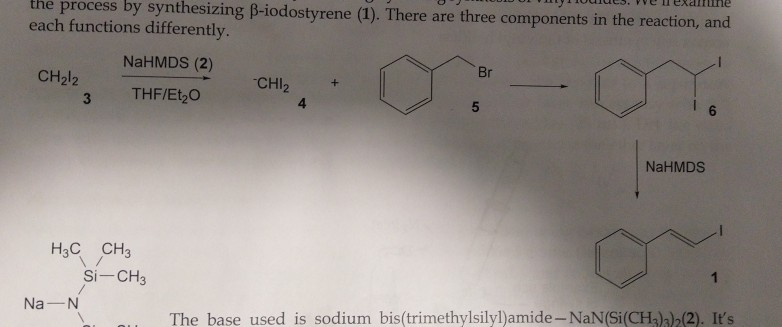
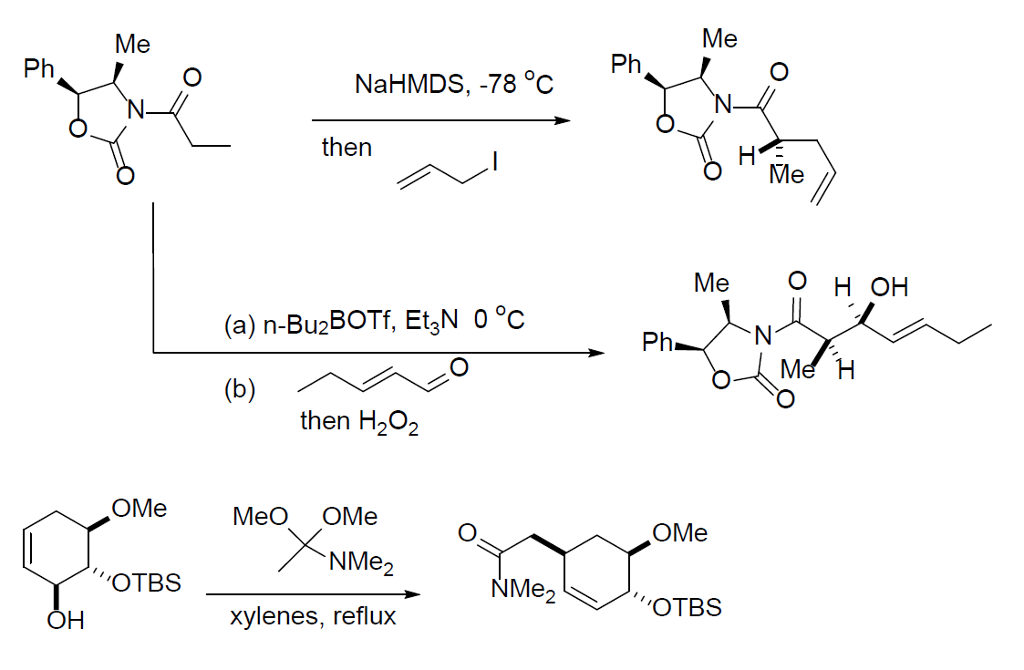
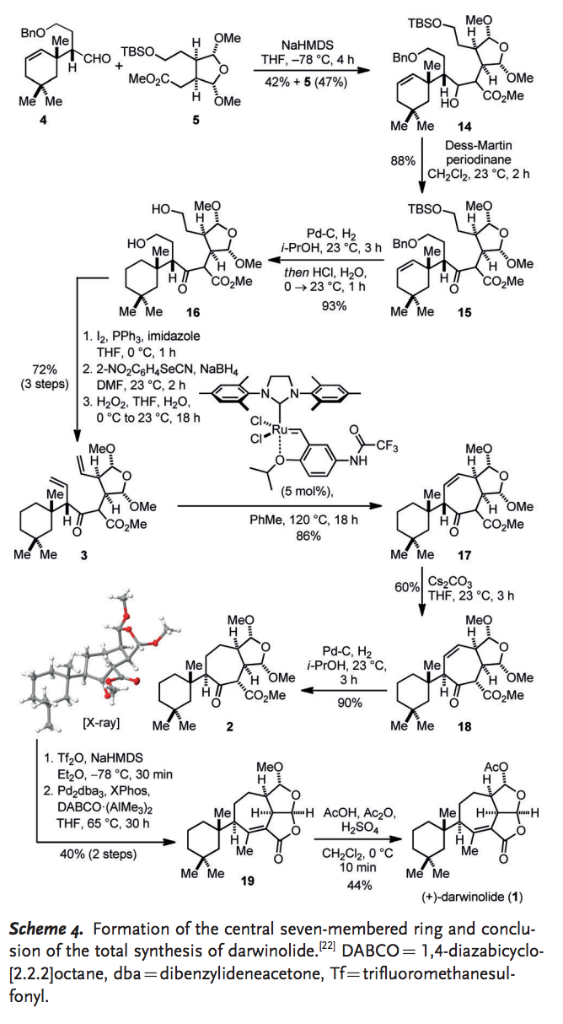
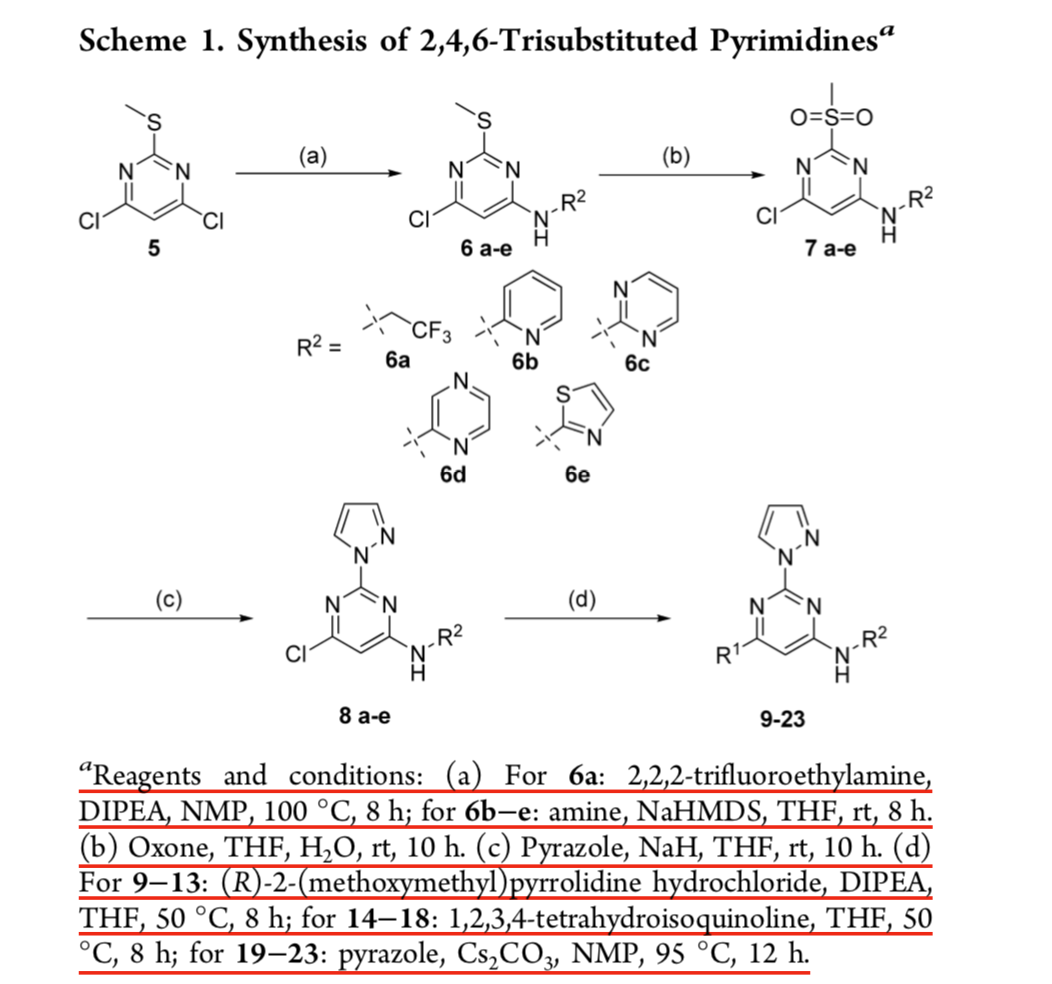
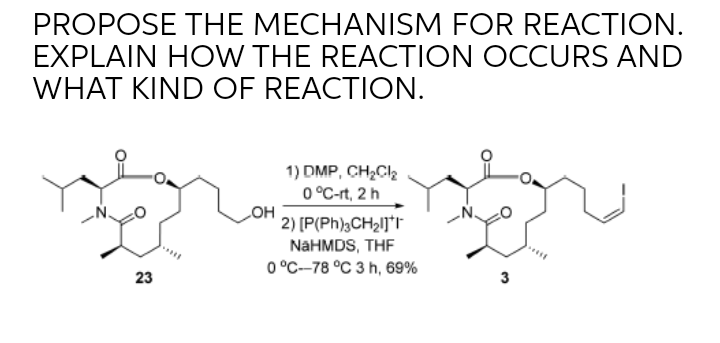
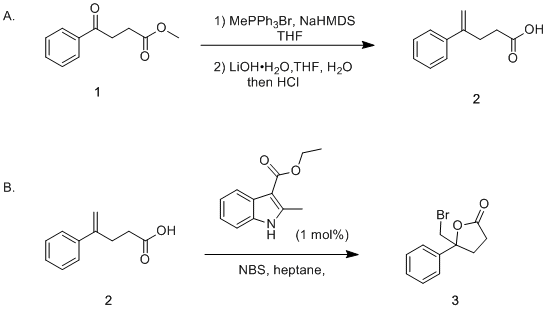
Catalysts | Free Full-Text | Transition Metal-Catalyzed α-Position Carbon–Carbon Bond Formations of Carbonyl Derivatives | HTML

