
Revisiting thioflavin T (ThT) fluorescence as a marker of protein fibrillation – The prominent role of electrostatic interactions - ScienceDirect
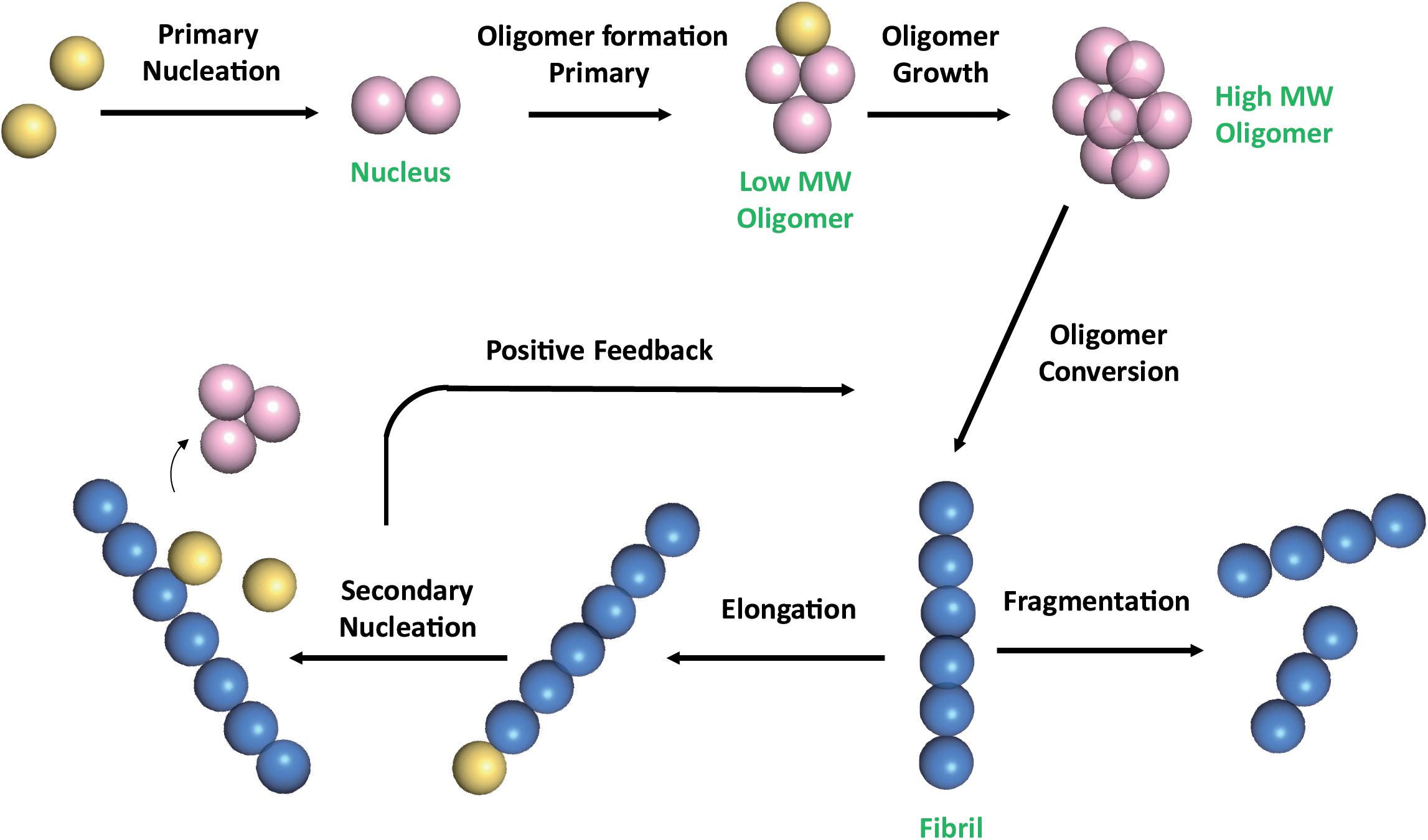
Frontiers | Natural Compounds as Inhibitors of Aβ Peptide Aggregation: Chemical Requirements and Molecular Mechanisms | Neuroscience

