Studies on kinetics and thermodynamics of oxidation of 3,4,5-trimethoxy benzaldehyde, benzaldehyde and N,N-dimethylamino benzaldehyde by tetraethylammonium bromochromate in dimethyl formamide and acetic acid mixture - ScienceDirect
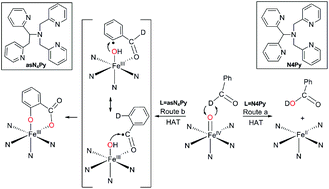
Steric control and the mechanism of benzaldehyde oxidation by polypyridyl oxoiron(iv) complexes: aromatic versus benzylic hydroxylation of aromatic aldehydes - Dalton Transactions (RSC Publishing)
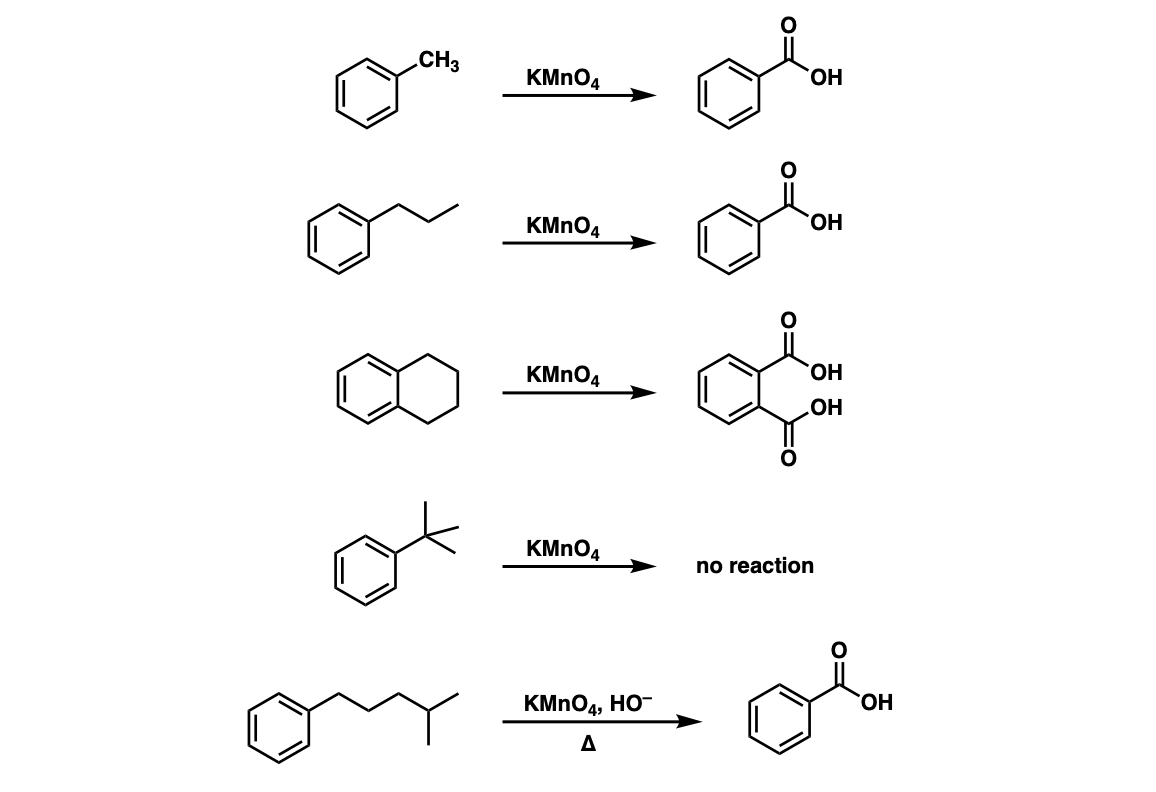
Oxidation of toluene to benzoic acid by O<sub>3</sub>/H<sub>2</sub>O<sub>2</sub> process enhanced usinghigh-gravity technology