Nickel(0)/NaHMDS adduct-mediated intramolecular alkylation of unactivated arenes via a homolytic aromatic substitution mechanism - Chemical Communications (RSC Publishing)
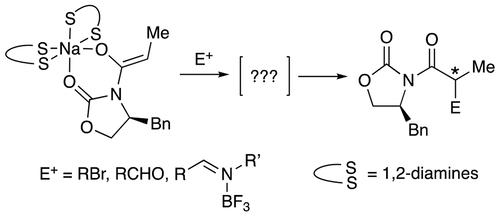
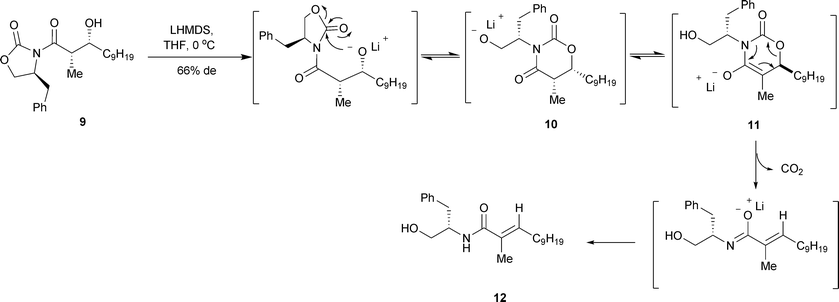
Structures and Reactivities of Sodiated Evans Enolates: Role of Solvation and Mixed Aggregation on the Stereochemistry and Mecha
A temporary stereocentre approach for the asymmetric synthesis of chiral cyclopropane-carboxaldehydes - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B908600E
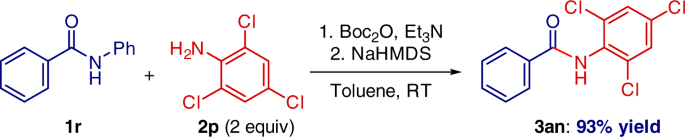
Highly selective transition-metal-free transamidation of amides and amidation of esters at room temperature | Nature Communications

AlMe3‐Mediated Regio‐ and Chemoselective Reactions of Indole with Carbamoyl Electrophiles - Velavan - 2013 - European Journal of Organic Chemistry - Wiley Online Library