Lithium Diisopropylamide: Solution Kinetics and Implications for Organic Synthesis - Collum - 2007 - Angewandte Chemie International Edition - Wiley Online Library
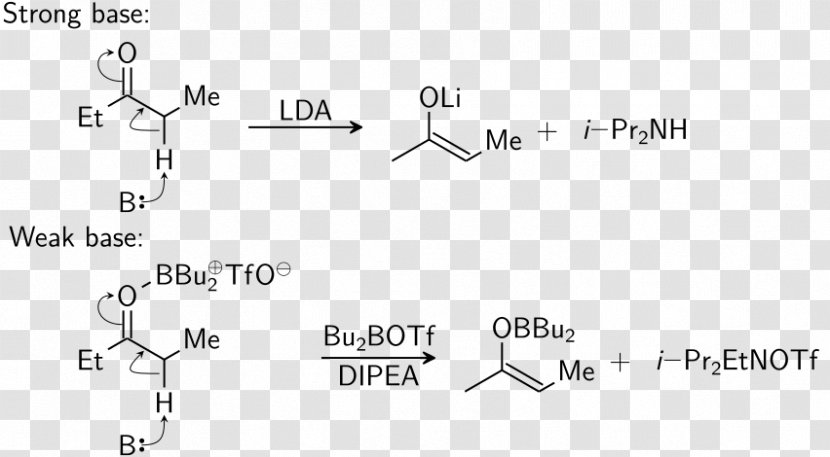
Triflate Aldol Reaction Dibutylboron Trifluoromethanesulfonate Condensation Lithium Diisopropylamide - Mechanism - Malonic Ester Synthesis Transparent PNG
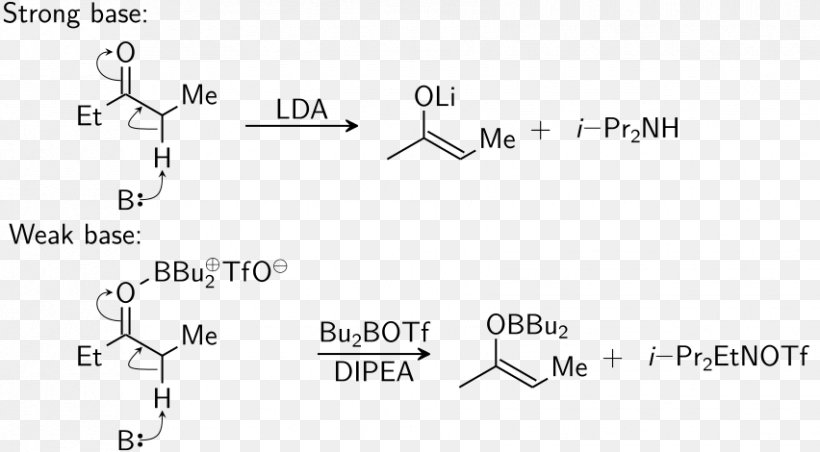
