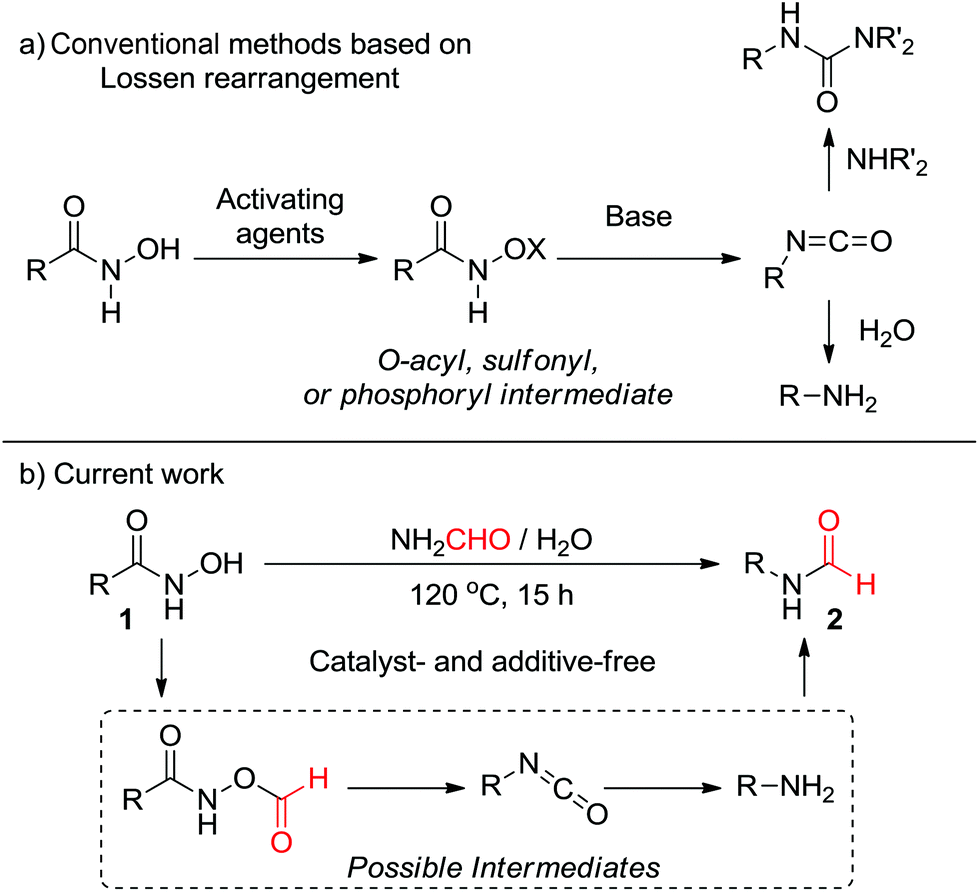
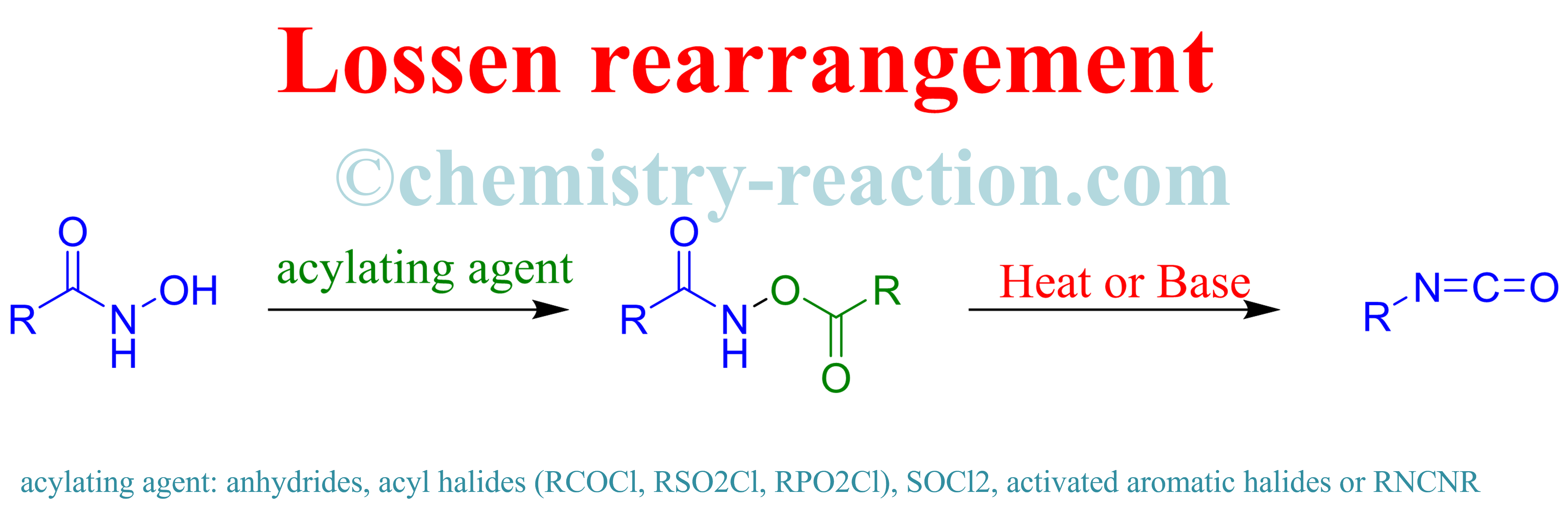
Consecutive Lossen rearrangement/transamidation reaction of hydroxamic acids under catalyst- and additive-free conditions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB00490K

