Catalytic reactions of oxetanes with protonic reagents and aprotic reagents leading to novel polymers - Kudo - 2007 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

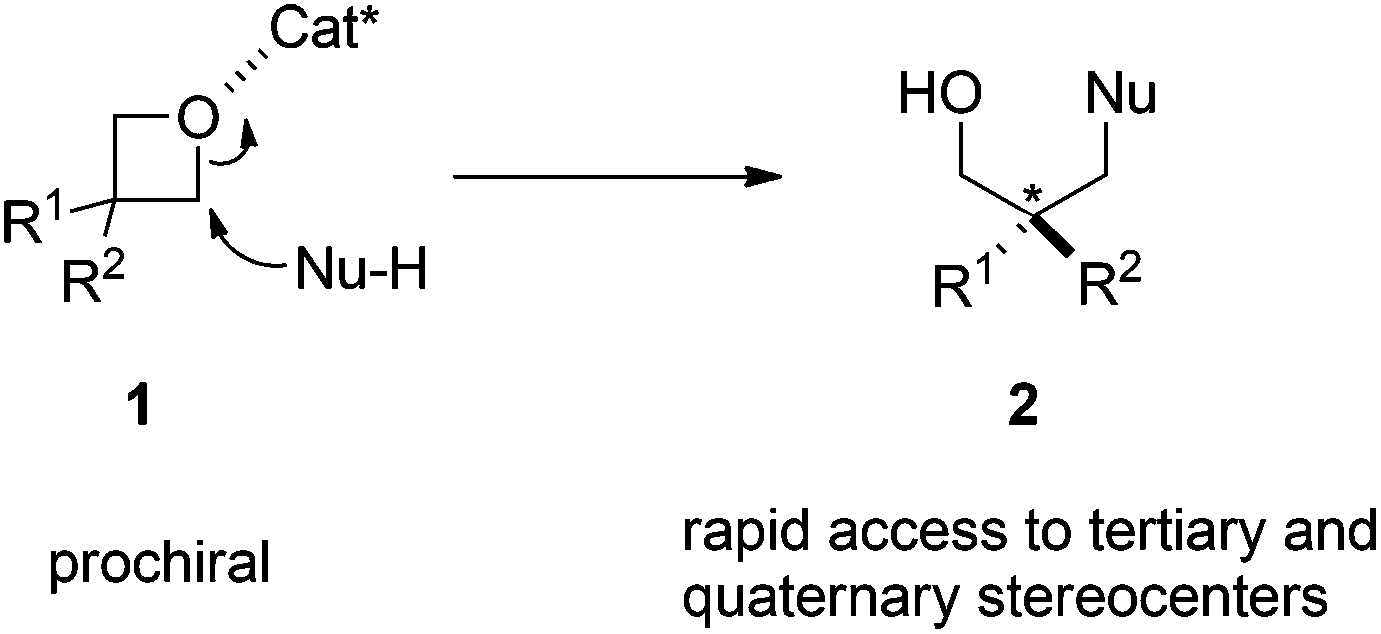
Enantioselective Oxetane Ring Opening with Chloride: Unusual Use of Wet Molecular Sieves for the Controlled Release of HCl - Yang - 2016 - Angewandte Chemie International Edition - Wiley Online Library

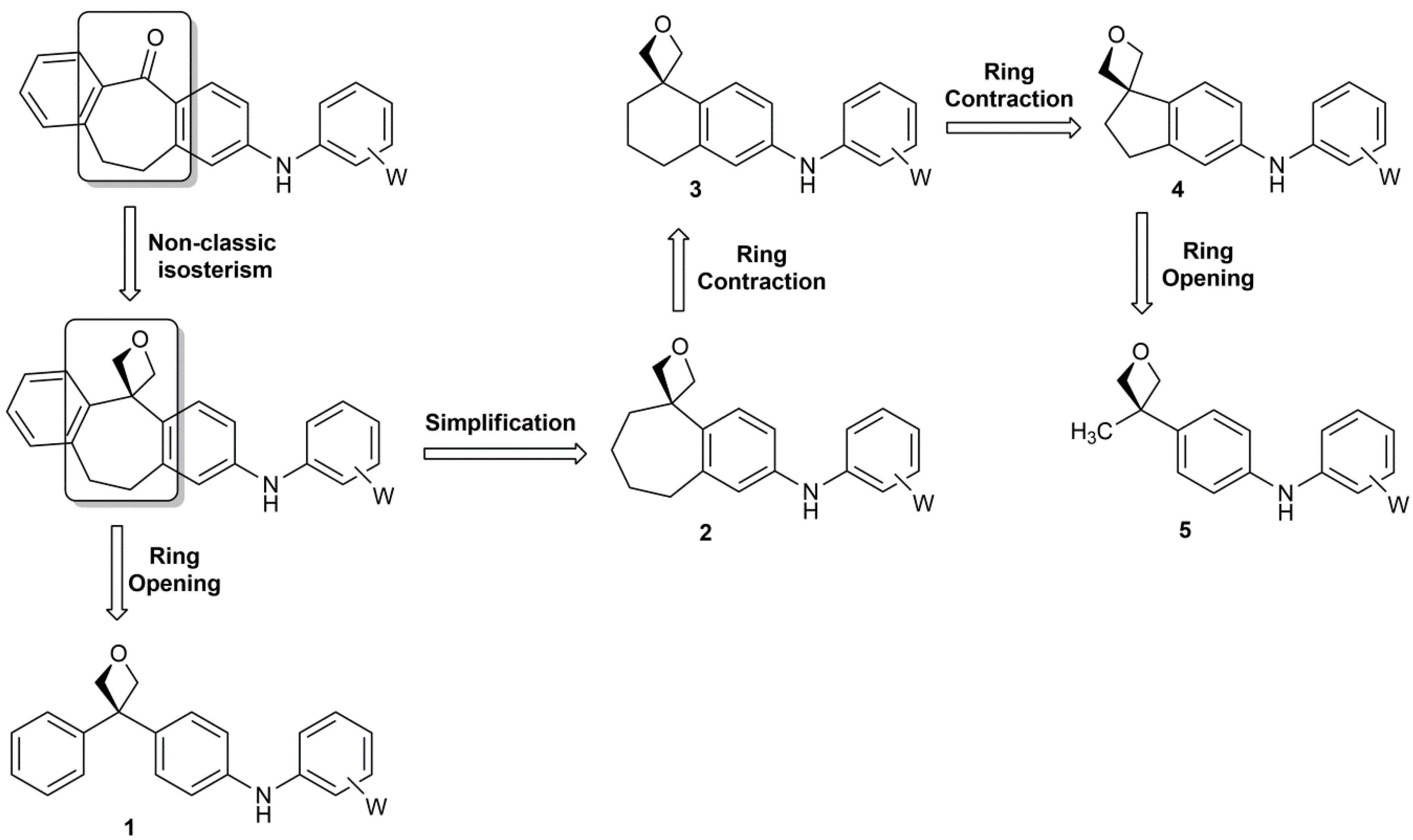
Application of oxetane ring opening toward stereoselective synthesis of zincophorin fragment - ScienceDirect