Triflate Aldol Reaction Dibutylboron Trifluoromethanesulfonate Aldol Condensation Lithium Diisopropylamide, PNG, 852x470px, Triflate, Acid Catalysis, Aldol, Aldol

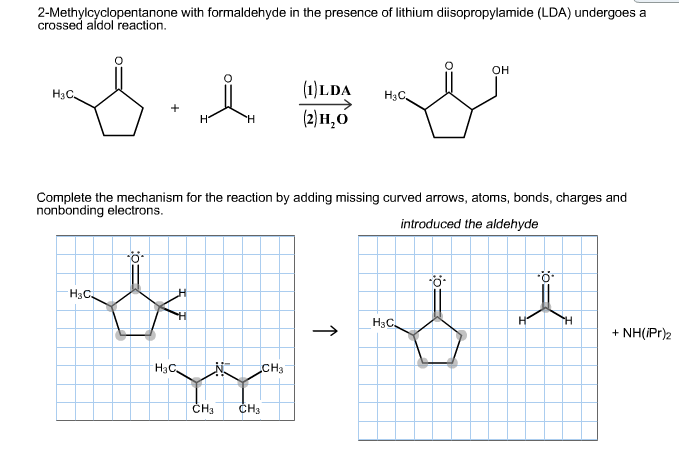
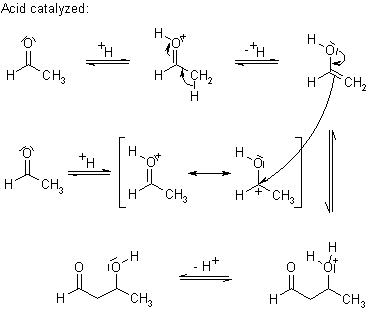
Draw a mechanism for the following reaction, and explain why an acid source is required after the reaction is complete. | Study.com

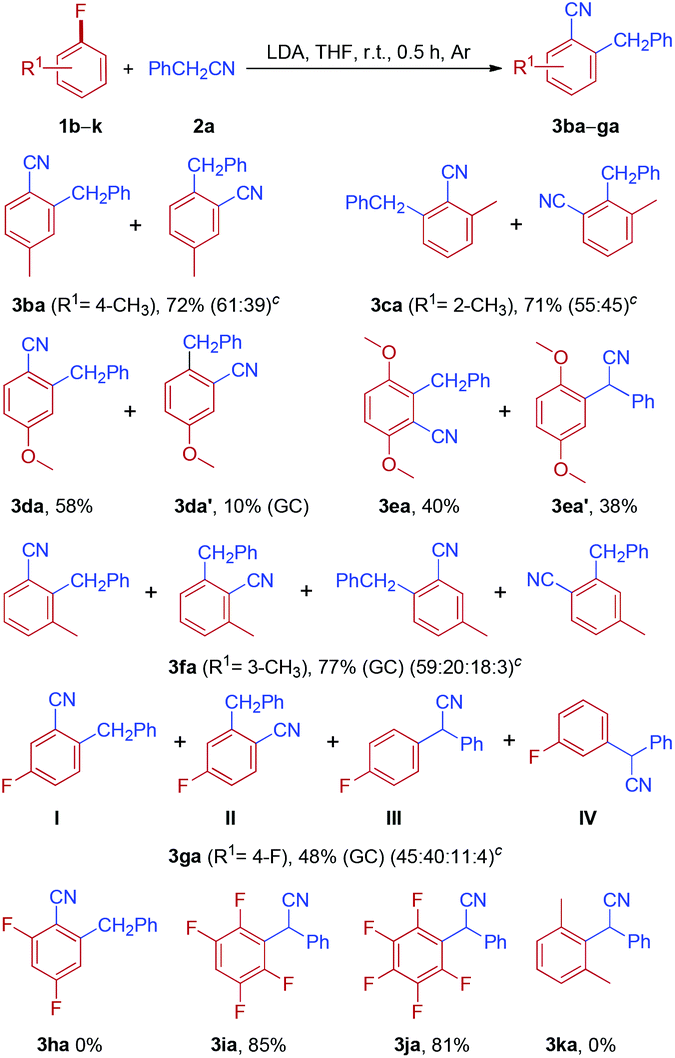
Facile Synthesis of 3-Arylindenes by HMPA-Promoted Direct Arylation of Indenes with Aryl Fluorides. - Abstract - Europe PMC

