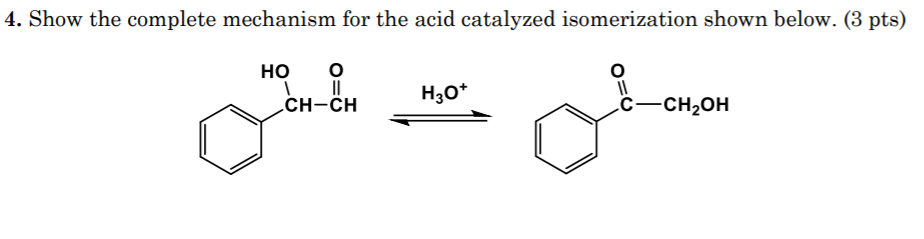
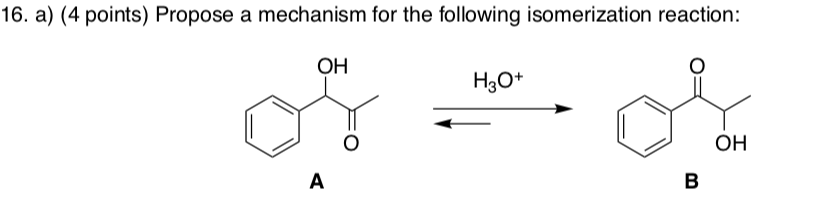
Compound A is difficult to prepare owing to its ready base-catalyzed isomerization to compound B. (1) Write a reasonable mechanism for this isomerization reaction. (2) Which compound is more stable? | Study.com
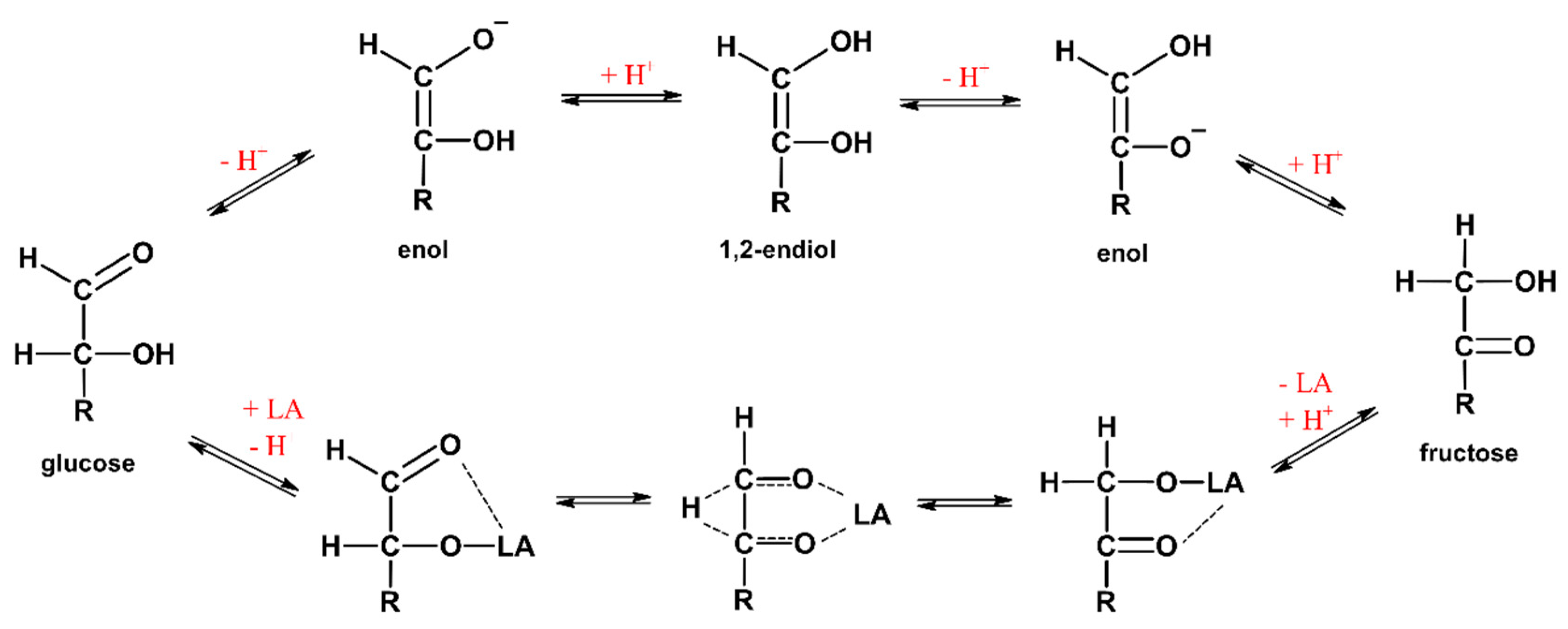
Processes | Free Full-Text | Isomerization of Glucose to Fructose in Hydrolysates from Lignocellulosic Biomass Using Hydrotalcite | HTML