DE102012100127A1 - Preparation of carbamates via a base-catalyzed Lossen rearrangement - Google Patents

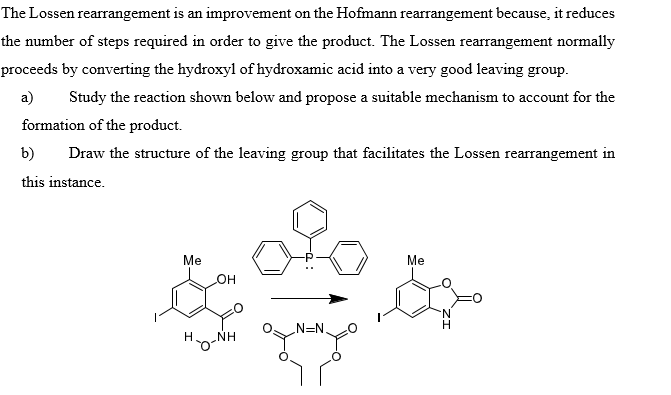
An Unusual Double Lossen Rearrangement Reaction: The Novel Molecular Detoxication Mechanism for Hydroxamic Acids
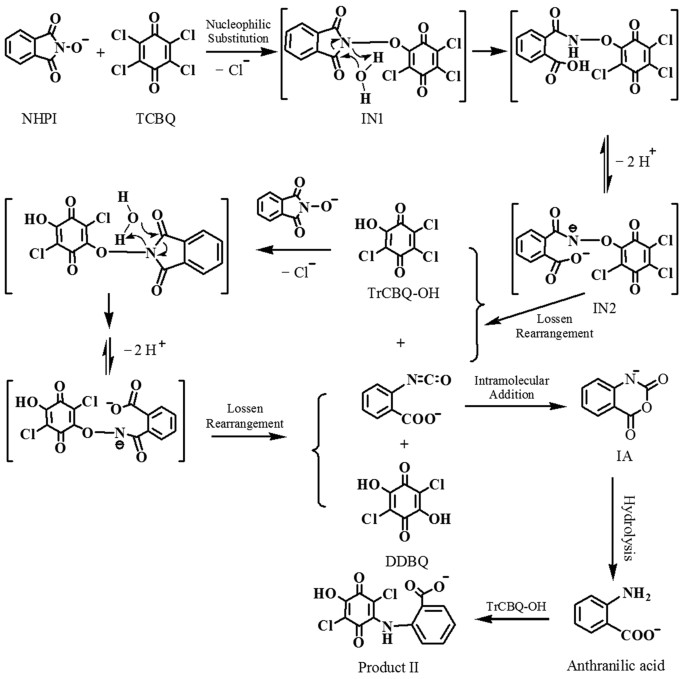
An Exceptionally Facile Two-Step Structural Isomerization and Detoxication via a Water-Assisted Double Lossen Rearrangement | Scientific Reports

Detoxifying carcinogenic polyhalogenated quinones by hydroxamic acids via an unusual double Lossen rearrangement mechanism | PNAS