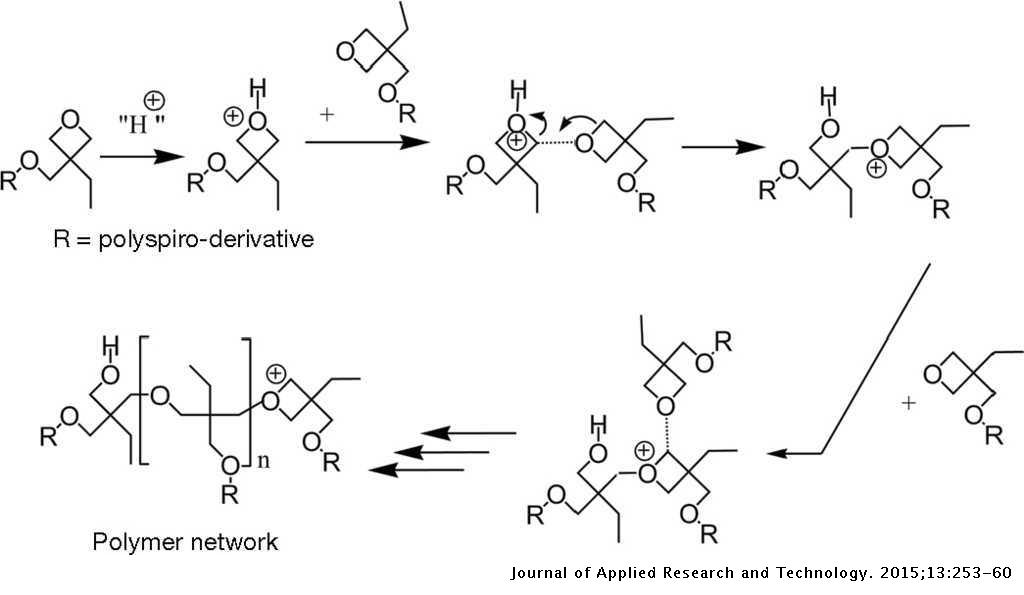
The I-V characteristics of organic hole-only devices based on crosslinked hole-transport layer | Journal of Applied Research and Technology. JART
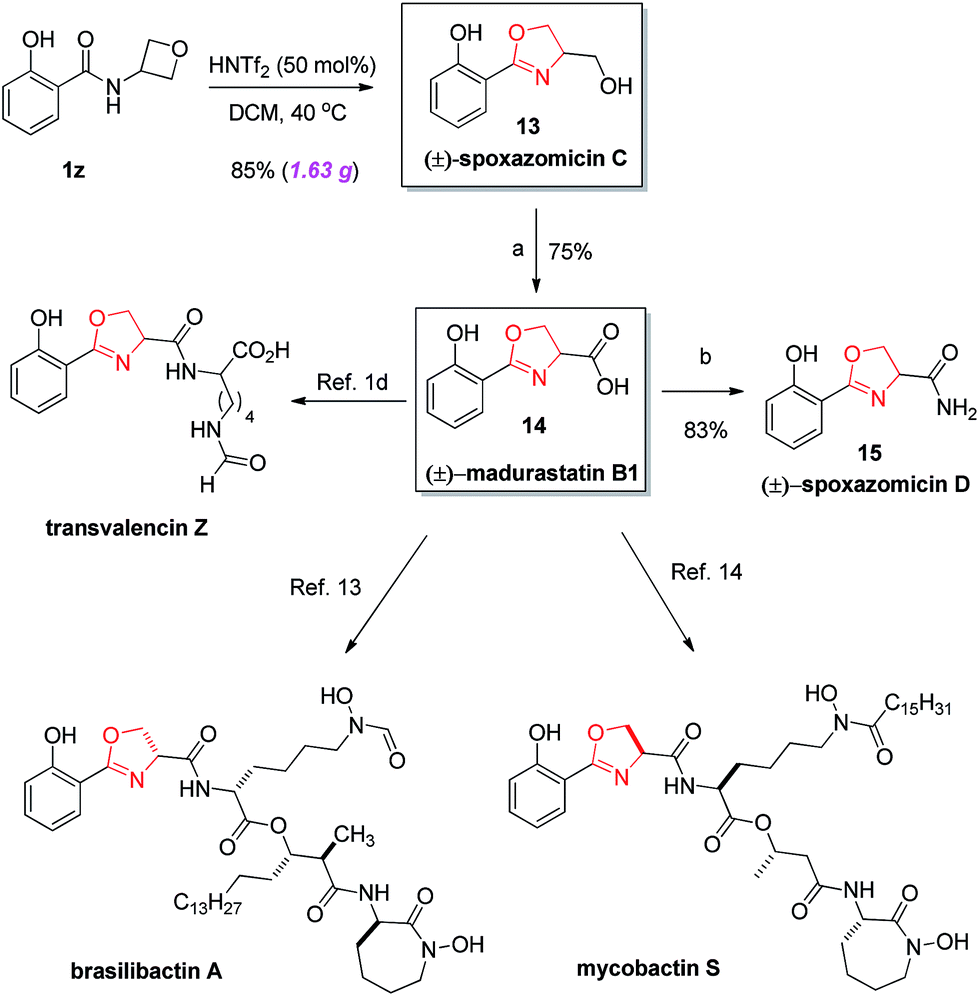
A mild catalytic synthesis of 2-oxazolines via oxetane ring-opening: rapid access to a diverse family of natural products - Chemical Science (RSC Publishing) DOI:10.1039/C9SC03843D
Catalytic reactions of oxetanes with protonic reagents and aprotic reagents leading to novel polymers

