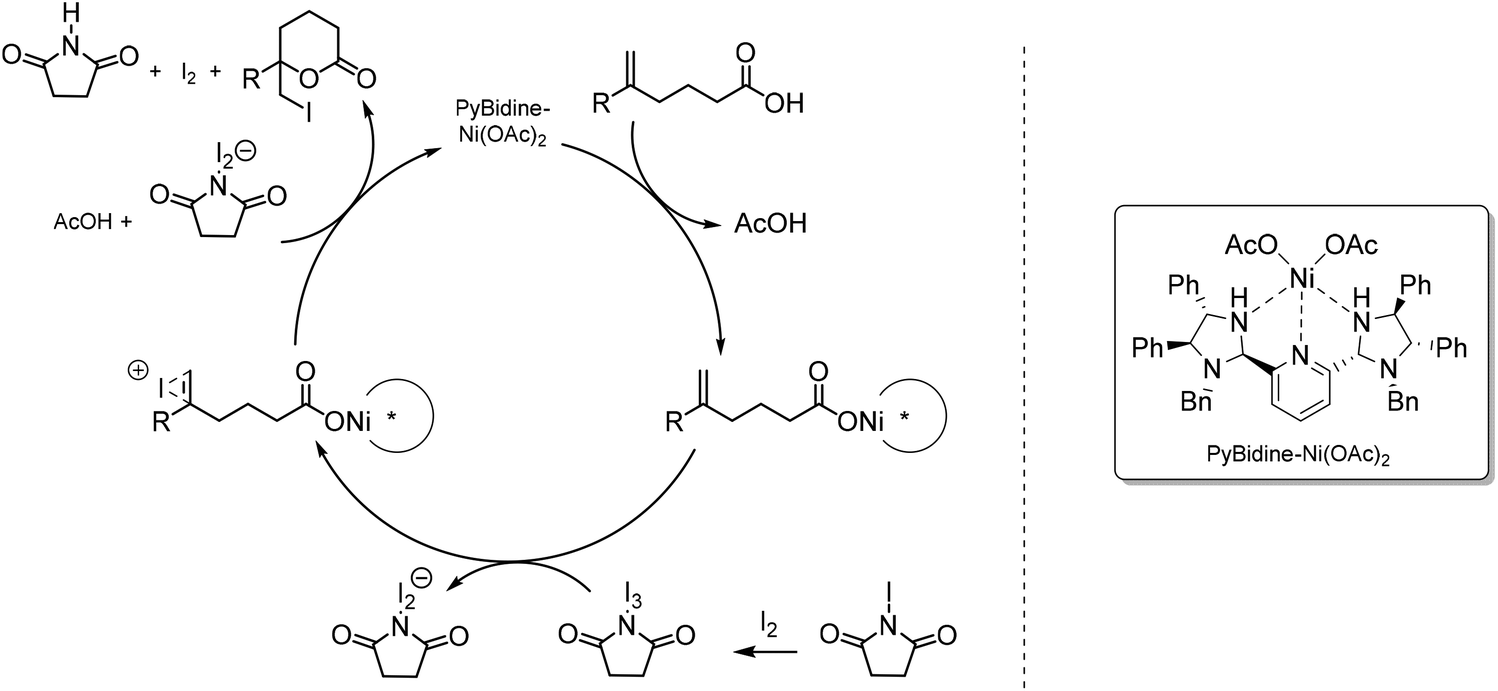
Asymmetric Iodolactonization: An Evolutionary Account - Nolsøe - 2014 - European Journal of Organic Chemistry - Wiley Online Library
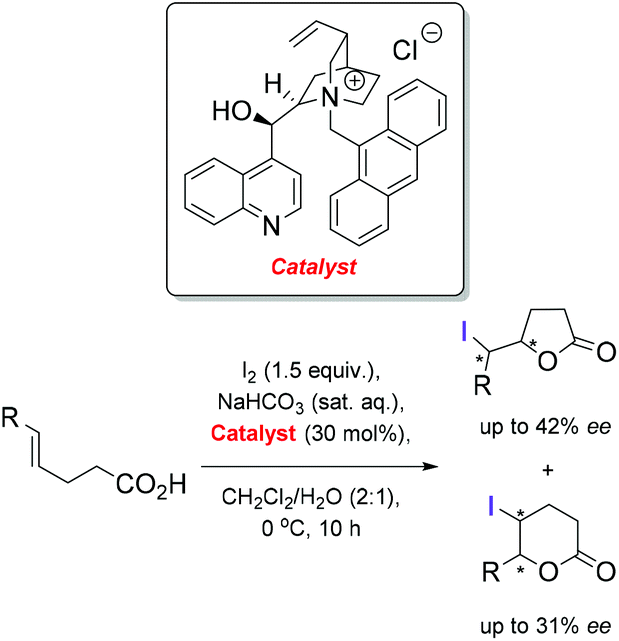
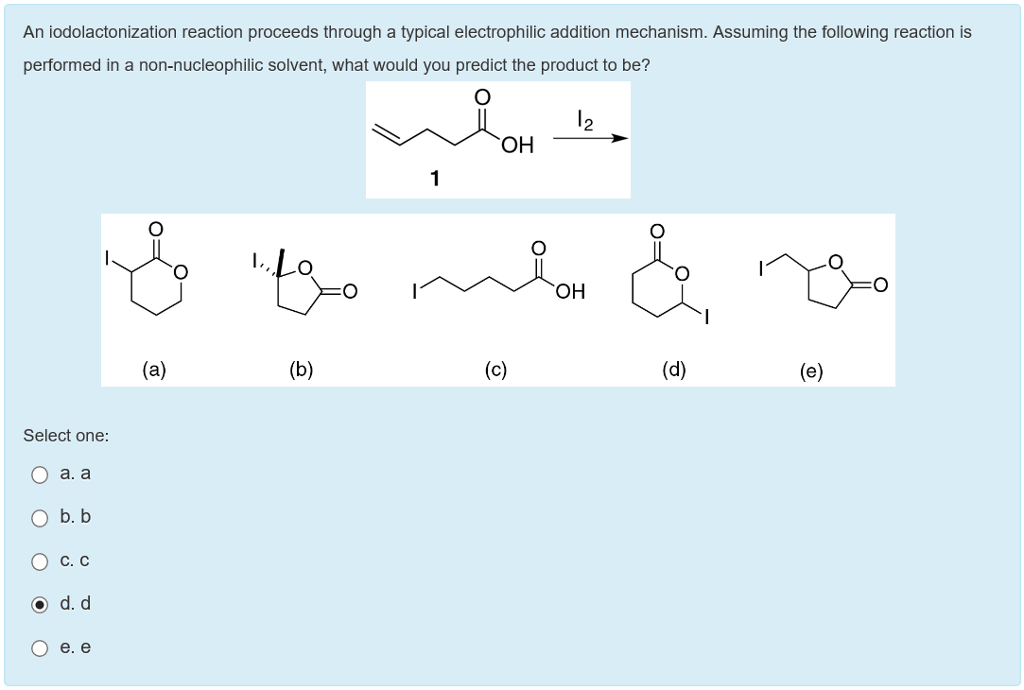
Catalytic enantioselective iodolactonization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB03160F

