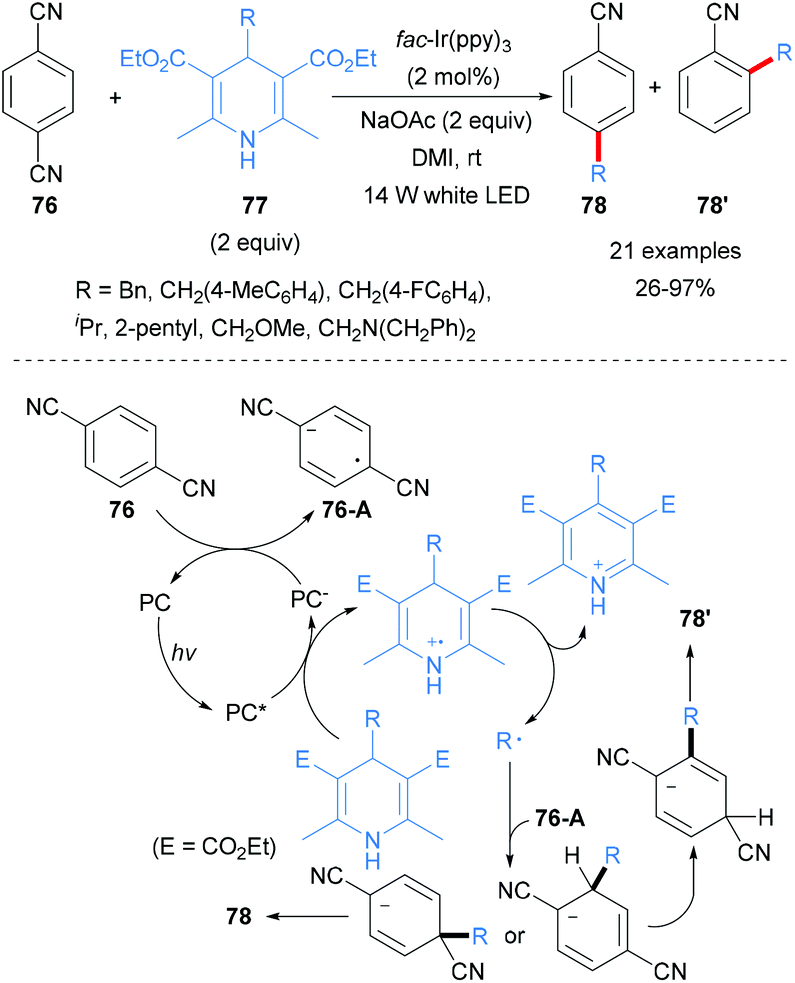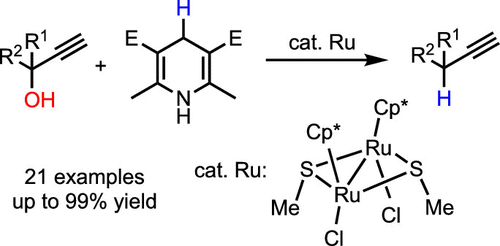
Ruthenium-Catalyzed Propargylic Reduction of Propargylic Alcohols with Hantzsch Ester,Organometallics - X-MOL
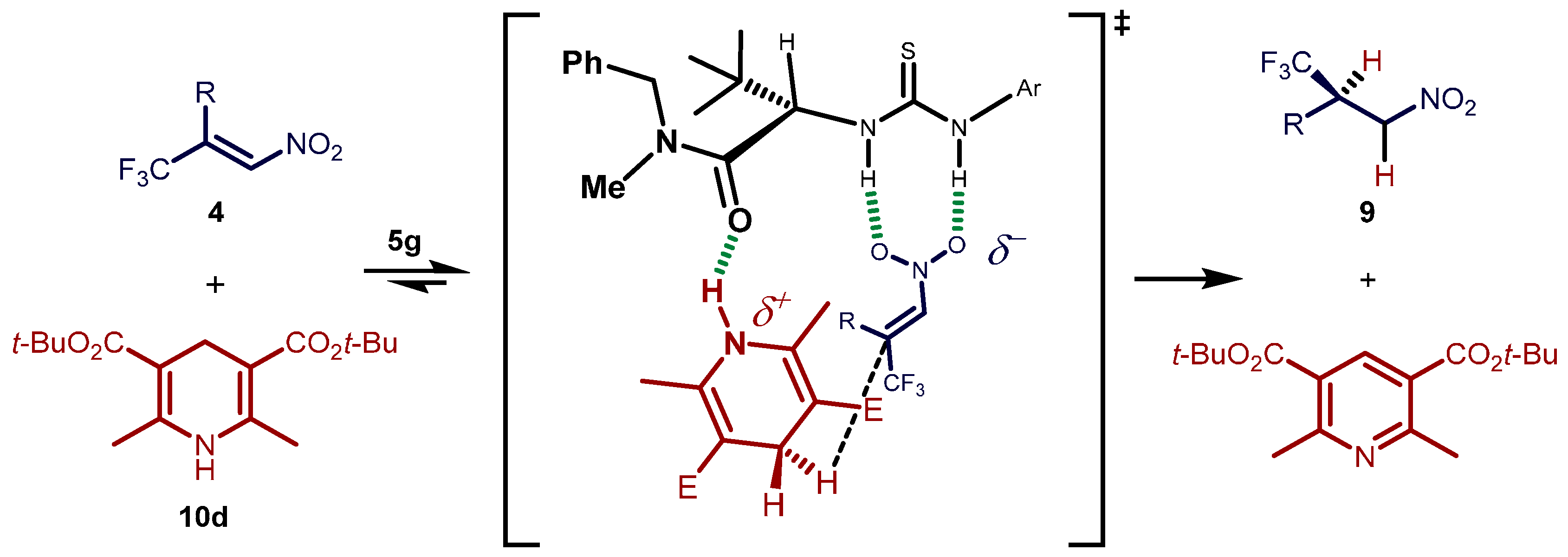
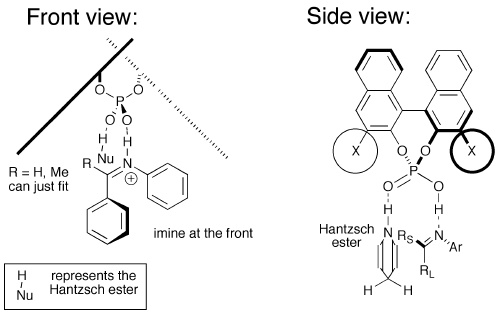
Molecules | Free Full-Text | A General Catalytic Enantioselective Transfer Hydrogenation Reaction of β,β-Disubstituted Nitroalkenes Promoted by a Simple Organocatalyst | HTML
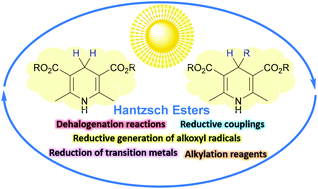
Hantzsch esters: an emerging versatile class of reagents in photoredox catalyzed organic synthesis - Organic & Biomolecular Chemistry (RSC Publishing)