Enantioselective Oxetane Ring Opening with Chloride: Unusual Use of Wet Molecular Sieves for the Controlled Release of HCl - Yang - 2016 - Angewandte Chemie International Edition - Wiley Online Library
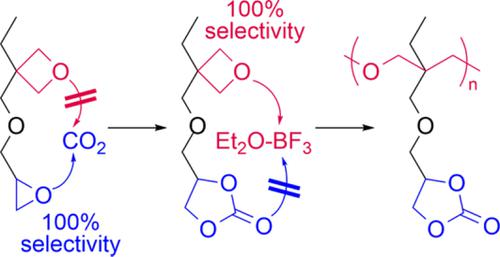
Synthesis and cationic ring‐opening polymerization of oxetane monomer containing five‐membered cyclic carbonate moiety via highly chemoselective addition of CO2,Journal of Polymer Science Part A: Polymer Chemistry - X-MOL

Enantioselective Oxetane Ring Opening with Chloride: Unusual Use of Wet Molecular Sieves for the Controlled Release of HCl - Yang - 2016 - Angewandte Chemie International Edition - Wiley Online Library
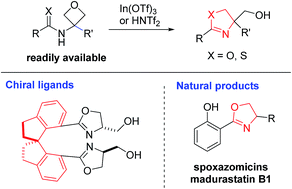
A mild catalytic synthesis of 2-oxazolines via oxetane ring-opening: rapid access to a diverse family of natural products - Chemical Science (RSC Publishing)
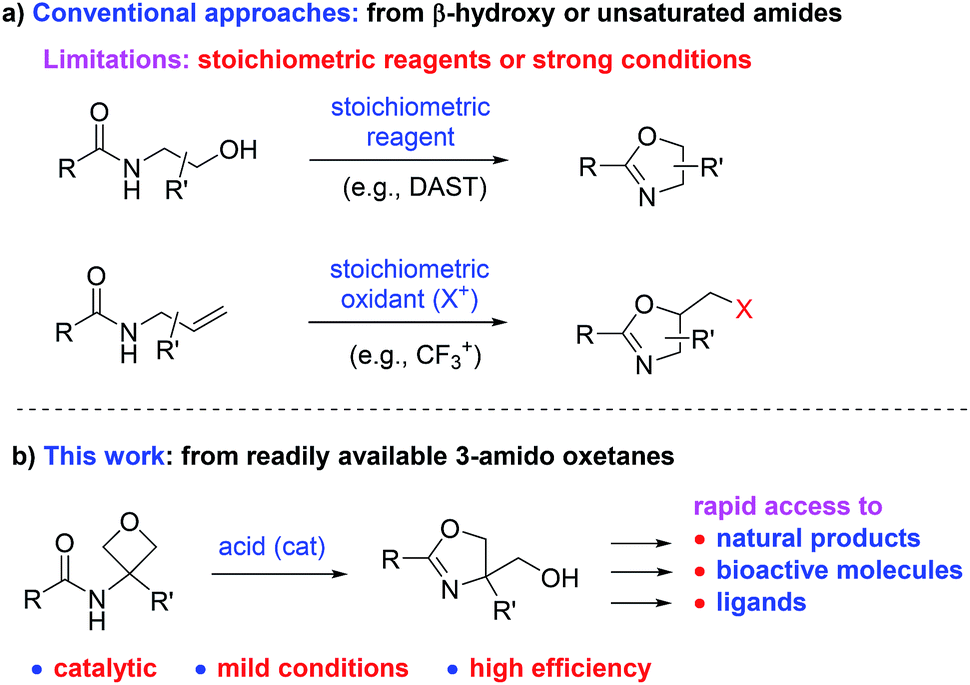
A mild catalytic synthesis of 2-oxazolines via oxetane ring-opening: rapid access to a diverse family of natural products - Chemical Science (RSC Publishing) DOI:10.1039/C9SC03843D