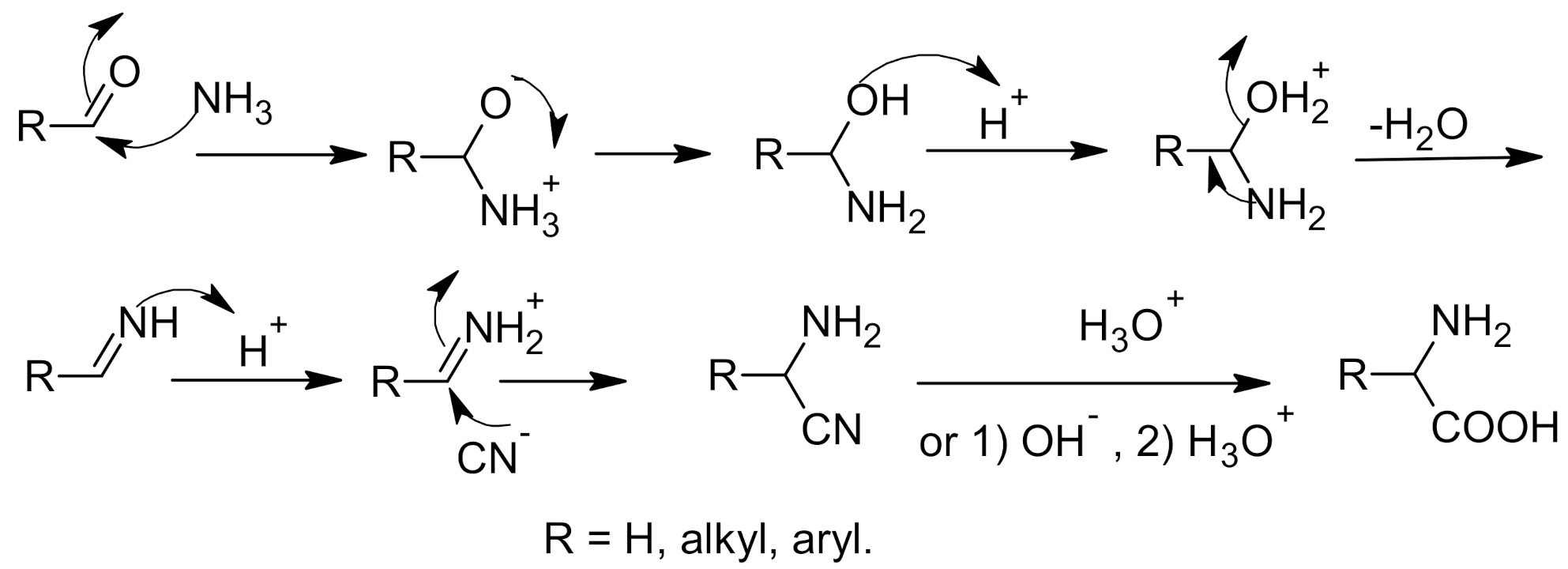
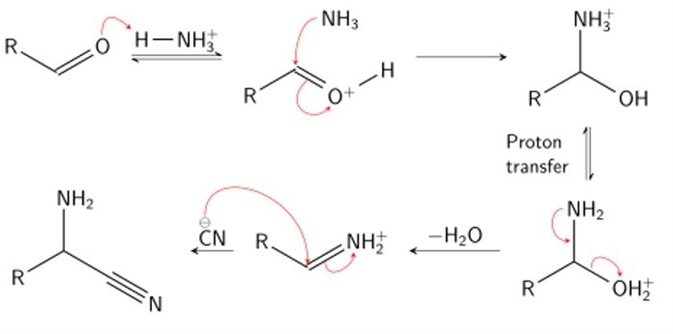
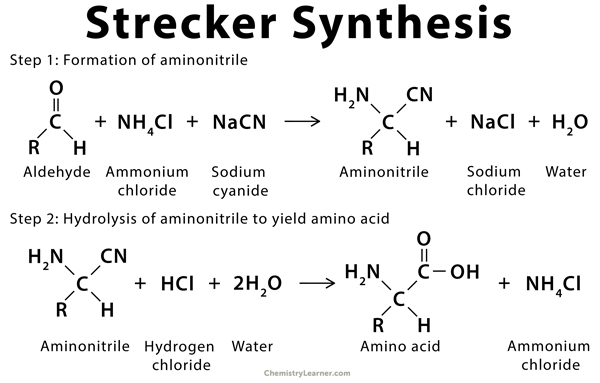
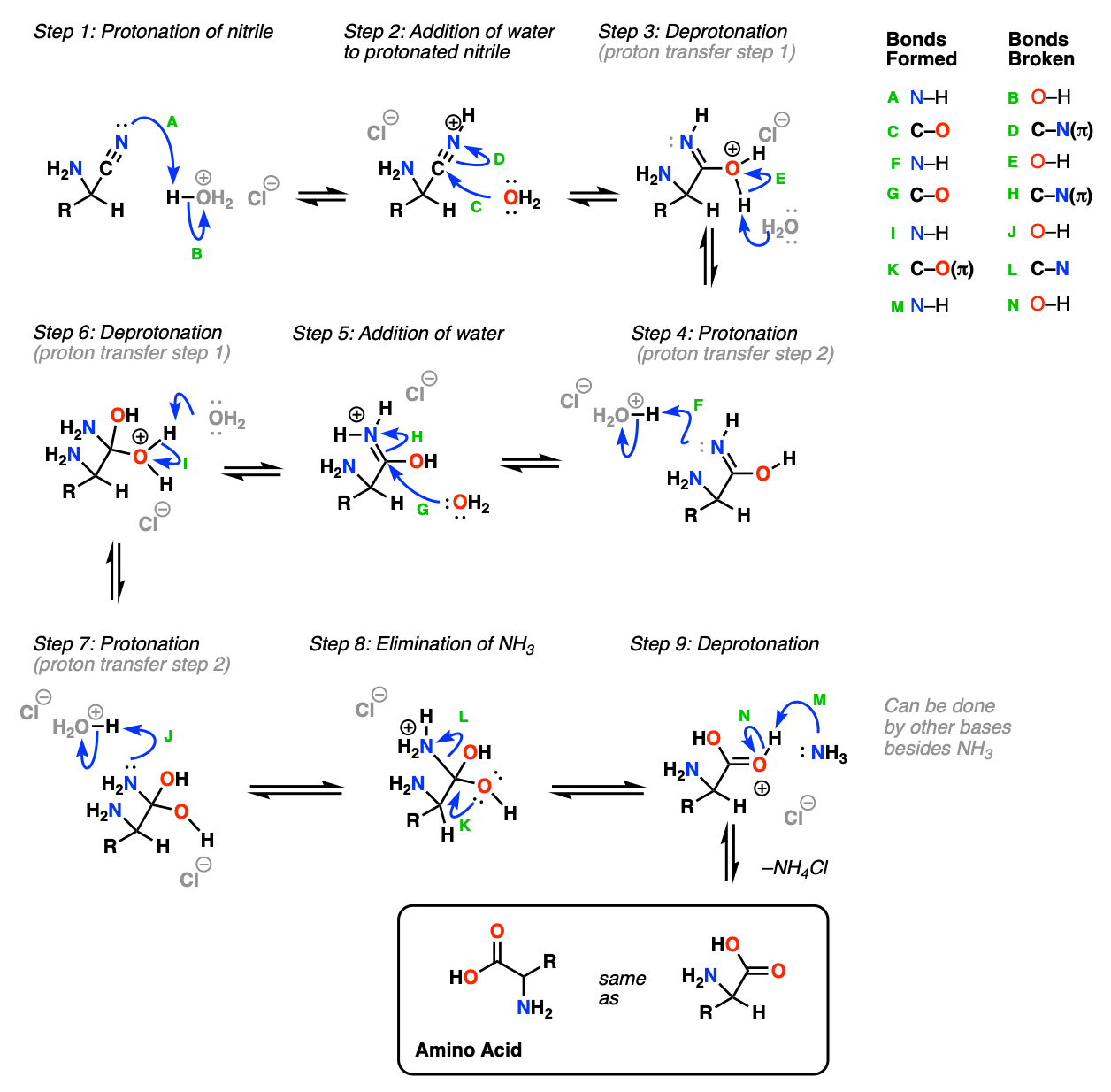
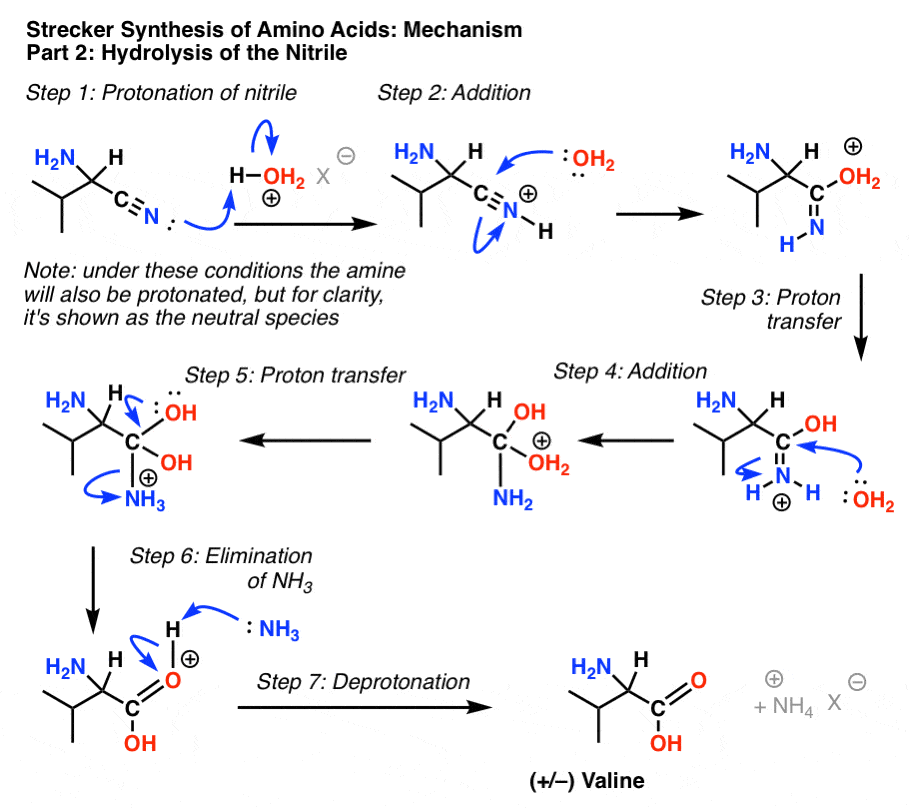