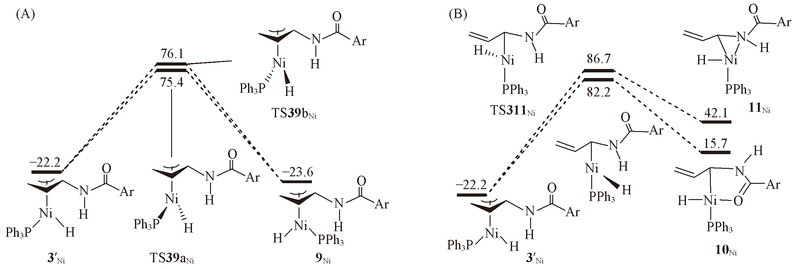
Radical Isomerization and Cycloisomerization Initiated by H• Transfer | Journal of the American Chemical Society
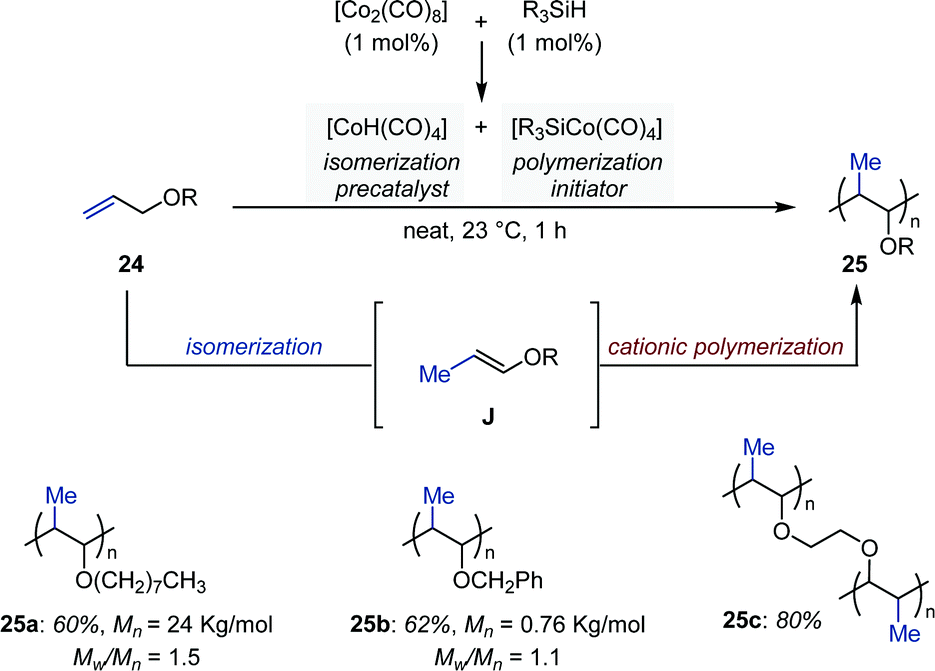
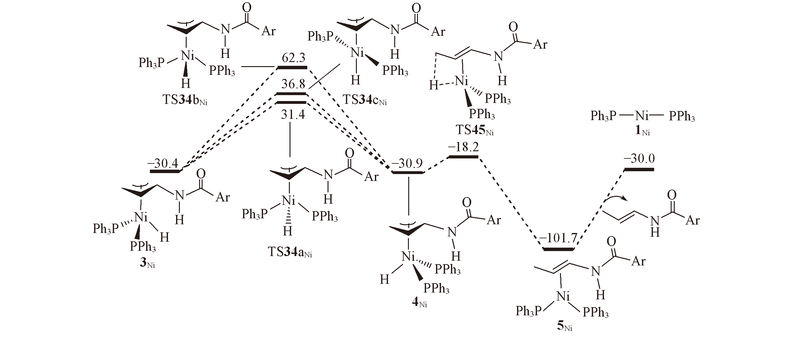
Transition metal-catalyzed alkene isomerization as an enabling technology in tandem, sequential and domino processes - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D0CS00449A