Asymmetric Iodolactonization: An Evolutionary Account - Nolsøe - 2014 - European Journal of Organic Chemistry - Wiley Online Library
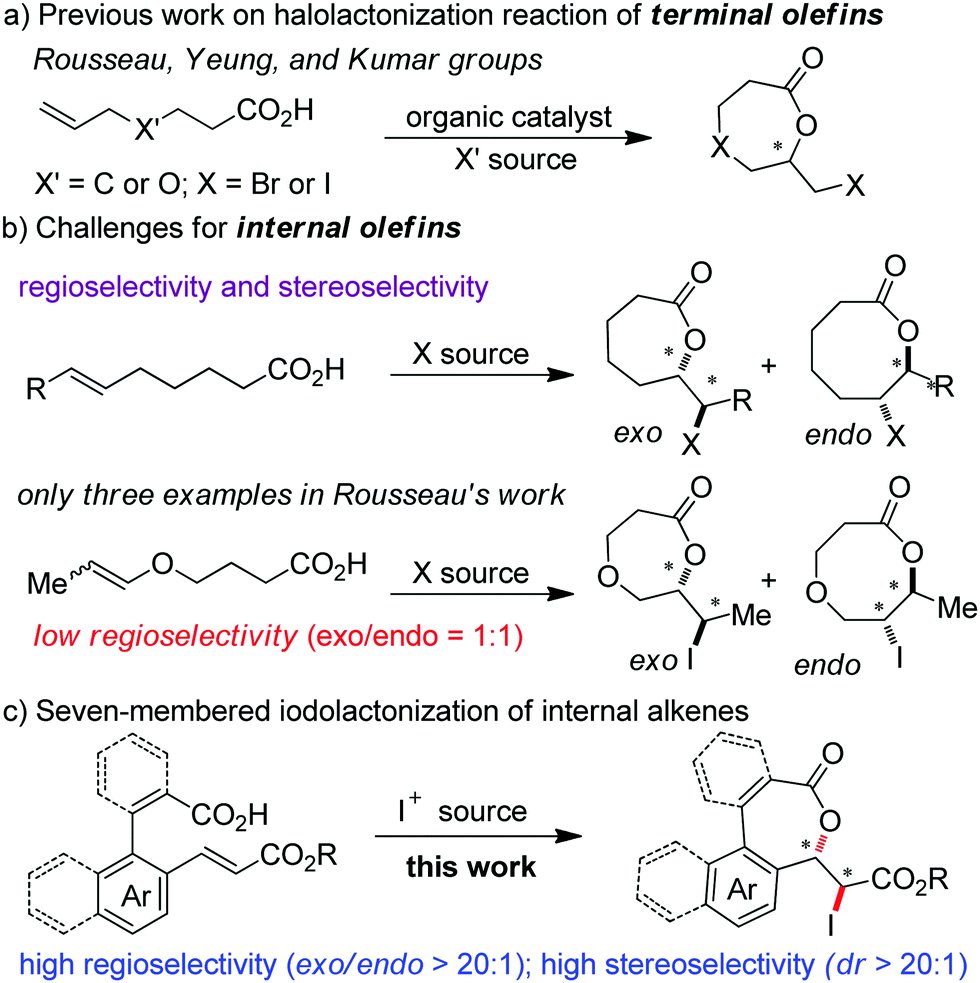
Synthesis of seven-membered lactones by regioselective and stereoselective iodolactonization of electron-deficient olefins - Chemical Communications (RSC Publishing) DOI:10.1039/C9CC10080F
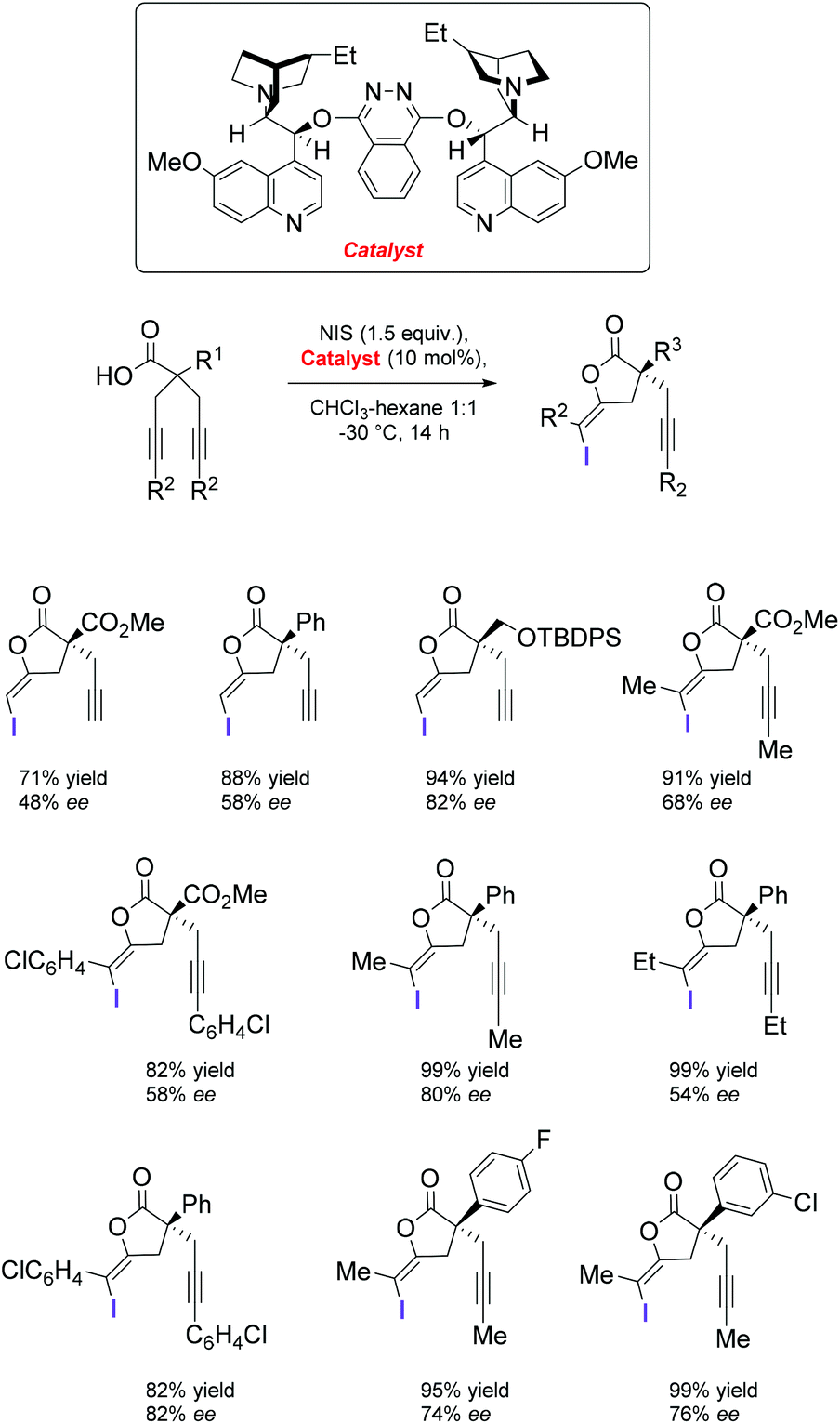
Catalytic enantioselective iodolactonization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB03160F
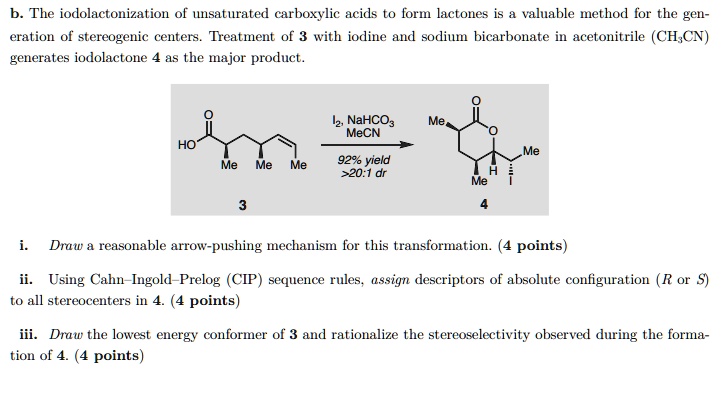
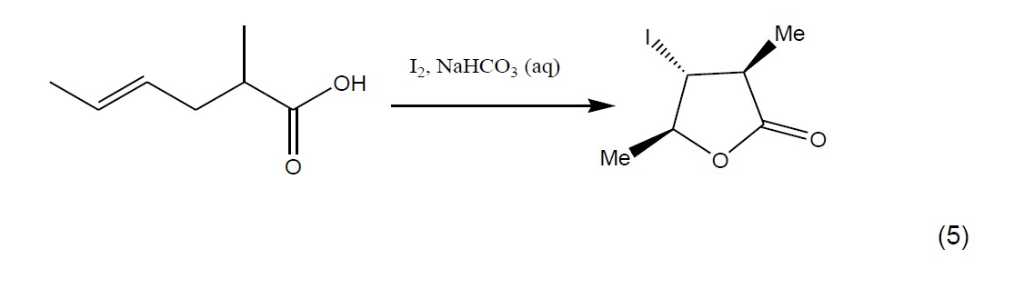
