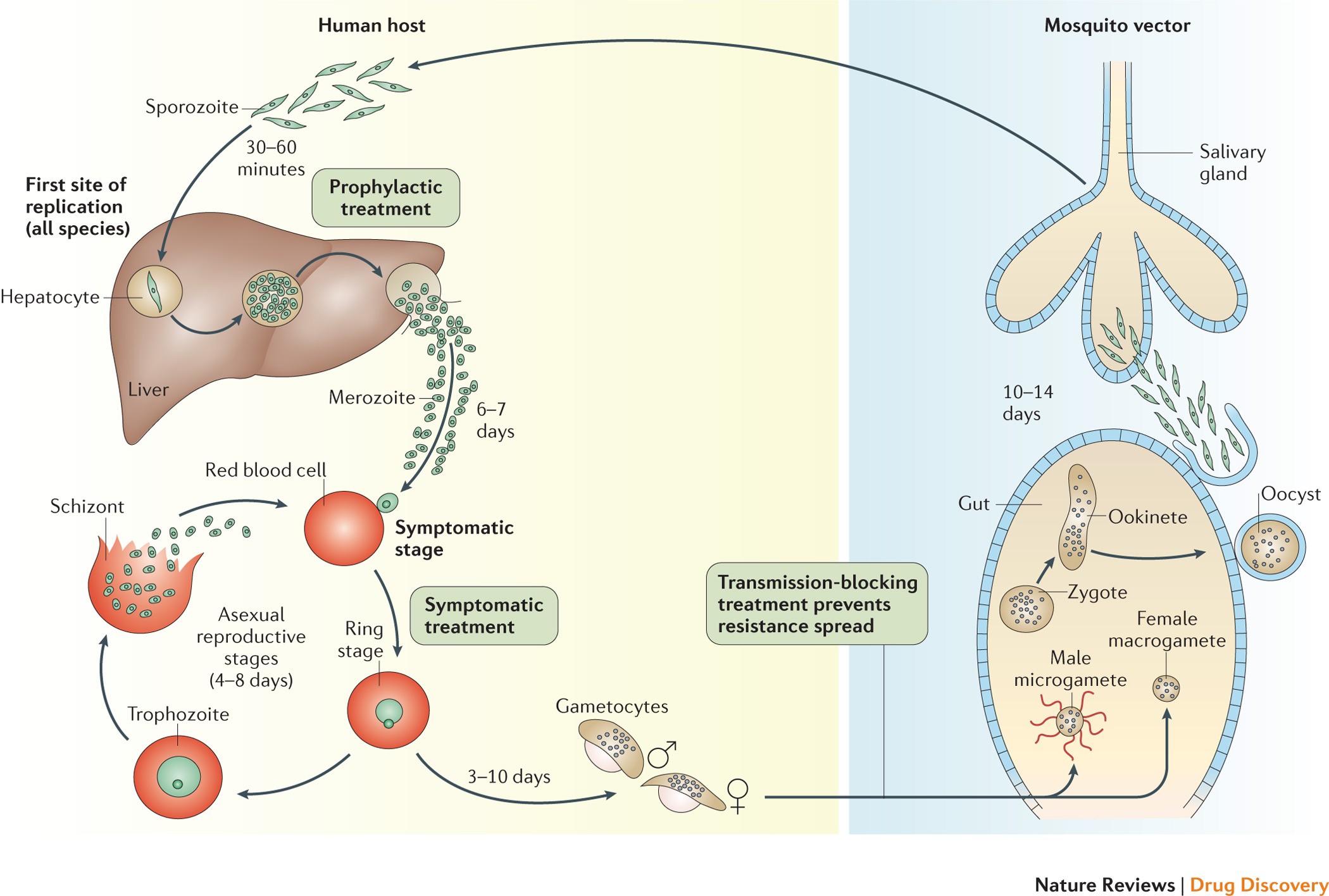
PLOS Neglected Tropical Diseases: The Use of Artemether-Lumefantrine for the Treatment of Uncomplicated Plasmodium vivax Malaria

Oxidative stress in malaria and artemisinin combination therapy: Pros and Cons - Kavishe - 2017 - The FEBS Journal - Wiley Online Library