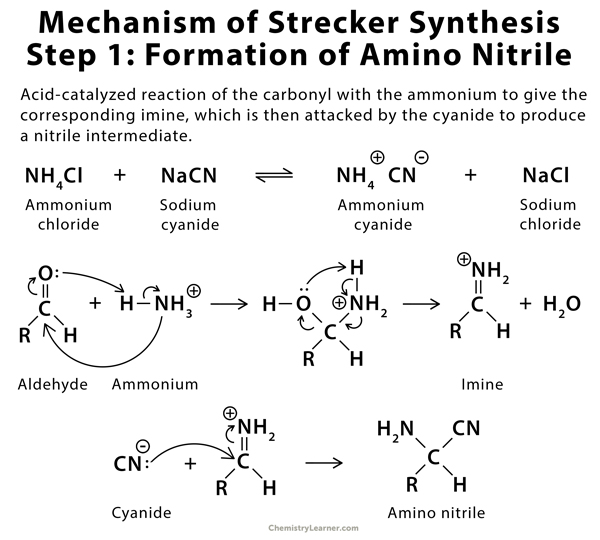
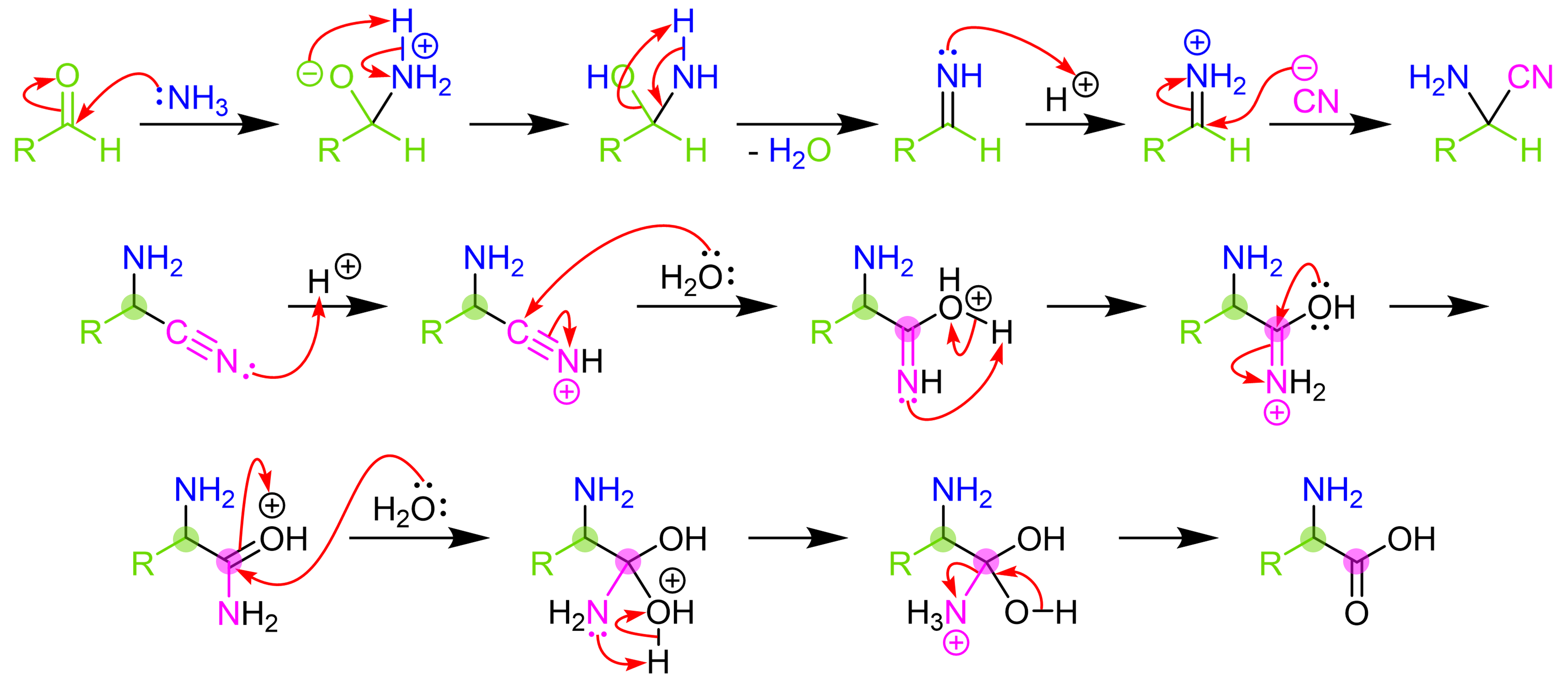
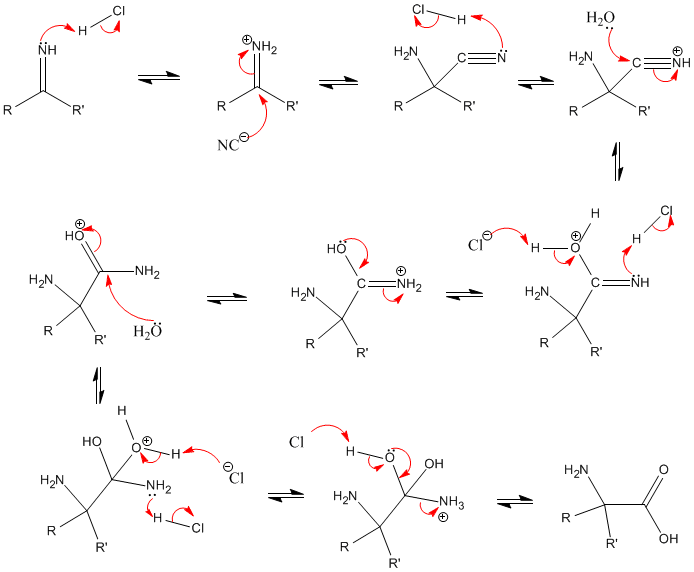
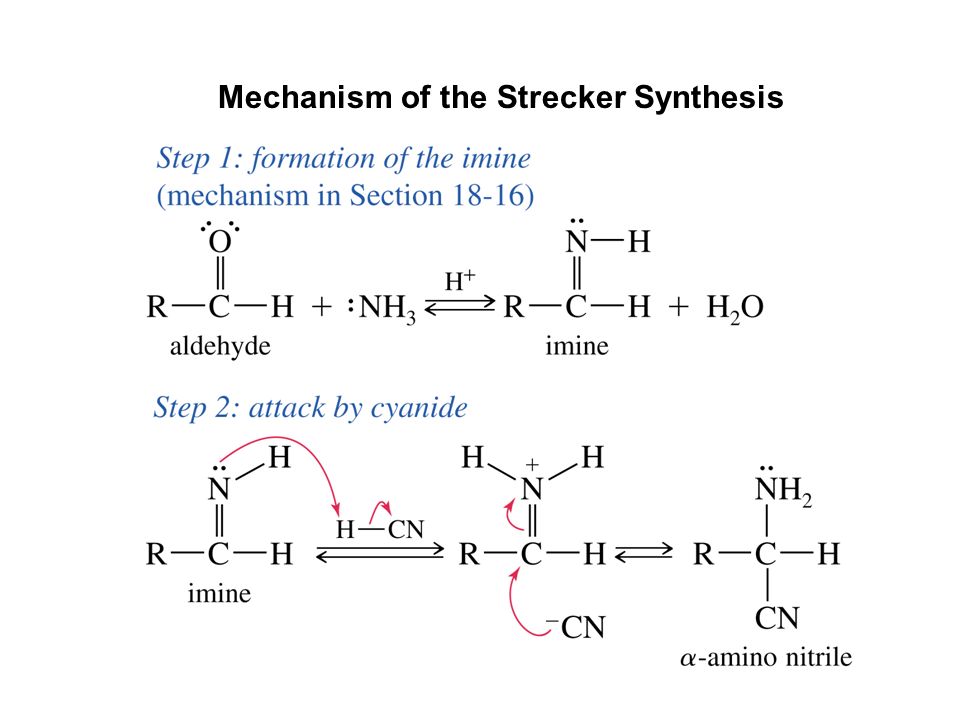
