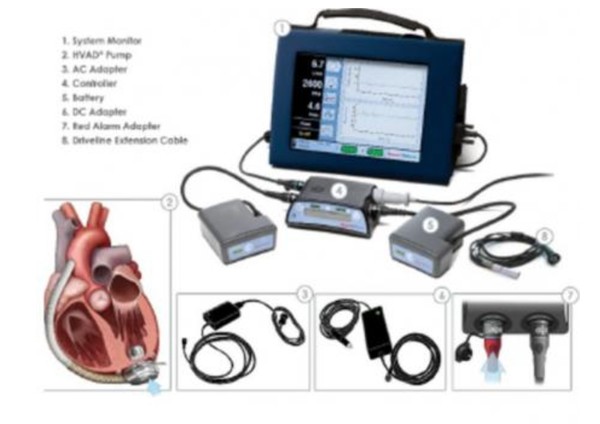
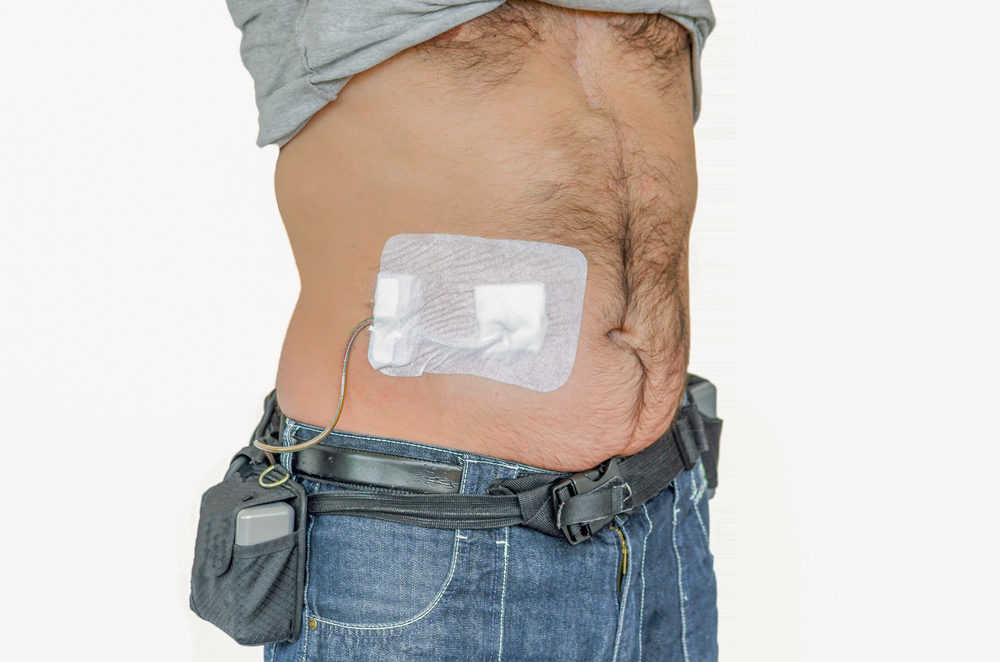
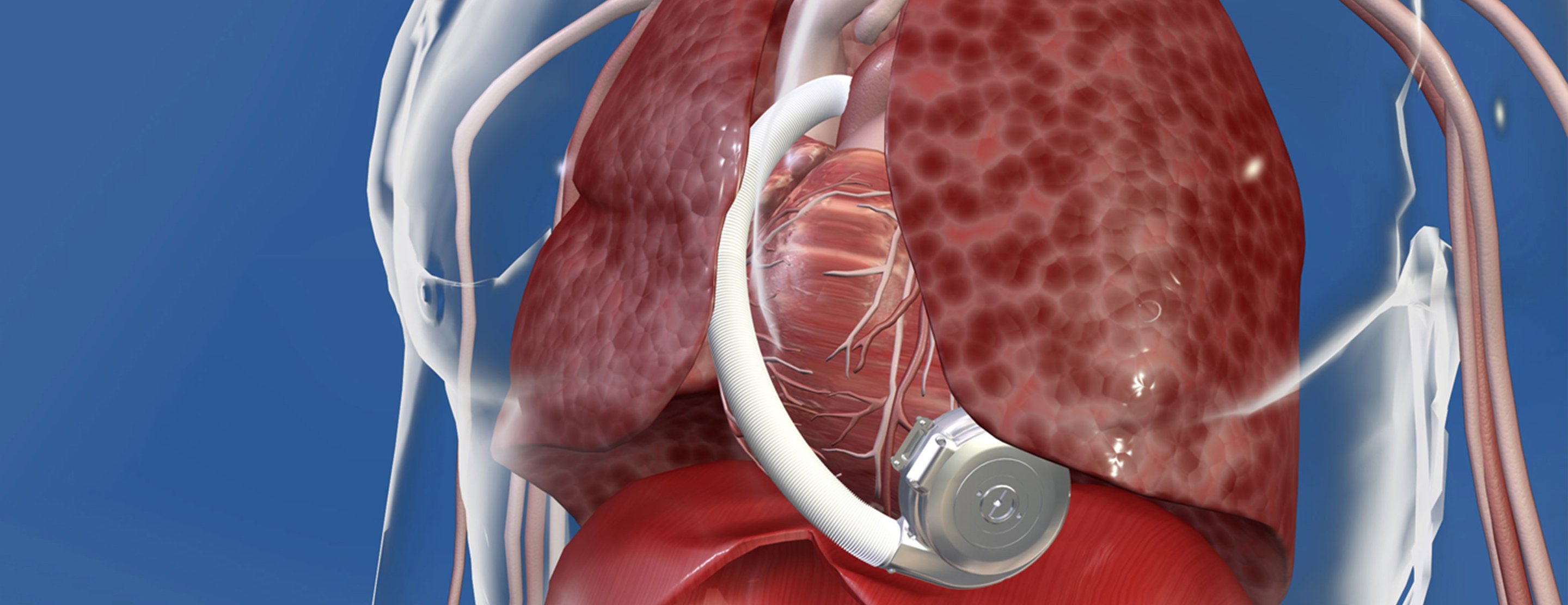
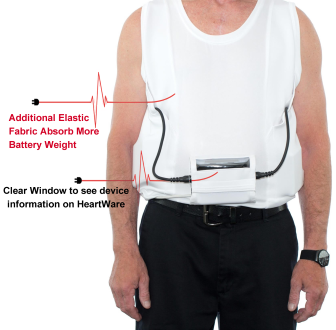
HeartWare Ventricular Assist Device AC adapters and batteries | Therapeutic Goods Administration (TGA)