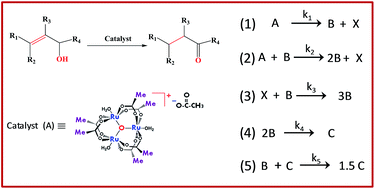
A new mechanism for allylic alcohol isomerization involving ruthenium nanoparticles as a 'true catalyst' generated through the self-assembly of supramolecular triruthenium clusters - RSC Advances (RSC Publishing)
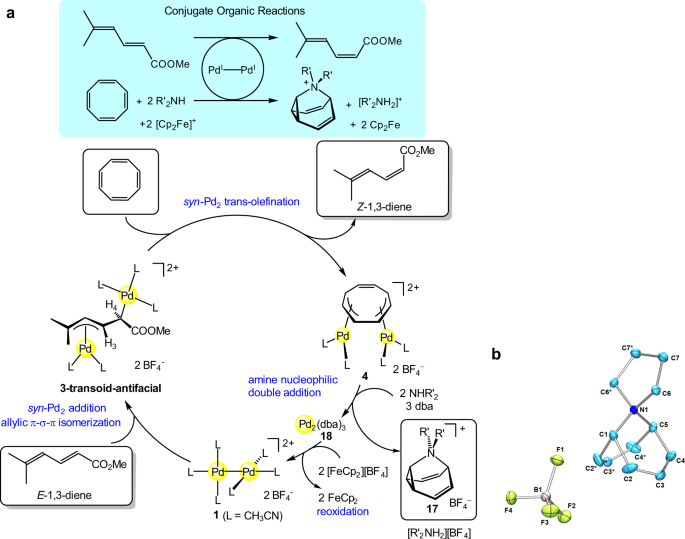
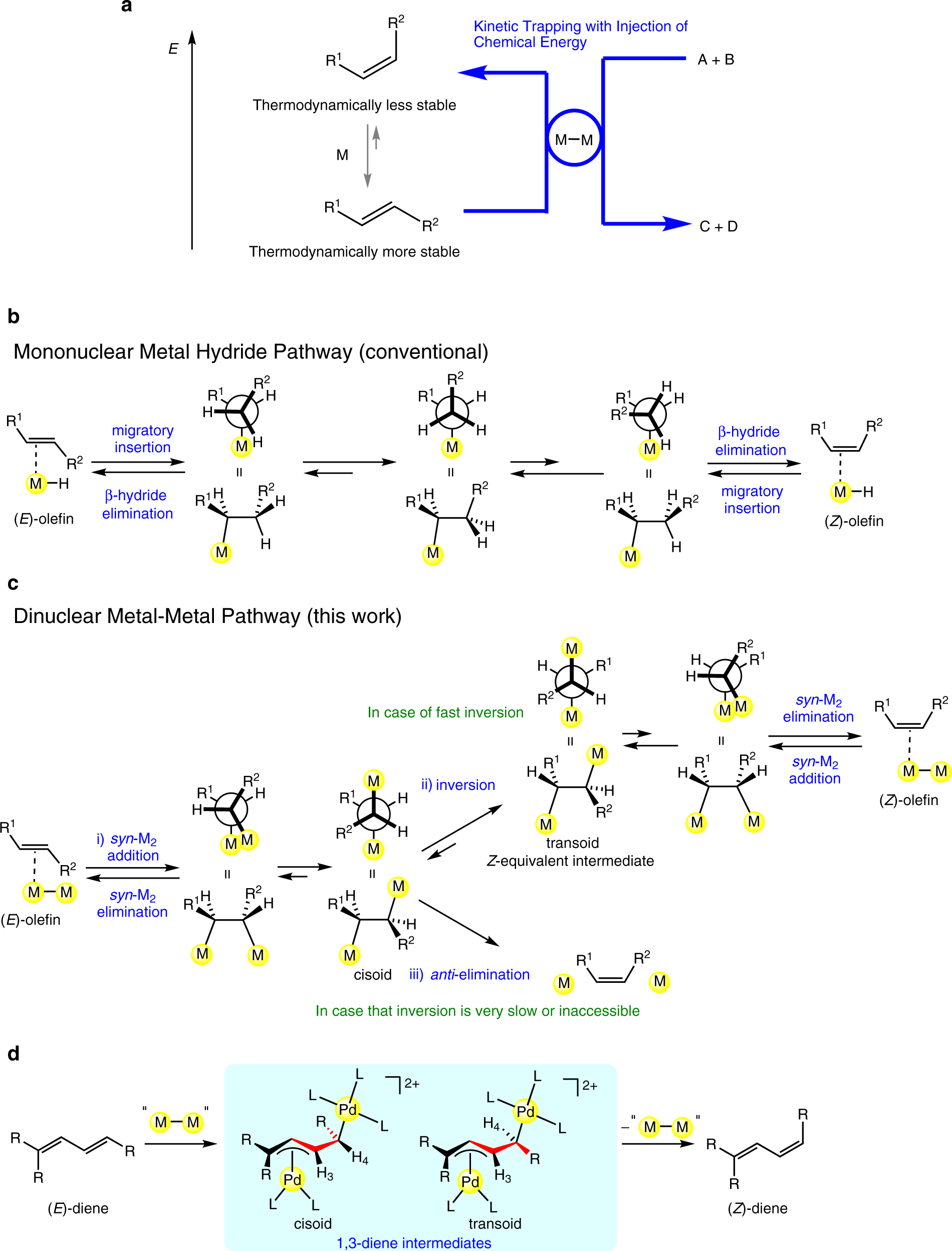
Selective E to Z isomerization of 1,3-Dienes Enabled by A Dinuclear Mechanism | Nature Communications