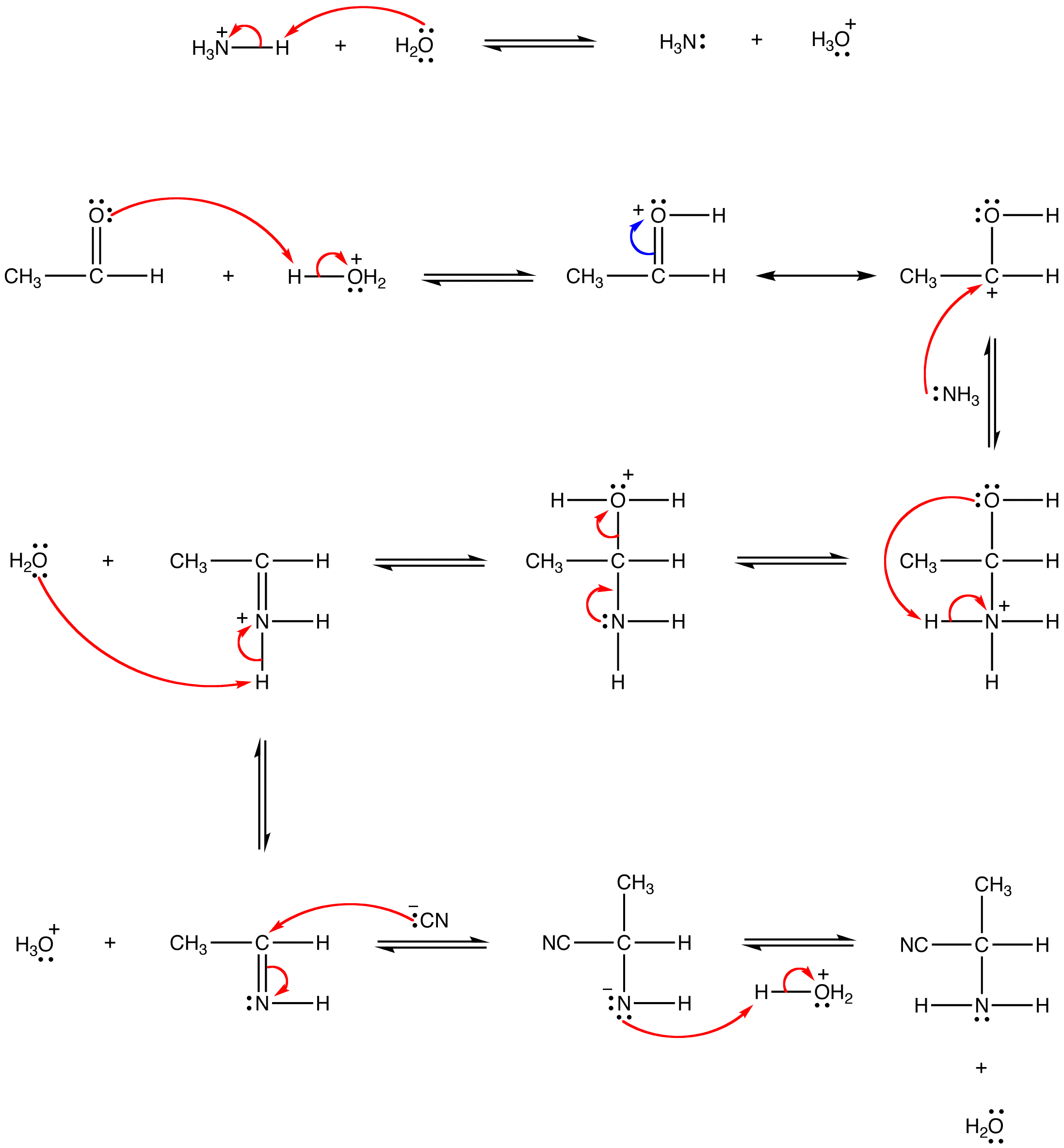
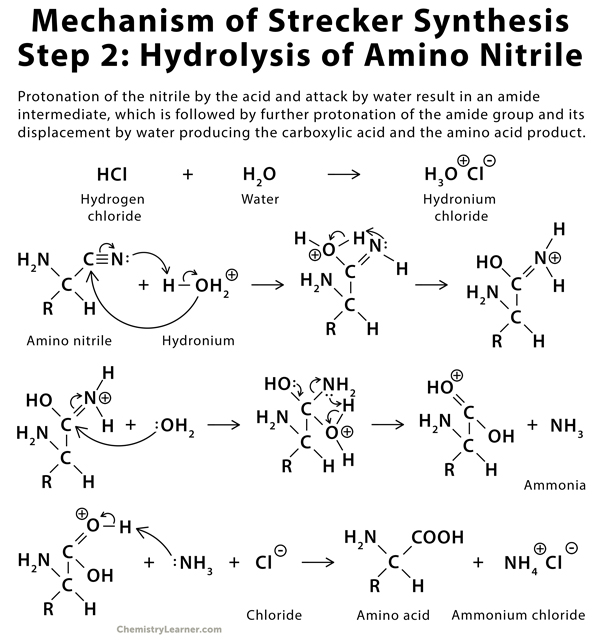
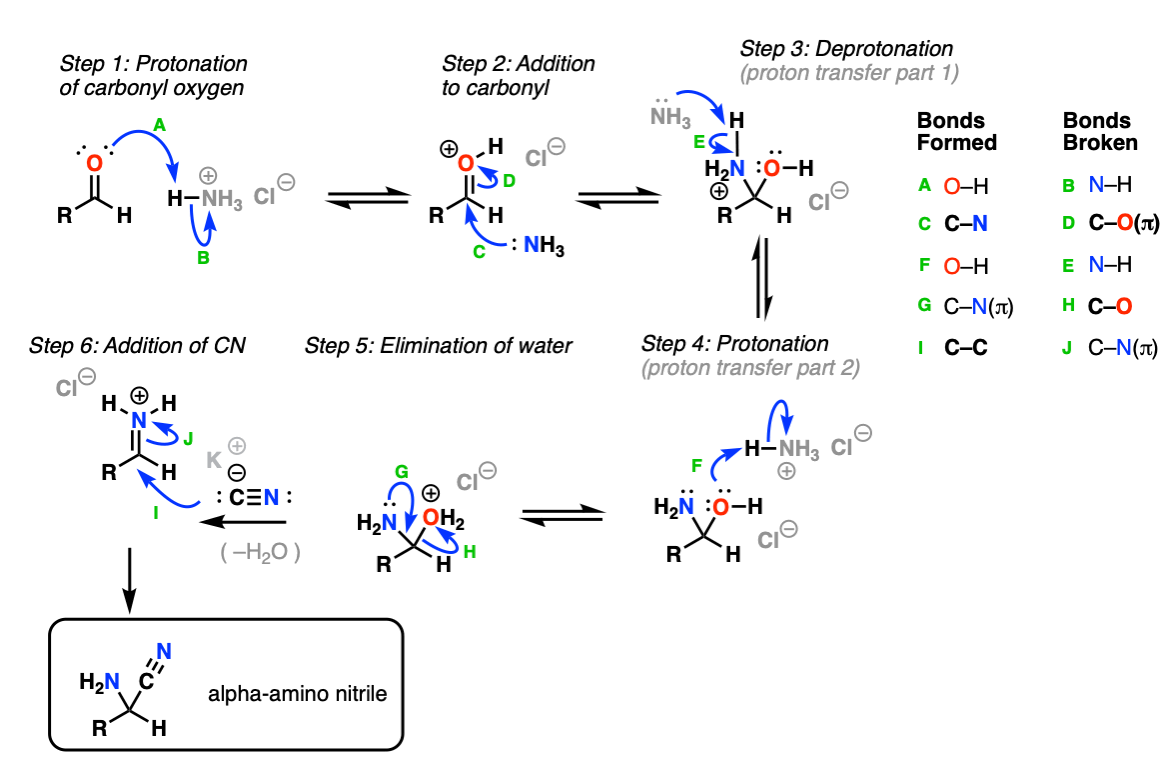
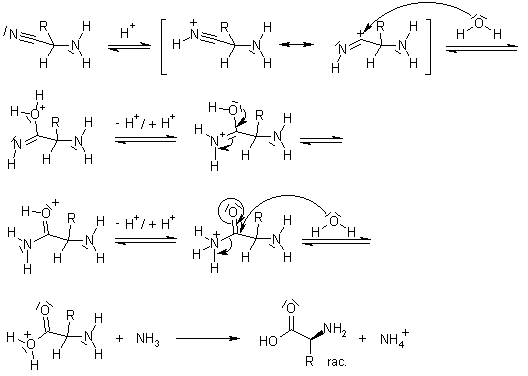Synthesis of α-Amino Acids - Nitrogen- and Phosphorus-Containing Compounds - MCAT Organic Chemistry Review

Strecker reaction and α-amino nitriles: Recent advances in their chemistry, synthesis, and biological properties - ScienceDirect
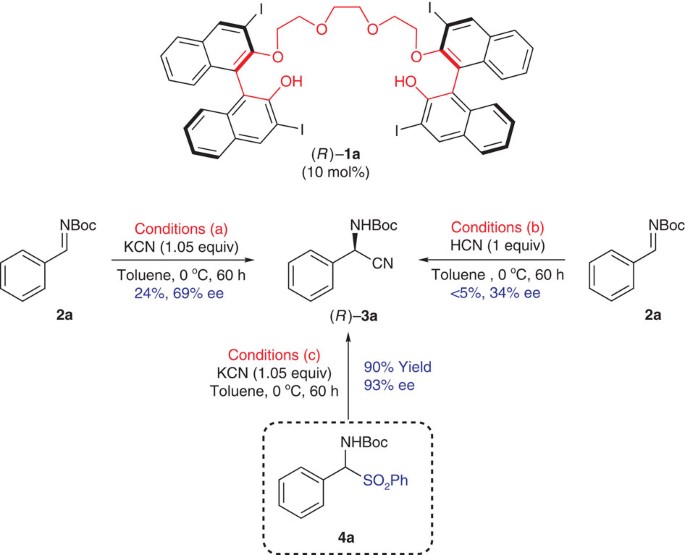
Scalable organocatalytic asymmetric Strecker reactions catalysed by a chiral cyanide generator | Nature Communications
