
Figure 2.1 from Kitchen wastewater treatment by adsorption process using banana trunk fibers (Musa Sapientum) and chitosan | Semantic Scholar

Comparison of the Thermodynamic Parameters Estimation for the Adsorption Process of the Metals from Liquid Phase on Activated Carbons | IntechOpen

Organic Dye Adsorption by Amphiphilic Tris‐Urea Supramolecular Hydrogel - Takeshita - 2017 - Chemistry – An Asian Journal - Wiley Online Library

Removal of Lignin from Wastewater Using an Industrial Waste as Adsorbent: A Statistical and Kinetic Modeling Approach | Journal of Hazardous, Toxic, and Radioactive Waste | Vol 26, No 2

Mechanism of adsorption of actives onto microporous functionalised calcium carbonate (FCC) | springerprofessional.de

SciELO - Brasil - Study of the Adsorption of Arsenic (III and V) by Magnetite Nanoparticles Synthetized via AACVD Study of the Adsorption of Arsenic (III and V) by Magnetite Nanoparticles Synthetized
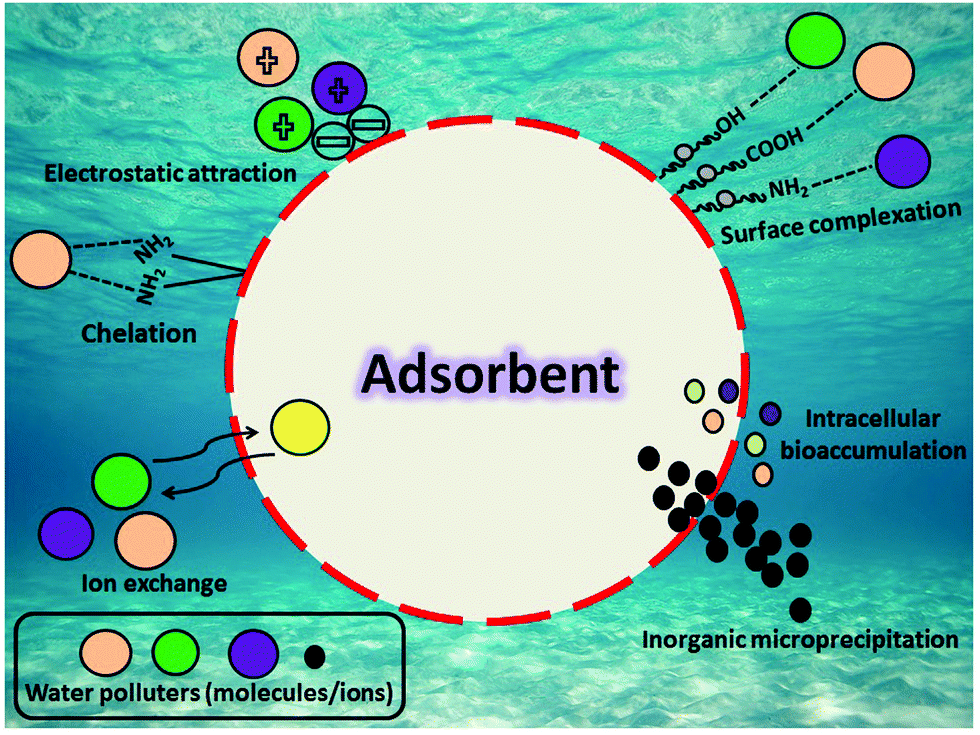
Perspectives regarding metal/mineral-incorporating materials for water purification: with special focus on Cr( vi ) removal - Materials Advances (RSC Publishing) DOI:10.1039/D0MA00153H
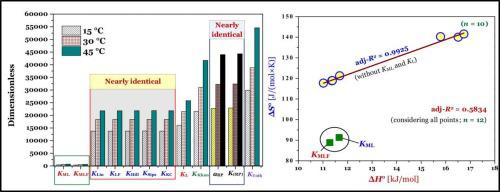
Thermodynamic parameters of liquid–phase adsorption process calculated from different equilibrium constants related to adsorption isotherms: A comparison study,Journal of Environmental Chemical Engineering - X-MOL

Mechanism of adsorption and regeneration of Pb(II) on mesoporous silica. | Download Scientific Diagram

Systematic Investigation on the Adsorption Performance and Mechanism of MnO2/TA Nanoflowers for Cu(II) Removal from Aqueous Solution - Gao - 2019 - ChemistrySelect - Wiley Online Library
PLOS ONE: Preparation of magnetic mesoporous silica from rice husk for aflatoxin B1 removal: Optimum process and adsorption mechanism