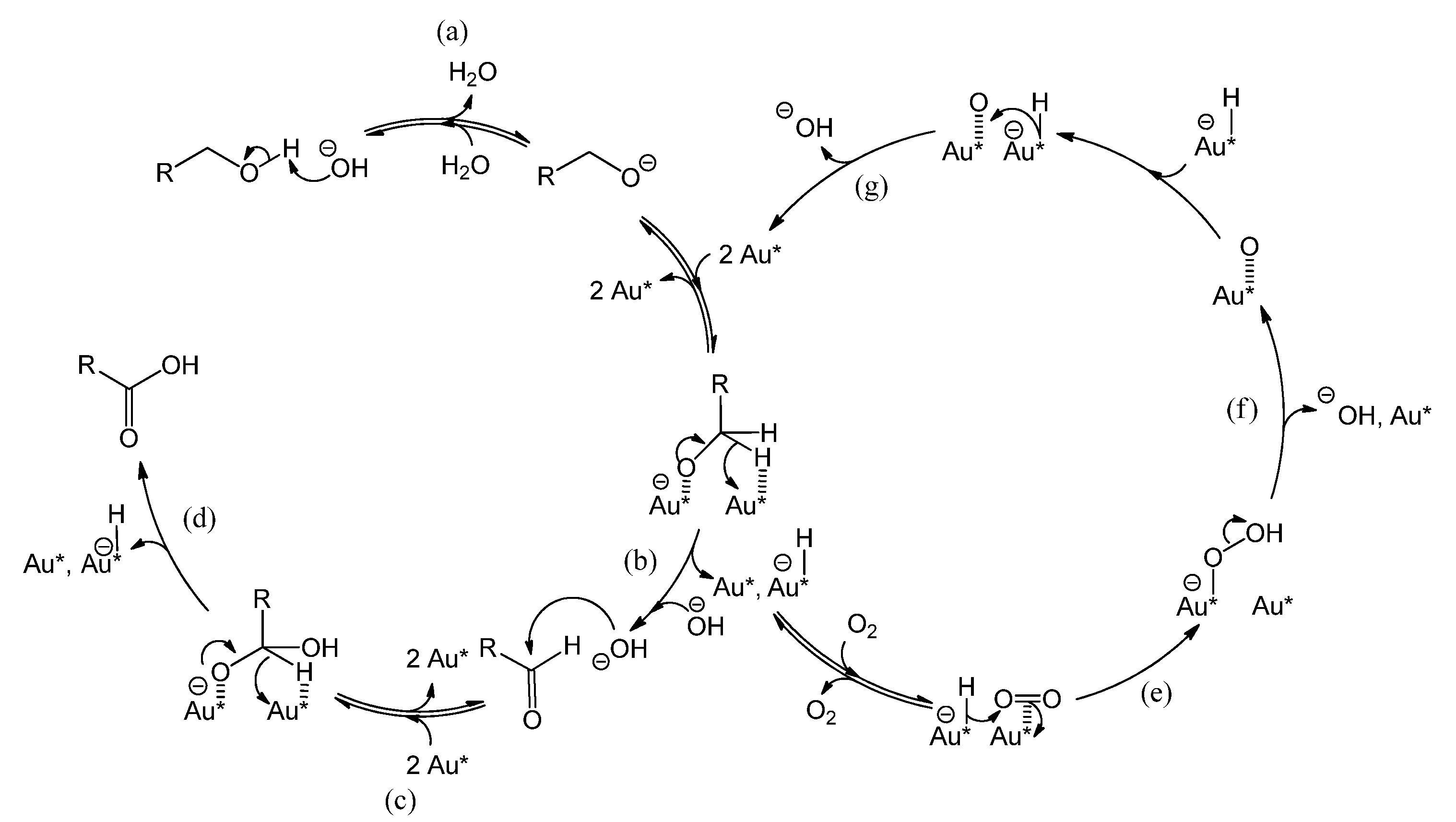
Catalysts | Free Full-Text | Inhibition of a Gold-Based Catalyst in Benzyl Alcohol Oxidation: Understanding and Remediation | HTML
![Special-selective C–H oxidation of toluene to benzaldehyde by a hybrid polyoxometalate photocatalyst including a rare [P6W48Fe6O180]30– anion-河南大学国际合作与交流处暨港澳台事务办公室 Special-selective C–H oxidation of toluene to benzaldehyde by a hybrid polyoxometalate photocatalyst including a rare [P6W48Fe6O180]30– anion-河南大学国际合作与交流处暨港澳台事务办公室](https://iao.henu.edu.cn/__local/B/93/88/7AA0FD387C42FB9C3337AAA9AD3_24BE5F8E_2C378.png)