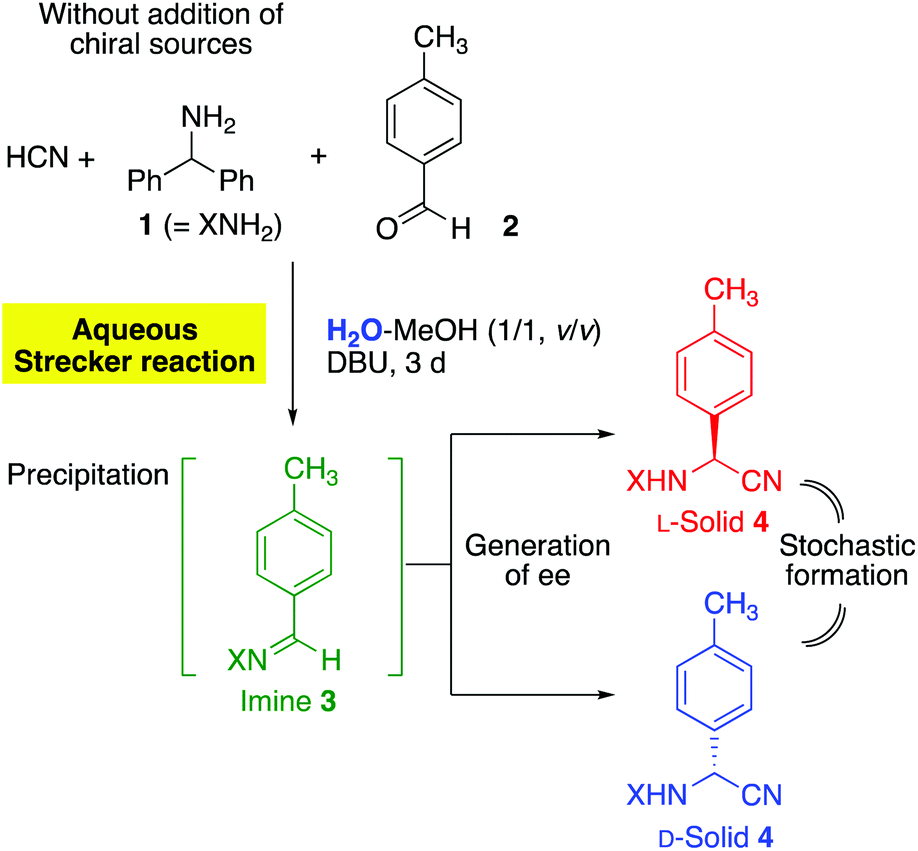
Absolute asymmetric Strecker synthesis in a mixed aqueous medium: reliable access to enantioenriched α-aminonitrile - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB03092H