Adsorption behavior and mechanism of chloramphenicols, sulfonamides, and non-antibiotic pharmaceuticals on multi-walled carbon nanotubes - ScienceDirect

Polymers | Free Full-Text | Advances in Amine-Surface Functionalization of Inorganic Adsorbents for Water Treatment and Antimicrobial Activities: A Review | HTML
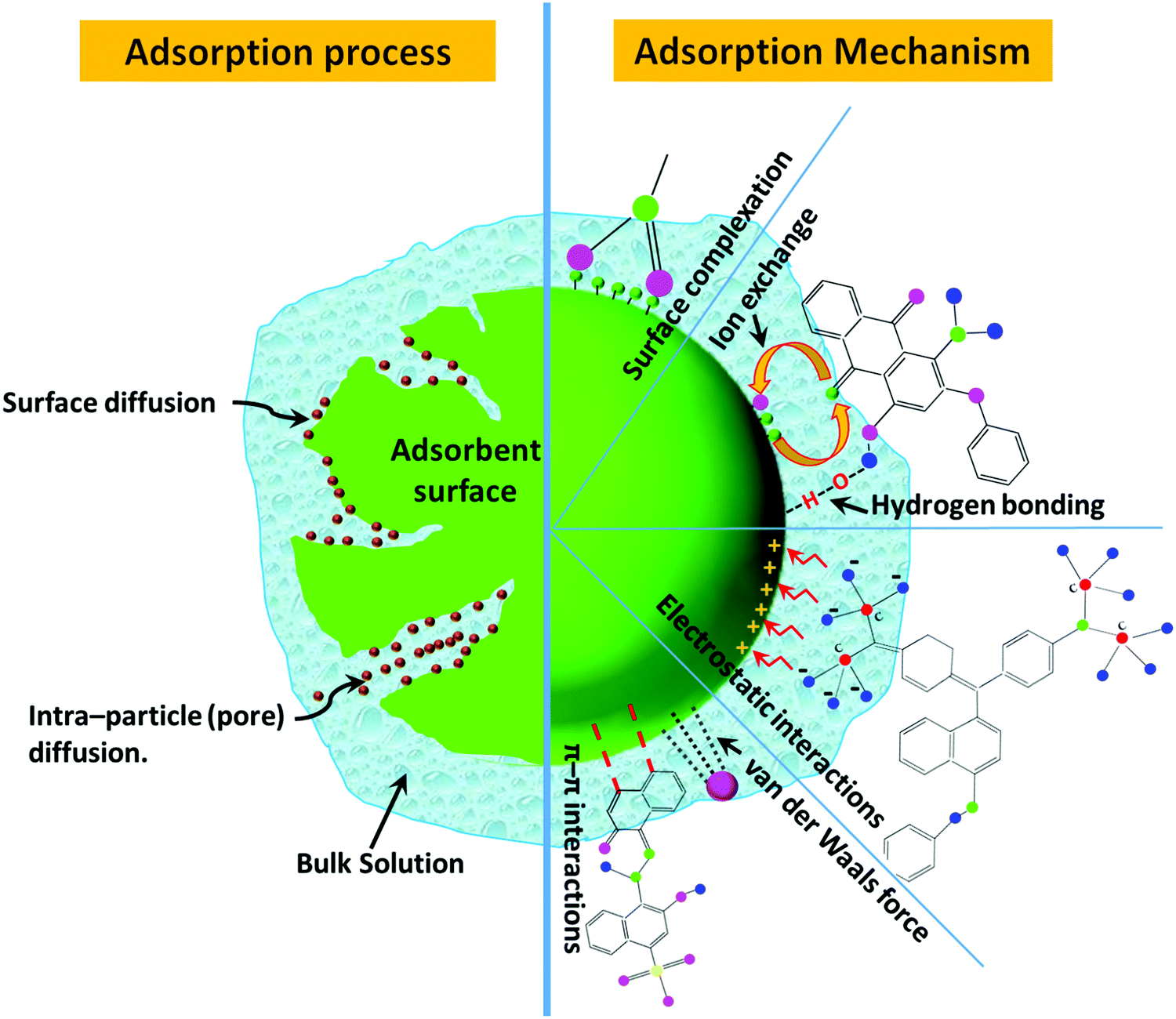
Recent advances on the removal of dyes from wastewater using various adsorbents: a critical review - Materials Advances (RSC Publishing) DOI:10.1039/D1MA00354B

a) Schematic of adsorption process of MB. (b) Proposed mechanism for... | Download Scientific Diagram

Magnetic/Polyetherimide-Acrylonitrile Composite Nanofibers for Nickel Ion Removal from Aqueous Solution. - Abstract - Europe PMC