Binding mode of Thioflavin T and other molecular probes in the context of amyloid fibrils—current status | Semantic Scholar
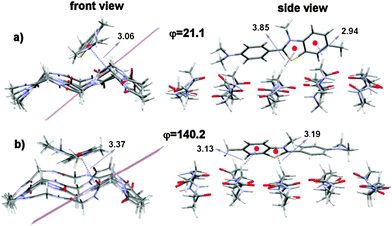
Crystal structure of thioflavin-T and its binding to amyloid fibrils : insights at the molecular level - Chemical Communications (RSC Publishing) DOI:10.1039/B912396B

Molecular Mechanism of Thioflavin-T Binding to the Surface of β-Rich Peptide Self-Assemblies - ScienceDirect
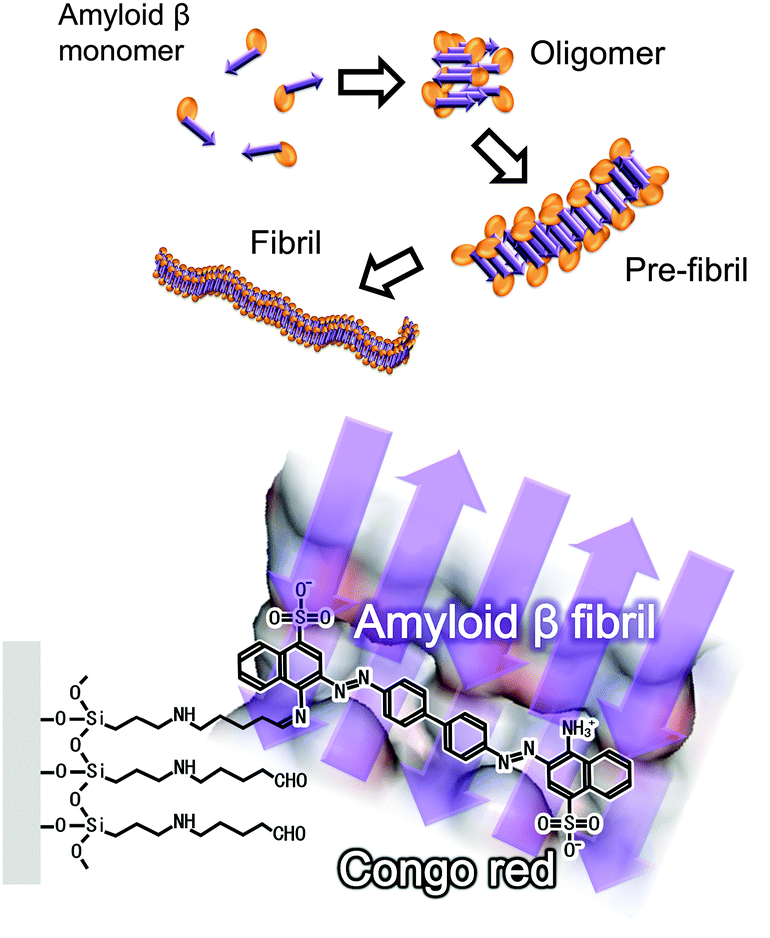
A label-free electrical assay of fibrous amyloid β based on semiconductor biosensing - Chemical Communications (RSC Publishing) DOI:10.1039/C3CC49460H
RopA and RopB aggregates binding with amyloid specific probe thioflavin... | Download Scientific Diagram

Binding of Congo Red to Amyloid Protofibrils of the Alzheimer Aβ9–40 Peptide Probed by Molecular Dynamics Simulations: Biophysical Journal
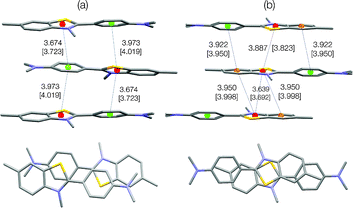
Crystal structure of thioflavin-T and its binding to amyloid fibrils : insights at the molecular level - Chemical Communications (RSC Publishing) DOI:10.1039/B912396B

Thioflavin T fluoresces as excimer in highly concentrated aqueous solutions and as monomer being incorporated in amyloid fibrils | Scientific Reports

