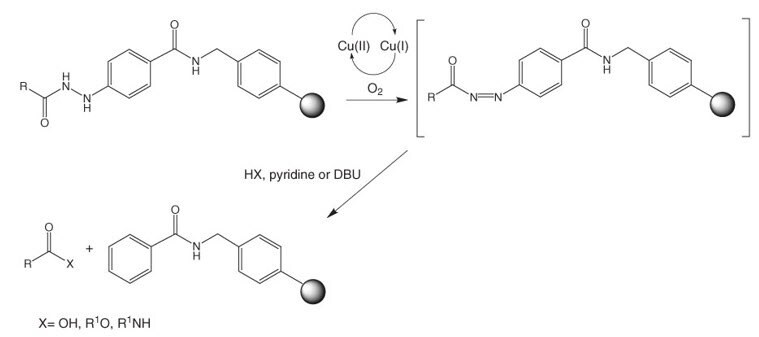
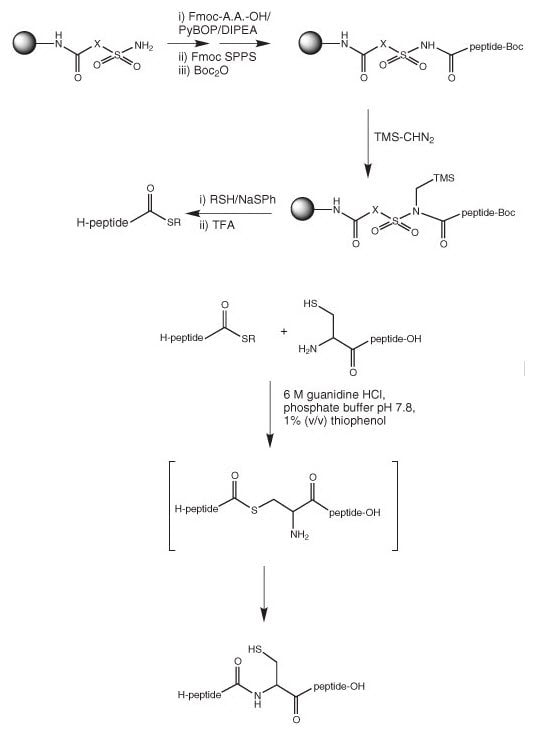
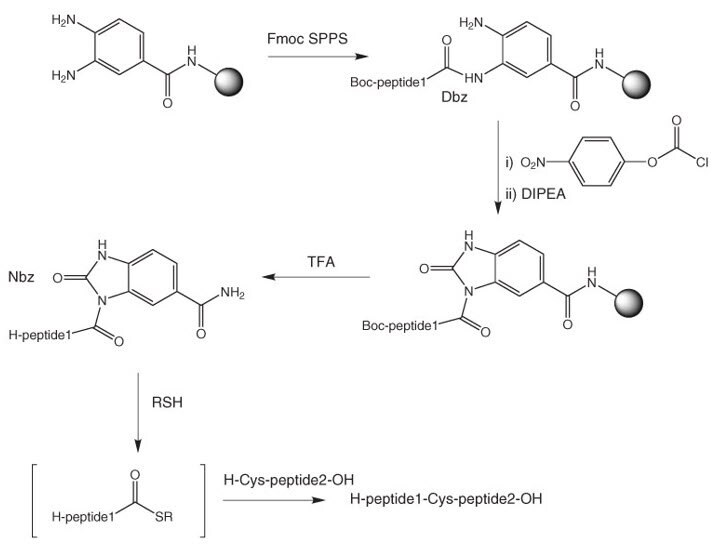
Development of a Backbone Cyclic Peptide Library as Potential Antiparasitic Therapeutics Using Microwave Irradiation | Protocol

Molecules | Free Full-Text | Deprotection Reagents in Fmoc Solid Phase Peptide Synthesis: Moving Away from Piperidine? | HTML
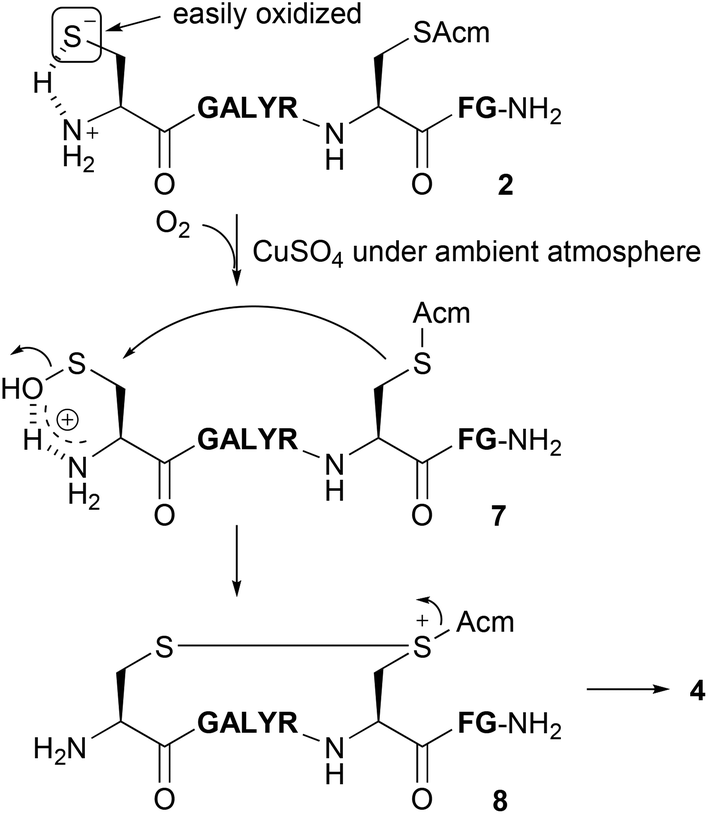
Deprotection of S -acetamidomethyl cysteine with copper( ii ) and 1,2-aminothiols under aerobic conditions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB01475C