Proposed mechanism for the oxidation of Hantzsch dihydropyridines by... | Download Scientific Diagram
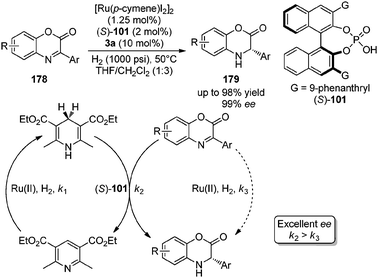
Transfer hydrogenation with Hantzsch esters and related organic hydride donors - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C1CS15268H

Mechanism of the highly selective transfer hydrogenation using Hantzsch... | Download Scientific Diagram

Reaction design and mechanistic proposal. One chiral rhodium catalyst... | Download Scientific Diagram
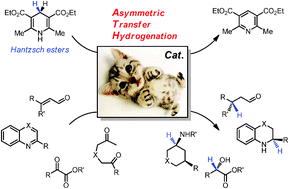
Transfer hydrogenation with Hantzsch esters and related organic hydride donors - Chemical Society Reviews (RSC Publishing)

Substituted Hantzsch Esters as Versatile Radical Reservoirs in Photoredox Reactions - Gu - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

Molecules | Free Full-Text | A General Catalytic Enantioselective Transfer Hydrogenation Reaction of β,β-Disubstituted Nitroalkenes Promoted by a Simple Organocatalyst | HTML
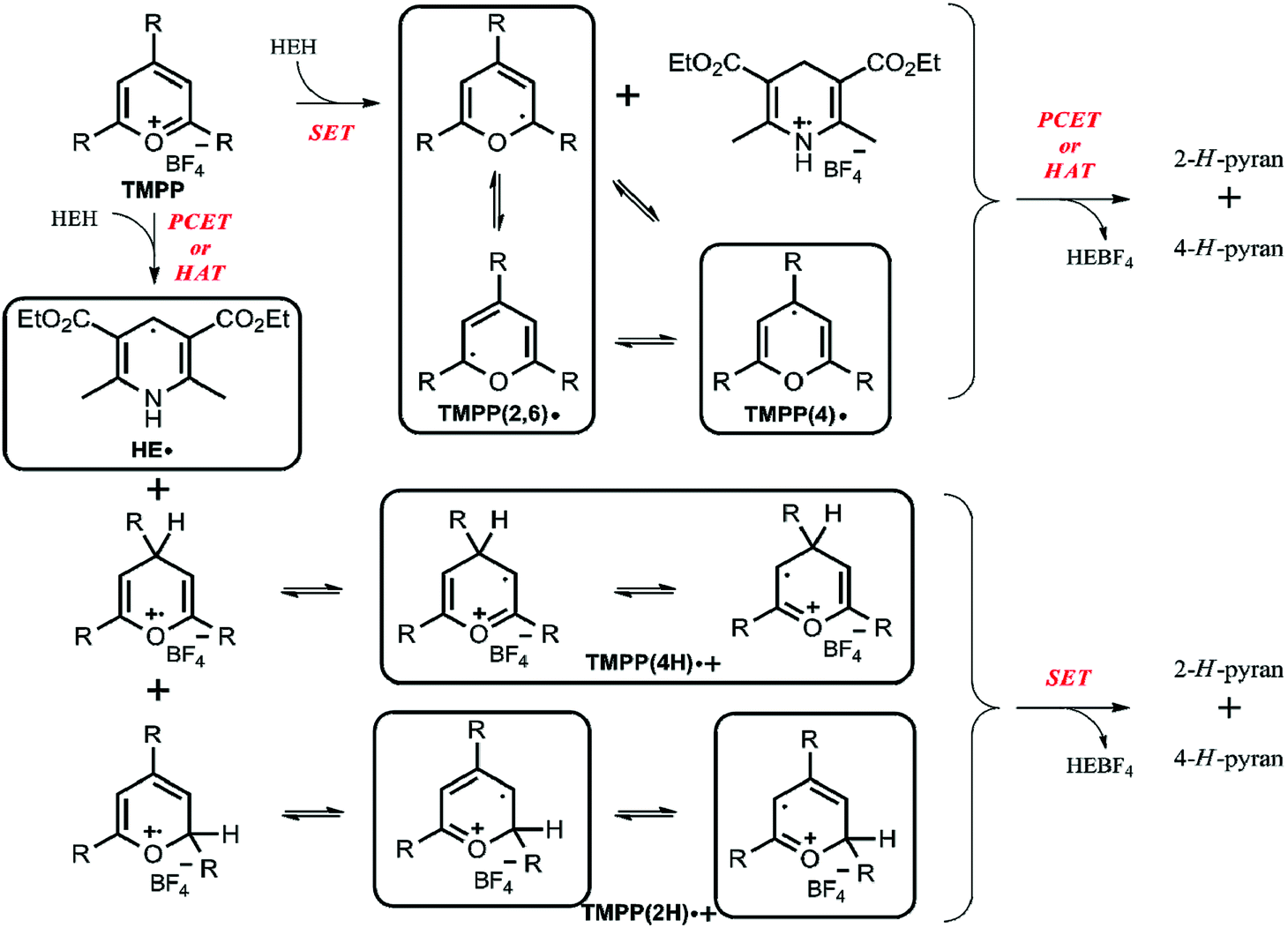
Electron paramagnetic resonance spectroscopic studies of the electron transfer reaction of Hantzsch ester and a pyrylium salt - RSC Advances (RSC Publishing) DOI:10.1039/C8RA05693E