Multi-omic Characterization of the Mode of Action of a Potent New Antimalarial Compound, JPC-3210, Against Plasmodium falciparum - ScienceDirect
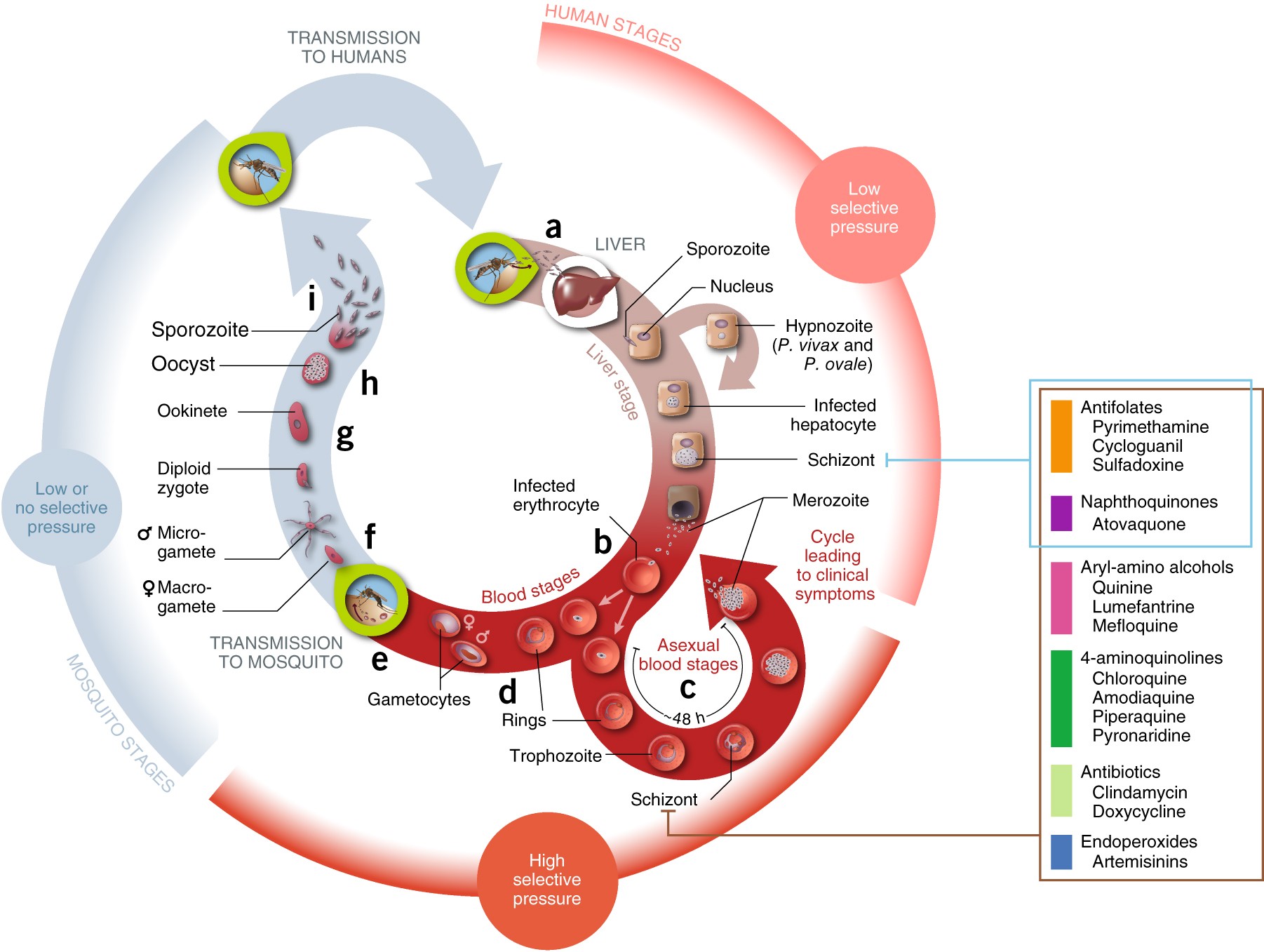
Antimalarial drug resistance: linking Plasmodium falciparum parasite biology to the clinic | Nature Medicine