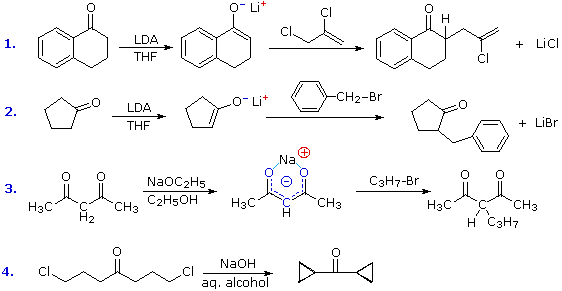
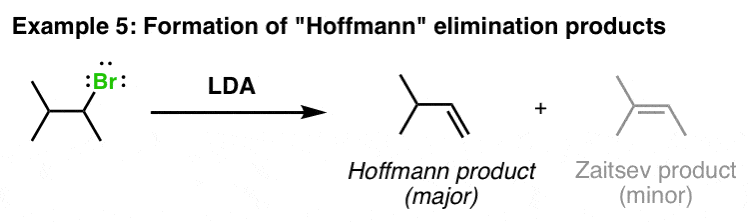
Synthesis of alkenes from ketones via arylsulphonyl-hydrazones; mechanistic views; the organic chemistry notebook series, a didactical approach, n27

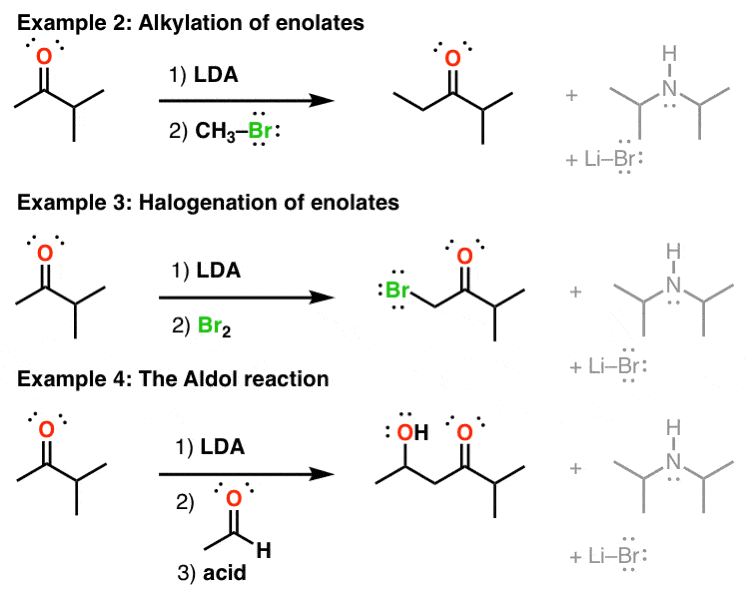
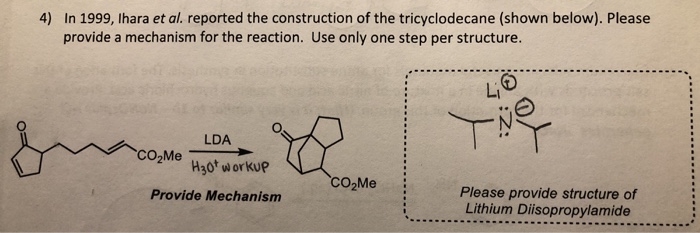
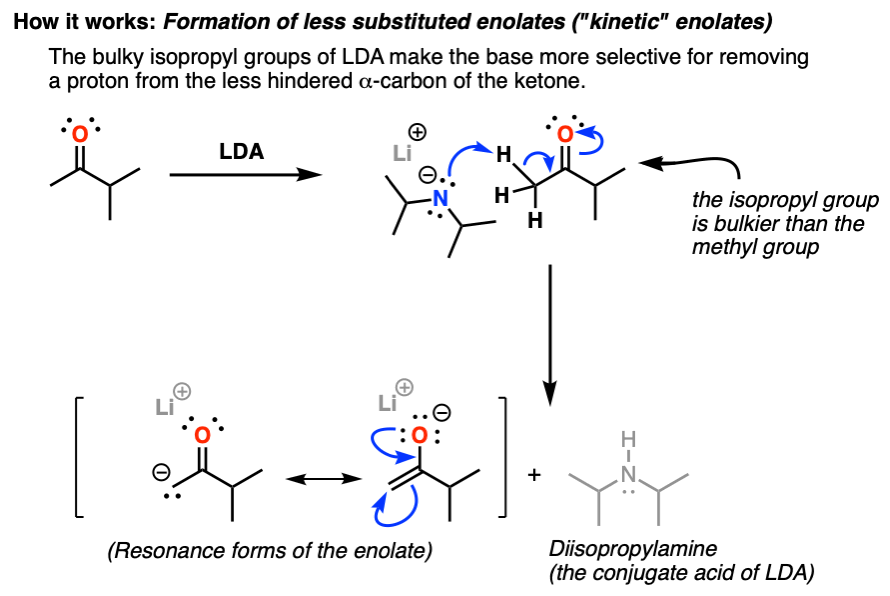
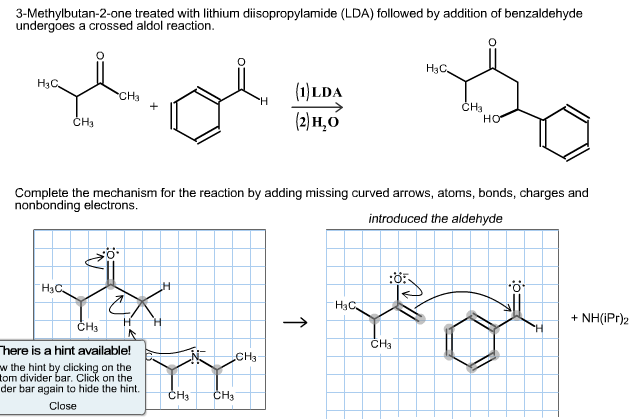
Lithium diisopropylamide (LDA): Properties as a selective and bulky base for Enolate generation. - YouTube