SOLVED:The iodolactonization of unsaturated carboxylic acids to form lactones is valuable method for the gen- eration of stereogenic centers_ Treatment of 3 with iodine and sodium bicarbonate in acetonitrile (CH,CN) generates iodolactone
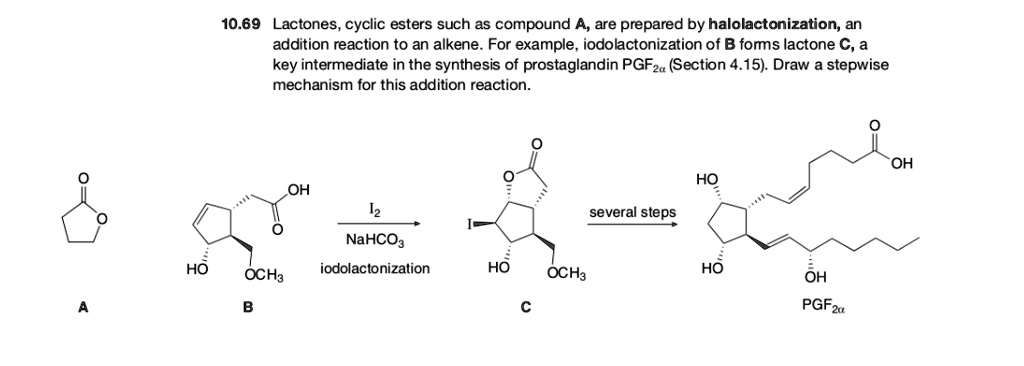
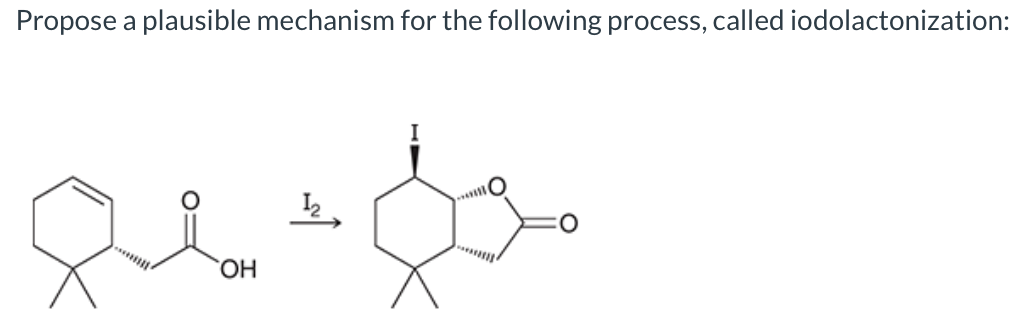
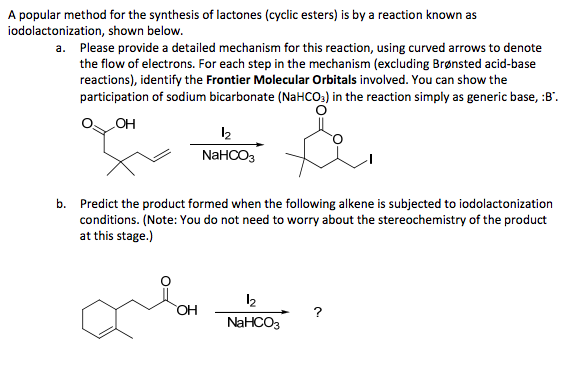
SOLVED:10.69 Lactones cyclic esters such as compound A, are prepared by halolactonization, an addition reaction t0 an alkene For example , iodolactonization of B foms lactone C, a key intermediate in the

Asymmetric Iodolactonization: An Evolutionary Account - Nolsøe - 2014 - European Journal of Organic Chemistry - Wiley Online Library