References in Efficacy and safety of artemether–lumefantrine compared with quinine in pregnant women with uncomplicated Plasmodium falciparum malaria: an open-label, randomised, non-inferiority trial - The Lancet Infectious Diseases
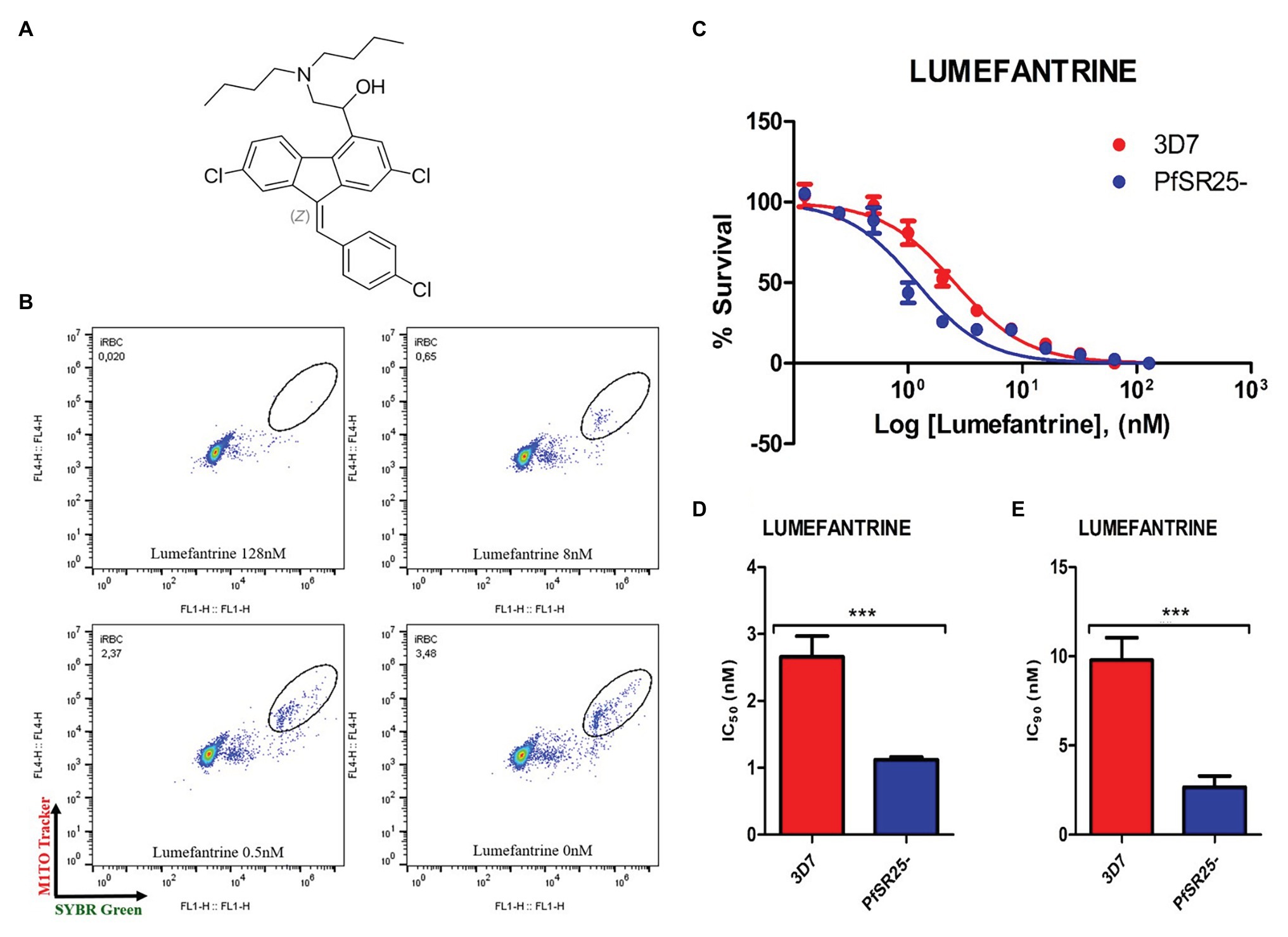



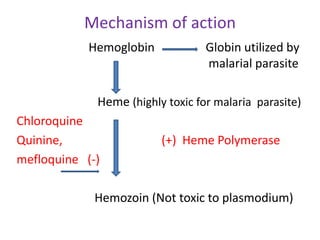



![treatment_of_malaria [TUSOM | Pharmwiki] treatment_of_malaria [TUSOM | Pharmwiki]](https://tmedweb.tulane.edu/pharmwiki/lib/exe/fetch.php/malaria_resistance.png?w=700&tok=1a46a7)



![treatment_of_malaria [TUSOM | Pharmwiki] treatment_of_malaria [TUSOM | Pharmwiki]](https://tmedweb.tulane.edu/pharmwiki/lib/exe/fetch.php/malaria_lifecycle.png)