Special-selective C–H oxidation of toluene to benzaldehyde by a hybrid polyoxometalate photocatalyst including a rare [P6W48Fe6O180]30– anion-河南大学国际合作与交流处暨港澳台事务办公室

Kinetics and Mechanism Modeling of Liquid-Phase Toluene Oxidation to Benzaldehyde Catalyzed by Mn–Mo Oxide | SpringerLink
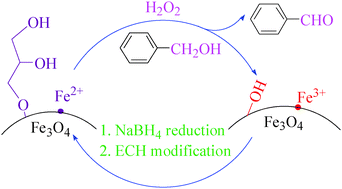
Selective oxidation of benzyl alcohol to benzaldehyde with H2O2 in water on epichlorohydrin-modified Fe3O4 microspheres - New Journal of Chemistry (RSC Publishing)
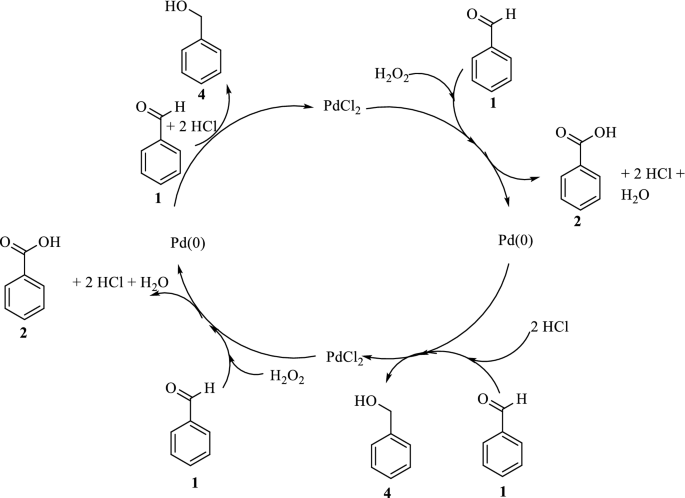
One-pot synthesis of benzaldehyde derivatives in PdCl2-catalyzed reactions with H2O2 in alcoholic solutions | SpringerLink

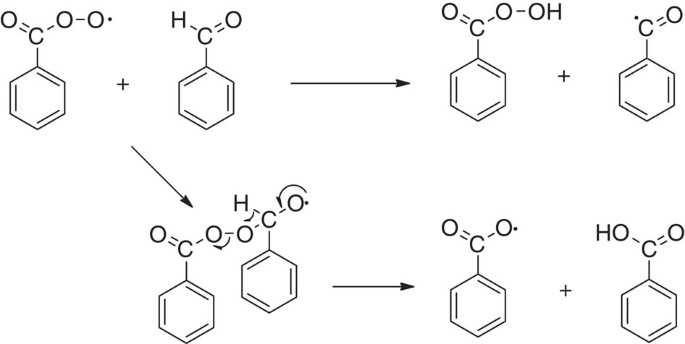