HeartWare Ventricular Assist Device AC adapters and batteries | Therapeutic Goods Administration (TGA)
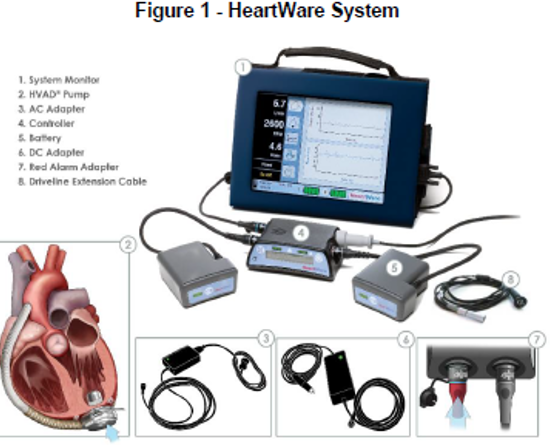
Medtronic, Inc. Recalls Instructions for Use and Patient Manual for HeartWare HVAD System to Update Information about Carrying Case, Driveline Cover, and Controller Power-Up Issues | FDA

CircuLite Synergy size compared to AA battery. Received from Heartware... | Download Scientific Diagram

REF 1650 Cardiac Pacemaker Batteries 14.4V Li Ion Rechargeable Battery for HeartWare|Replacement Batteries| - AliExpress

Medtronic Recalls HeartWare HVAD Battery Cables, Data Cables, Adapter Cables and Controller 2.0 Ports Due to Risk of Damage to Controller Ports | FDA