Direct Observation of Amyloid Fibril Growth Monitored by Thioflavin T Fluorescence*210 - Journal of Biological Chemistry
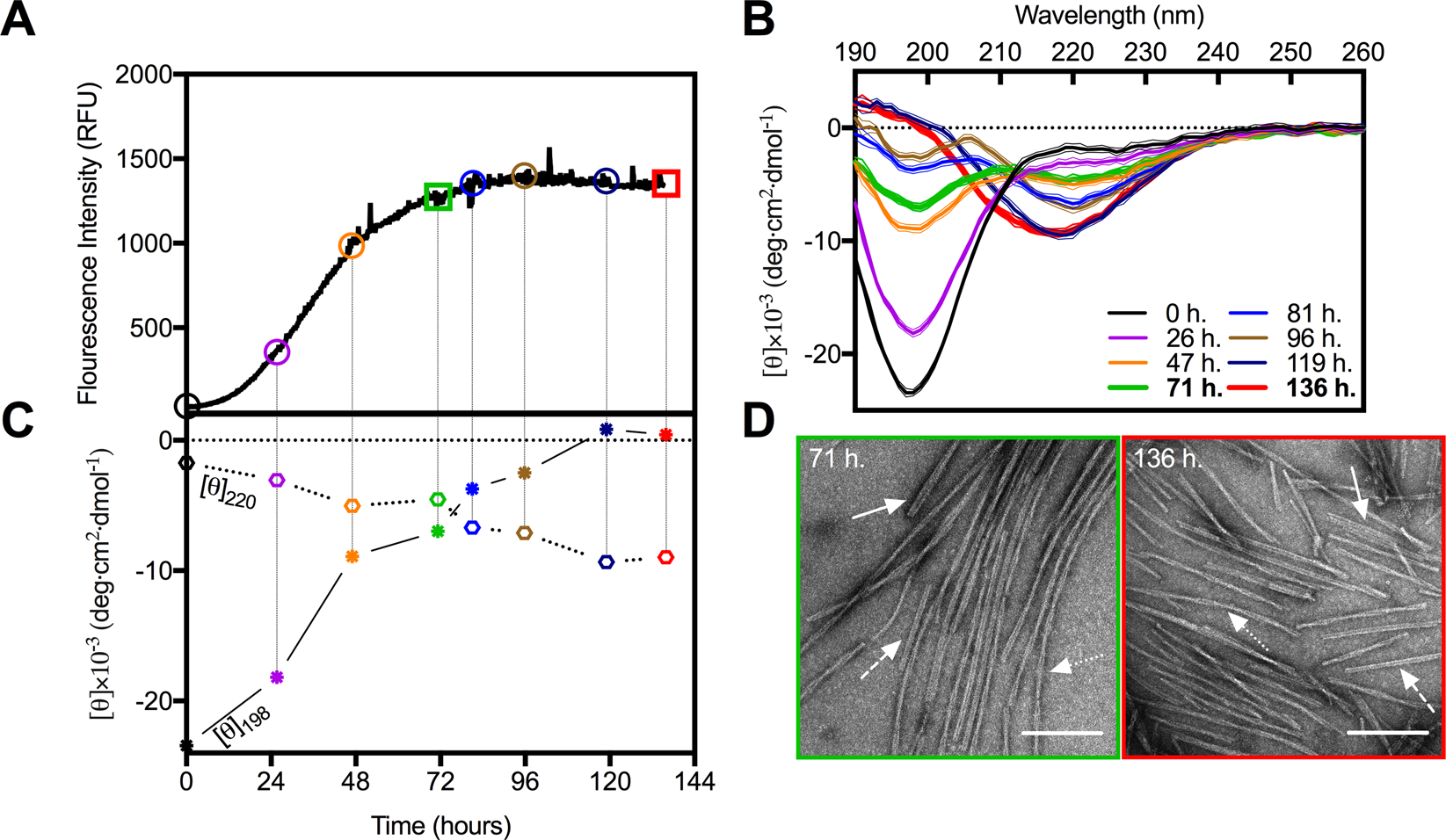
Early Stage Alpha-Synuclein Amyloid Fibrils are Reservoirs of Membrane- Binding Species | Scientific Reports
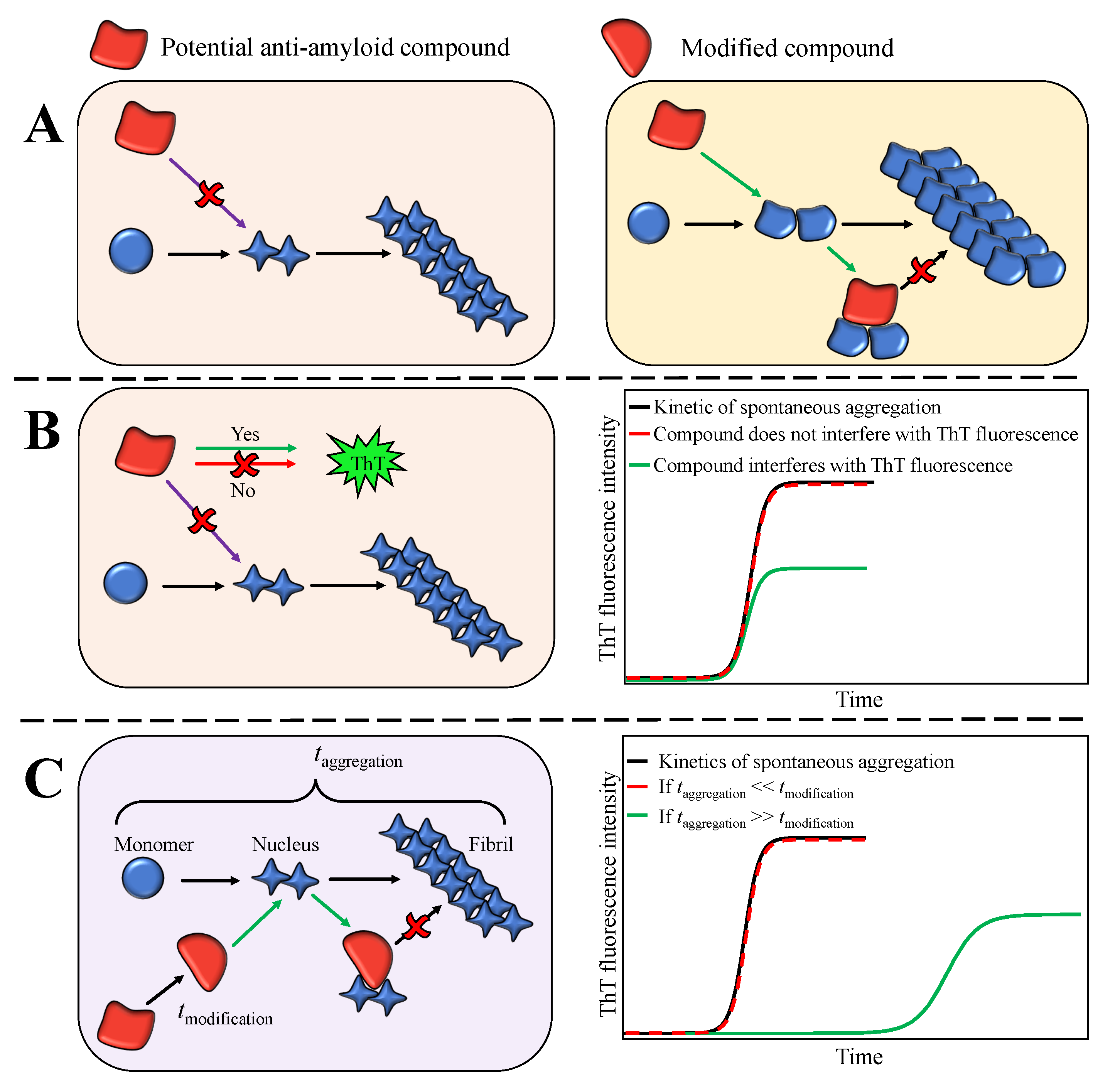



![Fibrillation mechanism controlled by CB[6]. (a) Thioflavin T (ThT)... | Download Scientific Diagram Fibrillation mechanism controlled by CB[6]. (a) Thioflavin T (ThT)... | Download Scientific Diagram](https://www.researchgate.net/publication/318504796/figure/fig3/AS:518719889985536@1500683867675/Fibrillation-mechanism-controlled-by-CB6-a-Thioflavin-T-ThT-assay-for-the-kinetics.png)


![Exploring the occurrence of thioflavin-T-positive insulin amyloid aggregation intermediates [PeerJ] Exploring the occurrence of thioflavin-T-positive insulin amyloid aggregation intermediates [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2021/10918/1/fig-1-full.png)