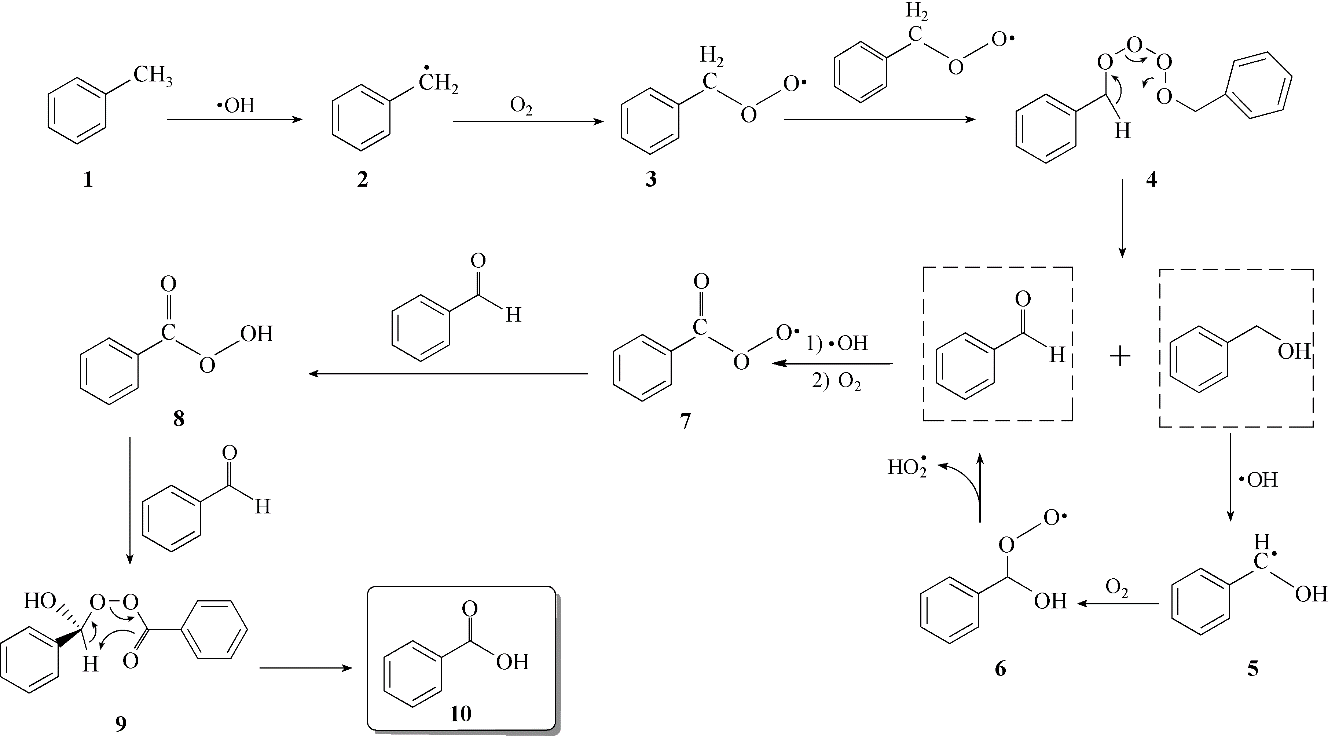
Figure 3 from The benzaldehyde oxidation paradox explained by the interception of peroxy radical by benzyl alcohol. | Semantic Scholar

Handy and highly efficient oxidation of benzylic alcohols to the benzaldehyde derivatives using heterogeneous Pd/AlO(OH) nanoparticles in solvent-free conditions | Scientific Reports

SciELO - Brasil - Accessing Basic Sites on Modified CoFe<sub>2</sub>O<sub>4</sub> Nanoparticles: Addressing the Selective Oxidation of Benzyl Alcohol and Unraveling the Au:Pd Ratio Effects by XPS Accessing Basic Sites on Modified CoFe<sub>2</sub>O<sub ...