Compound A is difficult to prepare owing to its ready base-catalyzed isomerization to compound B. (1) Write a reasonable mechanism for this isomerization reaction. (2) Which compound is more stable? | Study.com

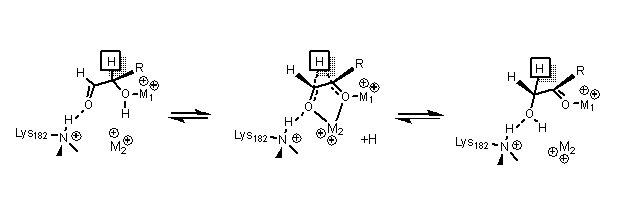
Mechanism of Glucose Isomerization Using a Solid Lewis Acid Catalyst in Water - Román‐Leshkov - 2010 - Angewandte Chemie International Edition - Wiley Online Library
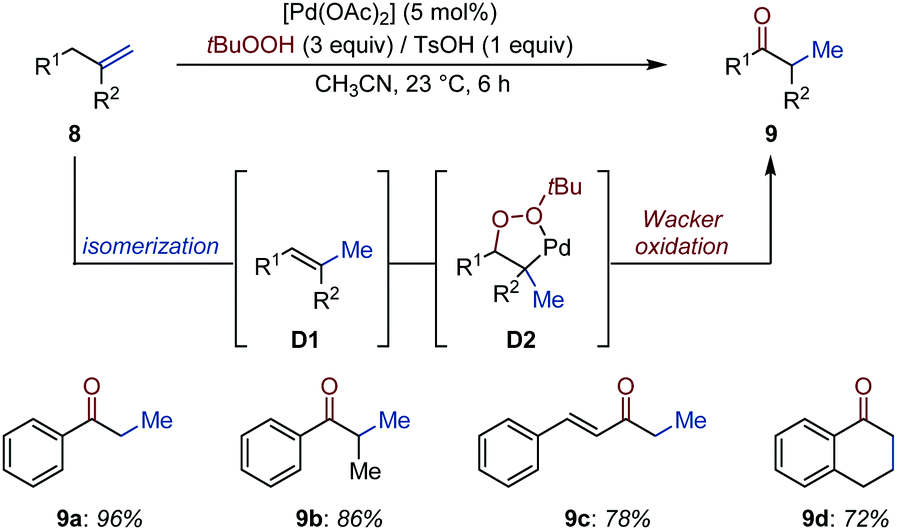
Transition metal-catalyzed alkene isomerization as an enabling technology in tandem, sequential and domino processes - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D0CS00449A

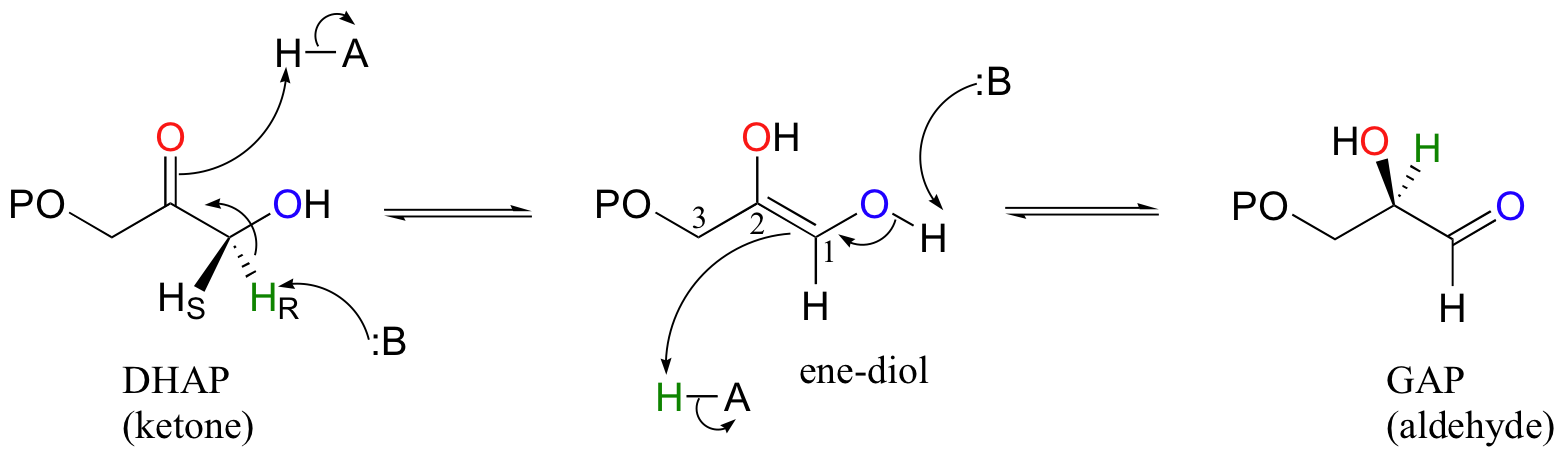
Evidence for tunneling in base-catalyzed isomerization of glyceraldehyde to dihydroxyacetone by hydride shift under formose conditions | PNAS