Microwave‐assisted cleavage of Alloc and Allyl Ester protecting groups in solid phase peptide synthesis - Wilson - 2016 - Journal of Peptide Science - Wiley Online Library

deprotection of various amino acids in the presence of other labile groups | Download Scientific Diagram

Fmoc-Arg(Pbf)-OH, CAS# 154445-77-9, N-α-Fmoc-N-g-(2,2,4,6,7-pentamethyldihydrobenzofuran-5-sulfonyl)-L-arginine - ChemPep Inc.

Deprotection kinetics for Fmoc-L-Leucine-OH and Fmoc-L-Arginine(Pbf)-OH... | Download Scientific Diagram

IJMS | Free Full-Text | Revisiting NO2 as Protecting Group of Arginine in Solid-Phase Peptide Synthesis | HTML
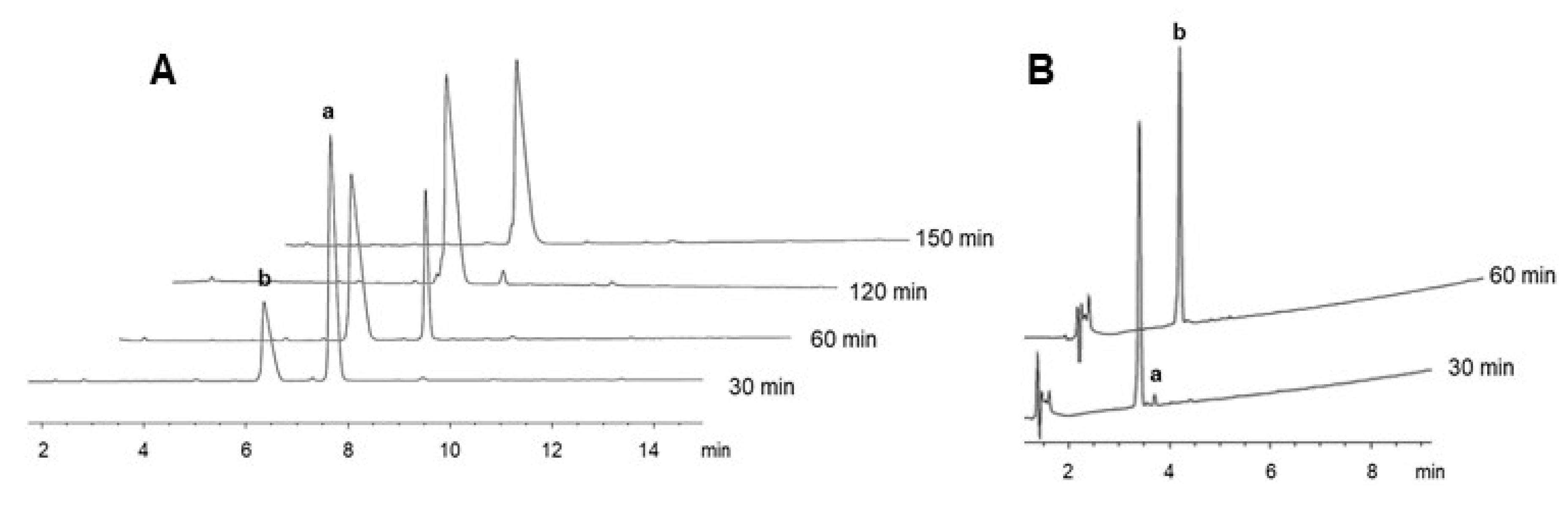
IJMS | Free Full-Text | Revisiting NO2 as Protecting Group of Arginine in Solid-Phase Peptide Synthesis | HTML

The 2,2,4,6,7-pentamethyldihydrobenzofuran-5-sulfonyl group (Pbf) as arginine side chain protectant - ScienceDirect

On‐resin conversion of Cys(Acm)‐containing peptides to their corresponding Cys(Scm) congeners - Mullen - 2010 - Journal of Peptide Science - Wiley Online Library

On‐resin conversion of Cys(Acm)‐containing peptides to their corresponding Cys(Scm) congeners - Mullen - 2010 - Journal of Peptide Science - Wiley Online Library

WO2014033466A1 - Method and compositions for removing acid-labile protecting groups - Google Patents

IJMS | Free Full-Text | Revisiting NO2 as Protecting Group of Arginine in Solid-Phase Peptide Synthesis | HTML