EP2615082A2 - Production of carbamates by means of base-catalyzed Lossen rearrangement - Google Patents

A Combined Experimental and Computational Investigation on the Unusual Molecular Mechanism of the Lossen Rearrangement Reaction Activated by Carcinogenic Halogenated Quinones | Semantic Scholar
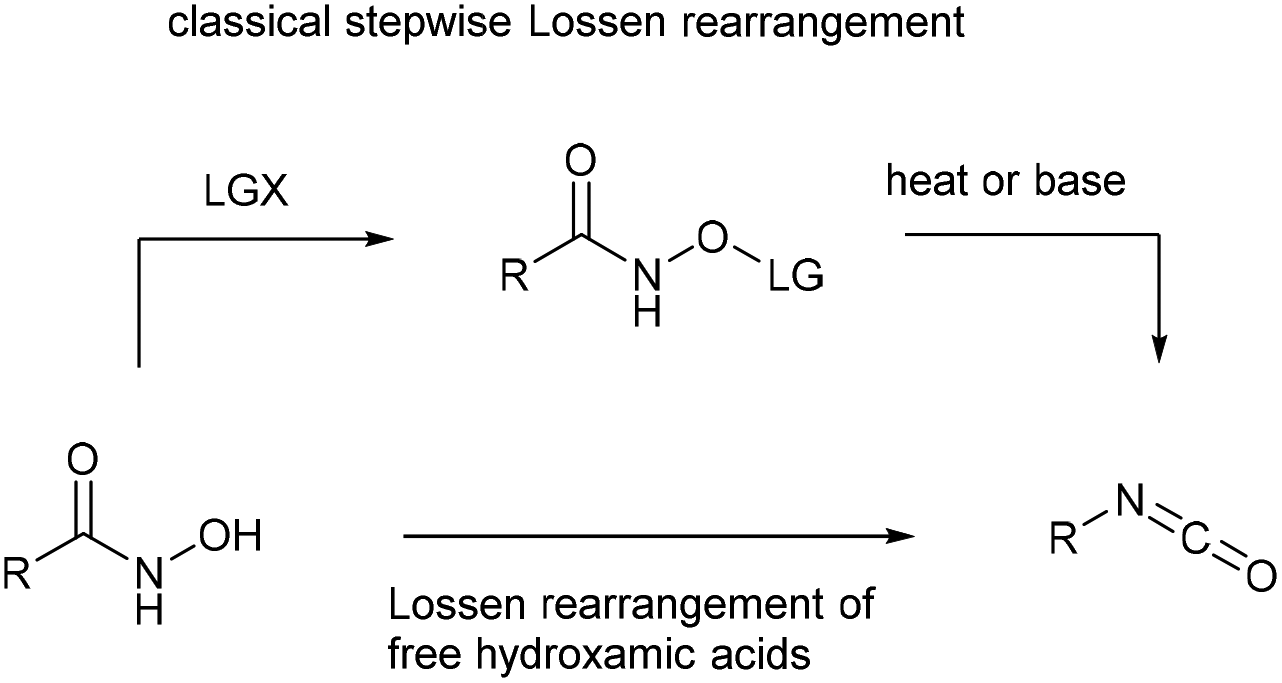
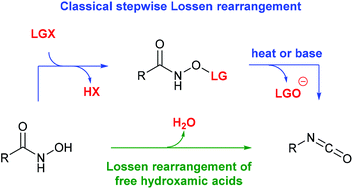
The Lossen rearrangement from free hydroxamic acids - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB00789J