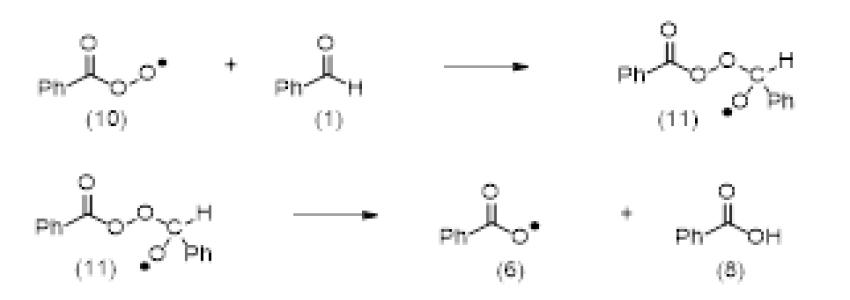
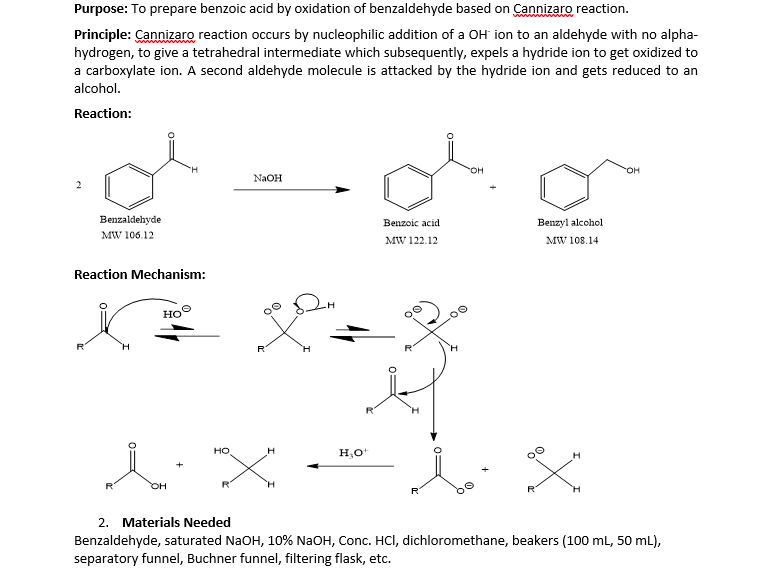
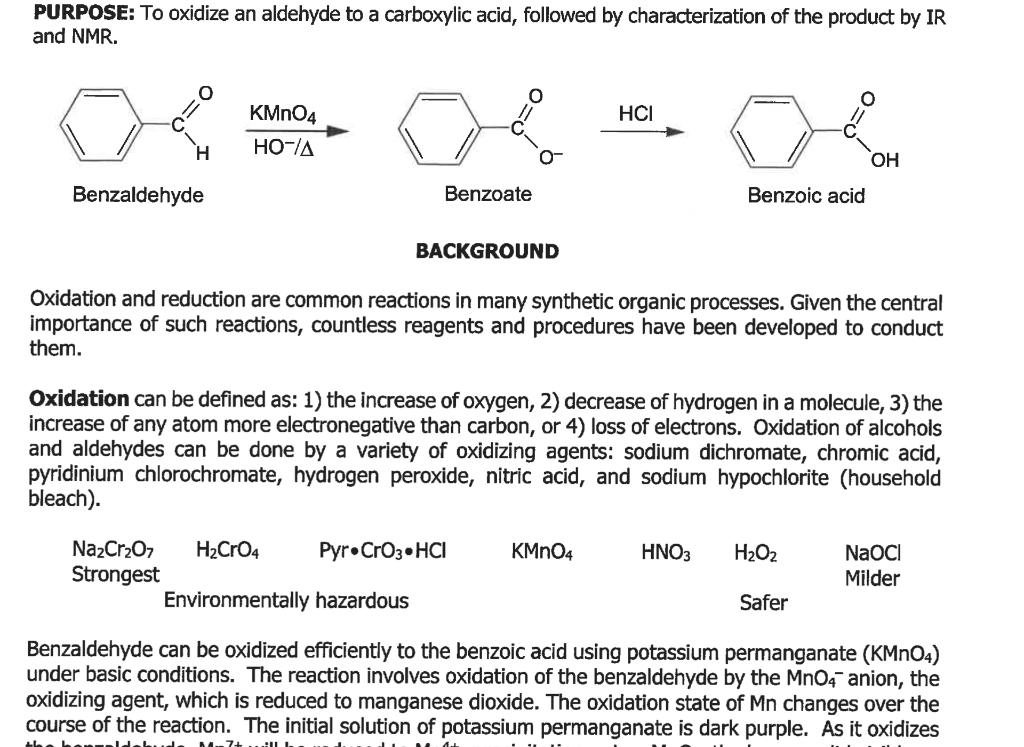
Seminary about (aut)oxidation of benzaldehyde F. Celerse Ludwig-Maximilians-Universität München, München, Allemagne, & Laboratoire de Chimie Théorique - UMR7616 UPMC & CNRS - Paris Mercredi 22 Mars 2017, 11h00 bibliothèque LCT, tour 12 ...