Enders SAMP/RAMP hydrazone-alkylation reaction Lithium diisopropylamide Enamine, Mechanism, blue, angle, white png | PNGWing

Syntheses of silyl triflates 19, 20, 23 and 24 Common intermediate 16... | Download Scientific Diagram
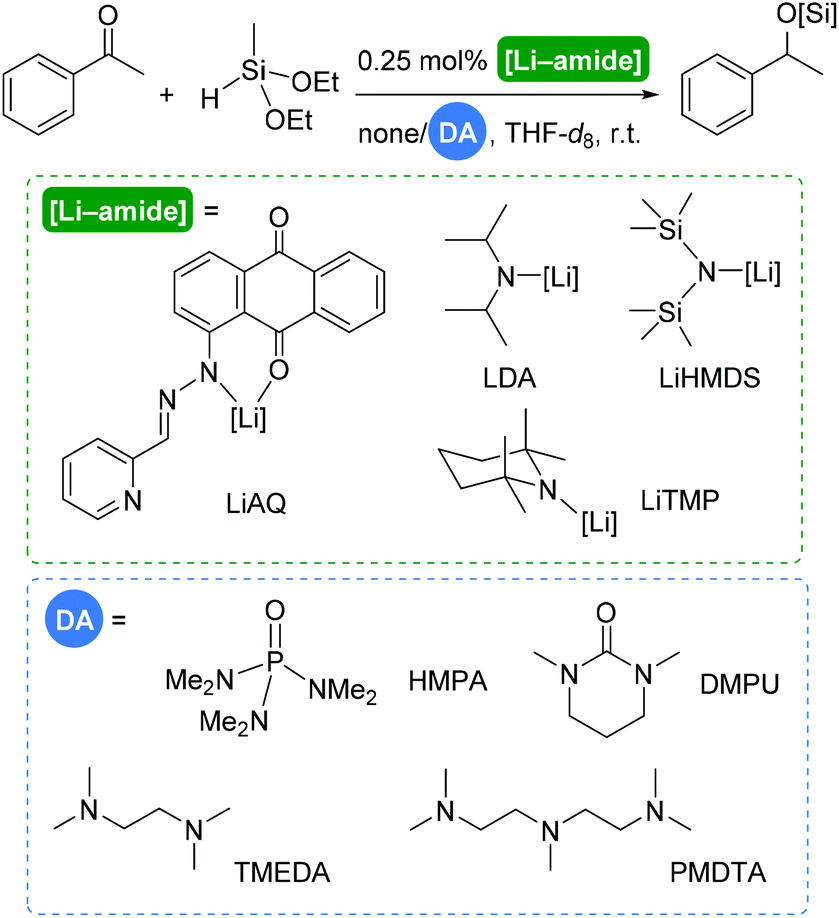
Accelerating role of deaggregation agents in lithium-catalysed hydrosilylation of carbonyl compounds - Dalton Transactions (RSC Publishing) DOI:10.1039/D0DT01540G