A Practical Approach to Ureas and Thiocarbamates: SO2F2‐Promoted Lossen Rearrangement of Hydroxamic Acid - Zhang - 2020 - ChemistrySelect - Wiley Online Library
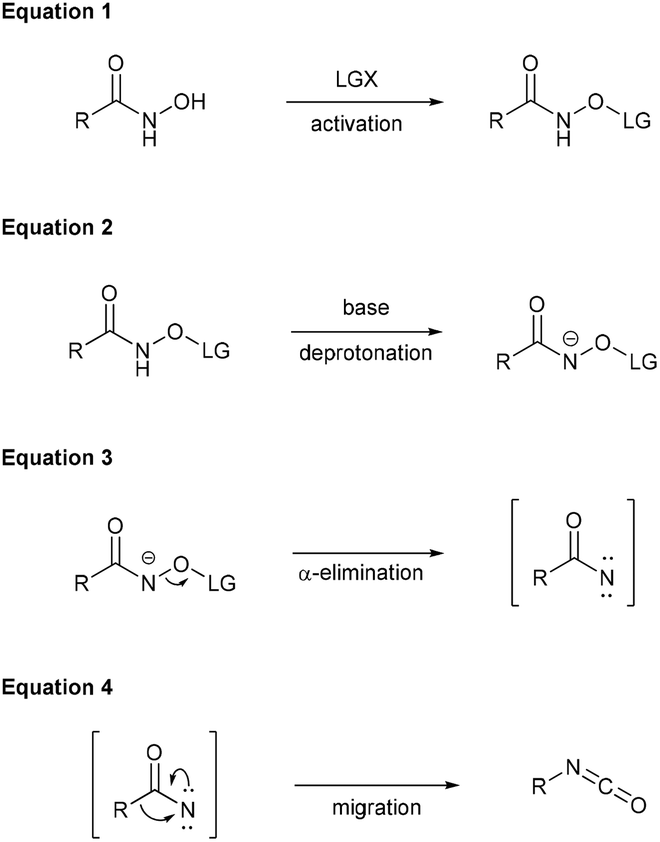
The Lossen rearrangement from free hydroxamic acids - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB00789J

Self-propagated Lossen rearrangement induced by a catalytic amount of activating agents under mild conditions - ScienceDirect

Lossen Rearrangements under Heck Reaction Conditions - AbdelHafez - 2014 - Advanced Synthesis & Catalysis - Wiley Online Library

