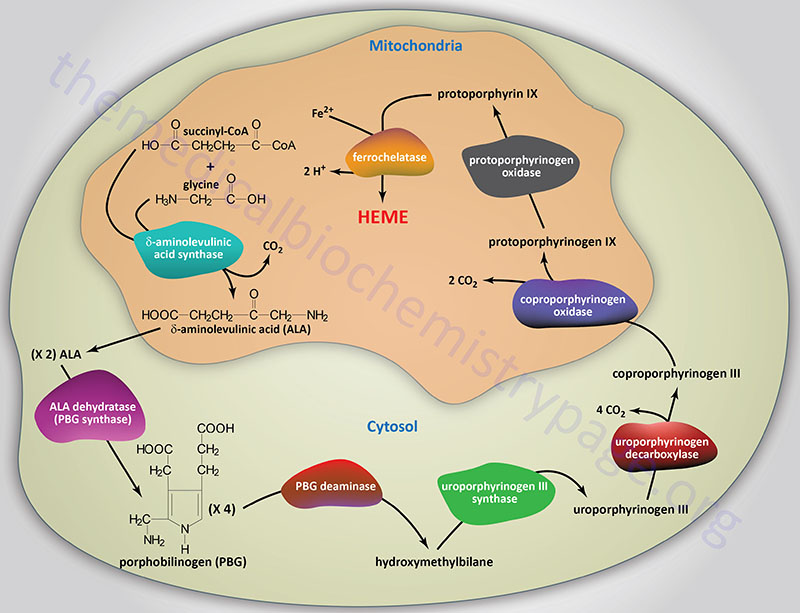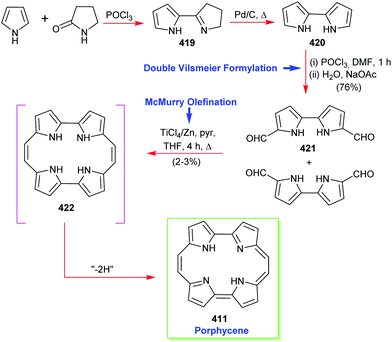
Classic highlights in porphyrin and porphyrinoid total synthesis and biosynthesis - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C7CS00719A

Synthesis and Functionalization of Porphyrins through Organometallic Methodologies | Chemical Reviews

Molecules | Free Full-Text | Synthesis, Characterization and Photodynamic Activity against Bladder Cancer Cells of Novel Triazole-Porphyrin Derivatives | HTML

Synthesis and characterization of free base and metal porphyrins and their interaction with CdTe QDs - ScienceDirect
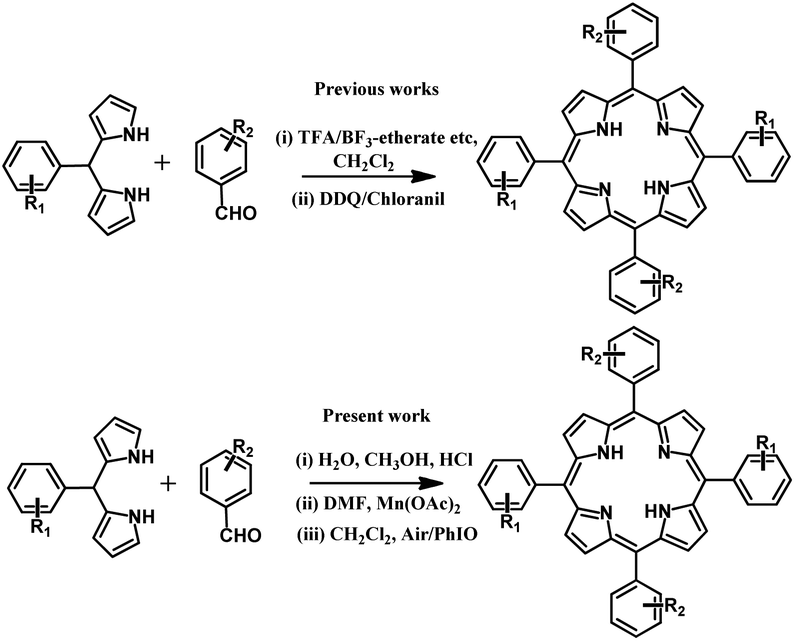
A new synthesis of porphyrins via a putative trans -manganese( iv )-dihydroxide intermediate - Dalton Transactions (RSC Publishing) DOI:10.1039/C9DT03573G

Acid Activation and Chemical Oxidation in the Synthesis of meso‐ Tetraphenylporphyrin using a Mixed‐Solvent System - Sun - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library
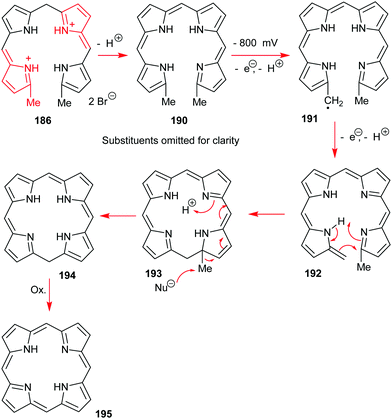
Classic highlights in porphyrin and porphyrinoid total synthesis and biosynthesis - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C7CS00719A