Asymmetric Strecker Reaction with Chiral Amines: a Catalyst‐Free Protocol Using Acetone Cyanohydrin in Water - Pori - 2013 - European Journal of Organic Chemistry - Wiley Online Library
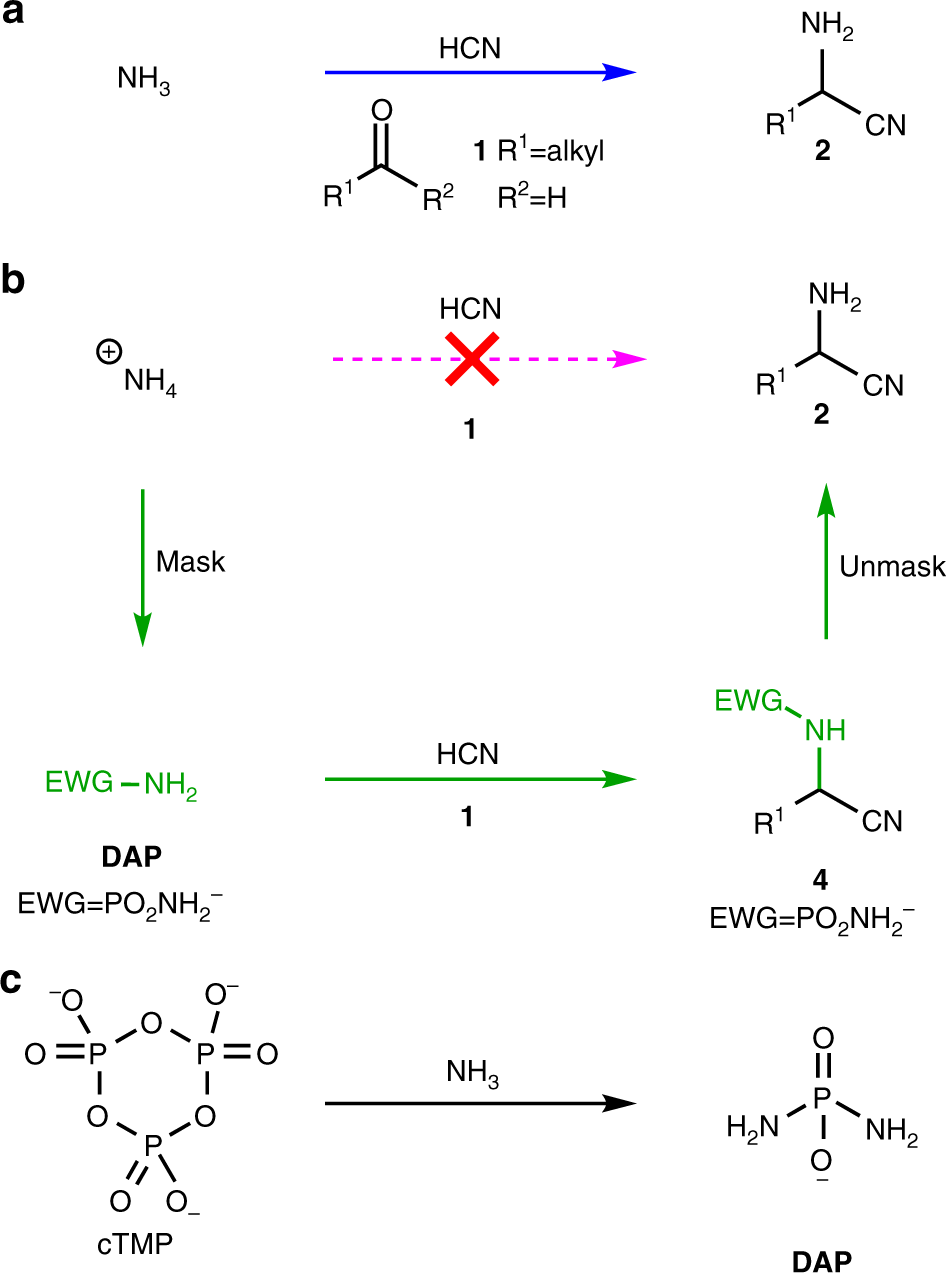
Selective prebiotic synthesis of phosphoroaminonitriles and aminothioamides in neutral water | Communications Chemistry