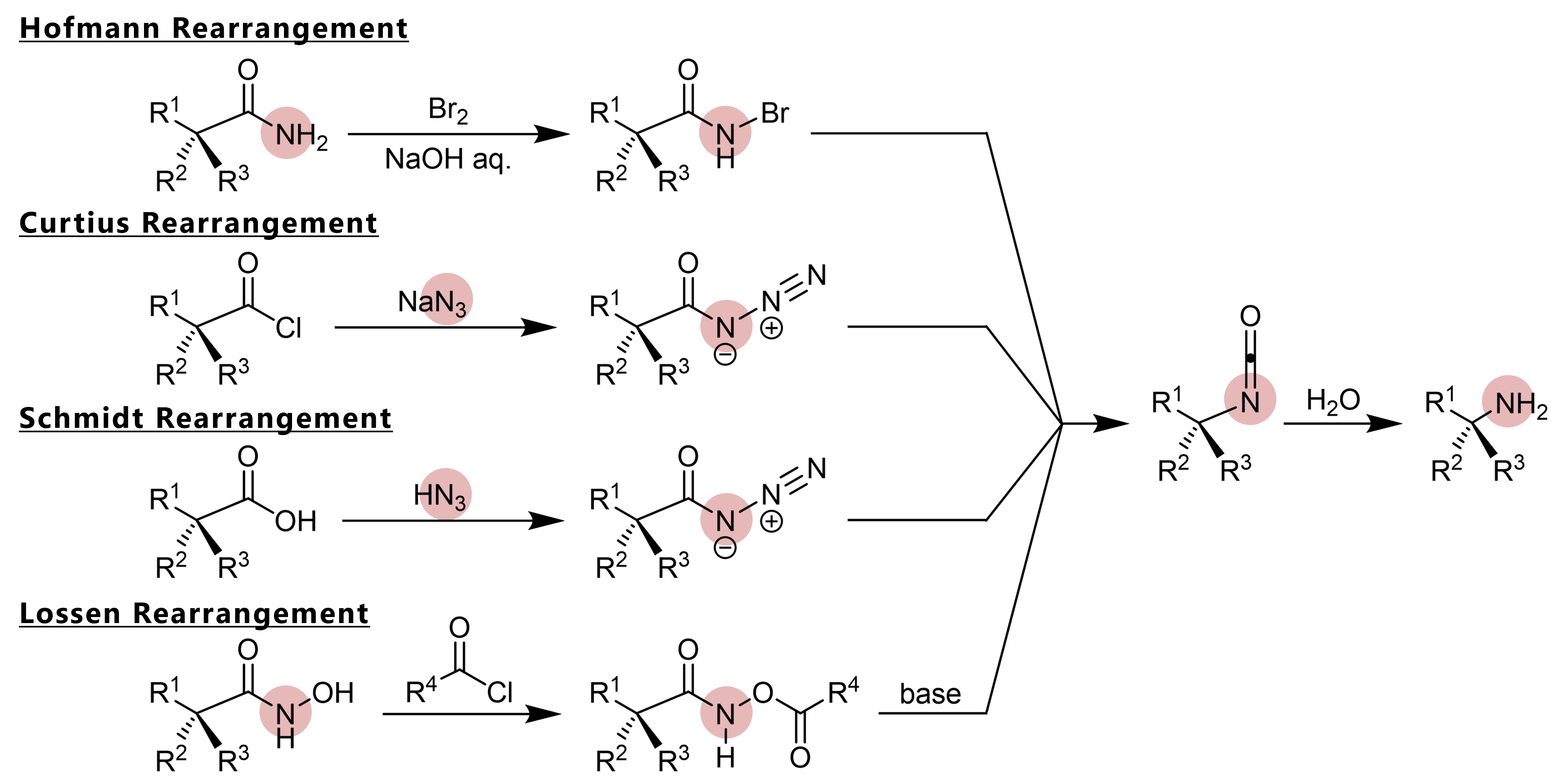

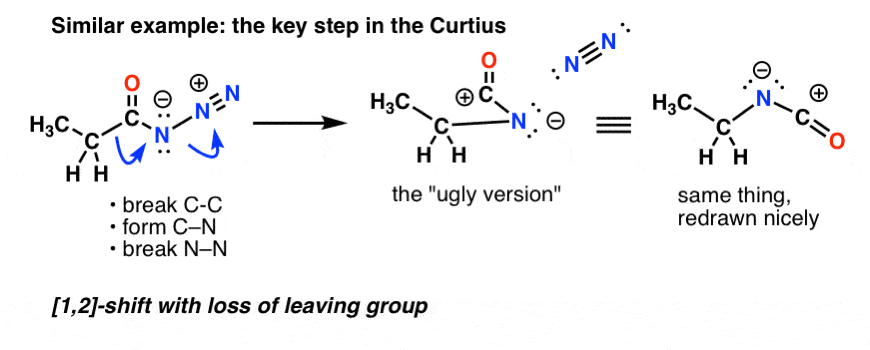
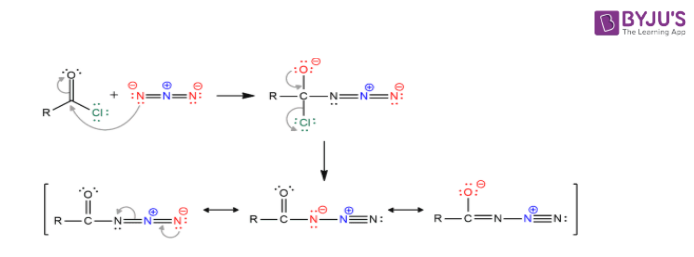



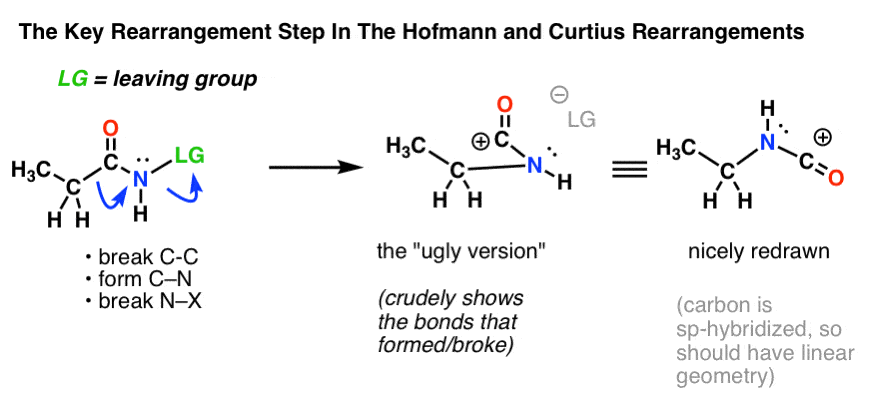
![11] Curtius Rearrangement 1890 – ChemInfoGraphic 11] Curtius Rearrangement 1890 – ChemInfoGraphic](https://cheminfographic.files.wordpress.com/2017/08/11_curtius_rearrangement_4.jpg)

