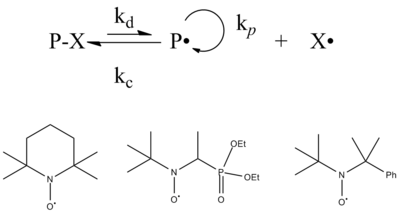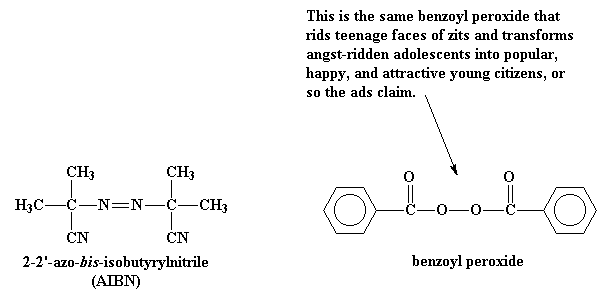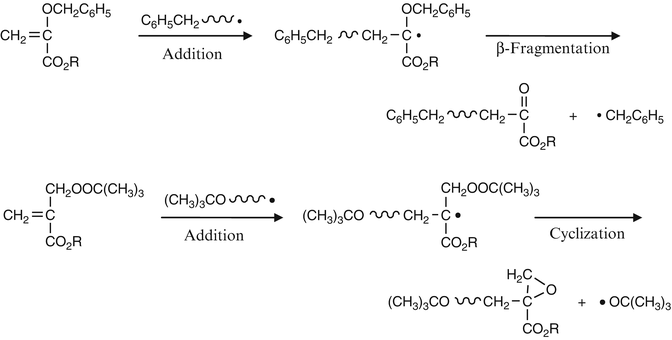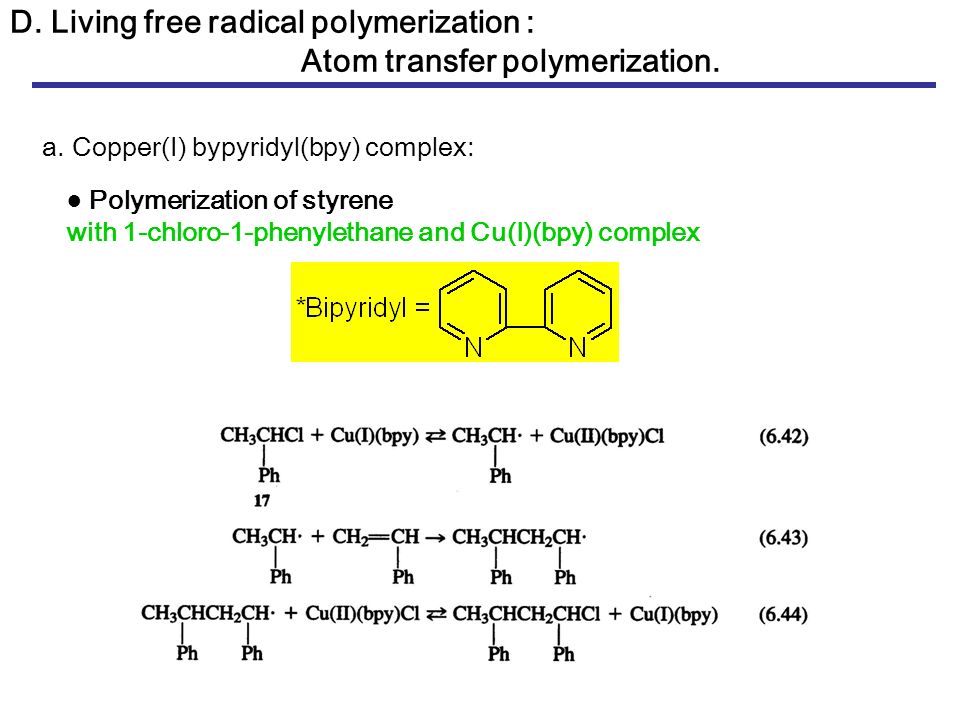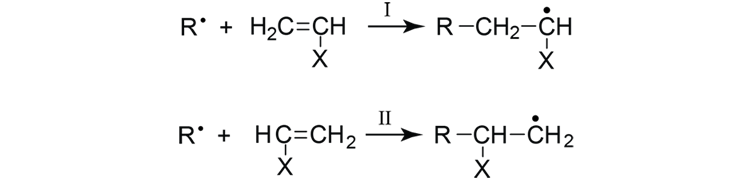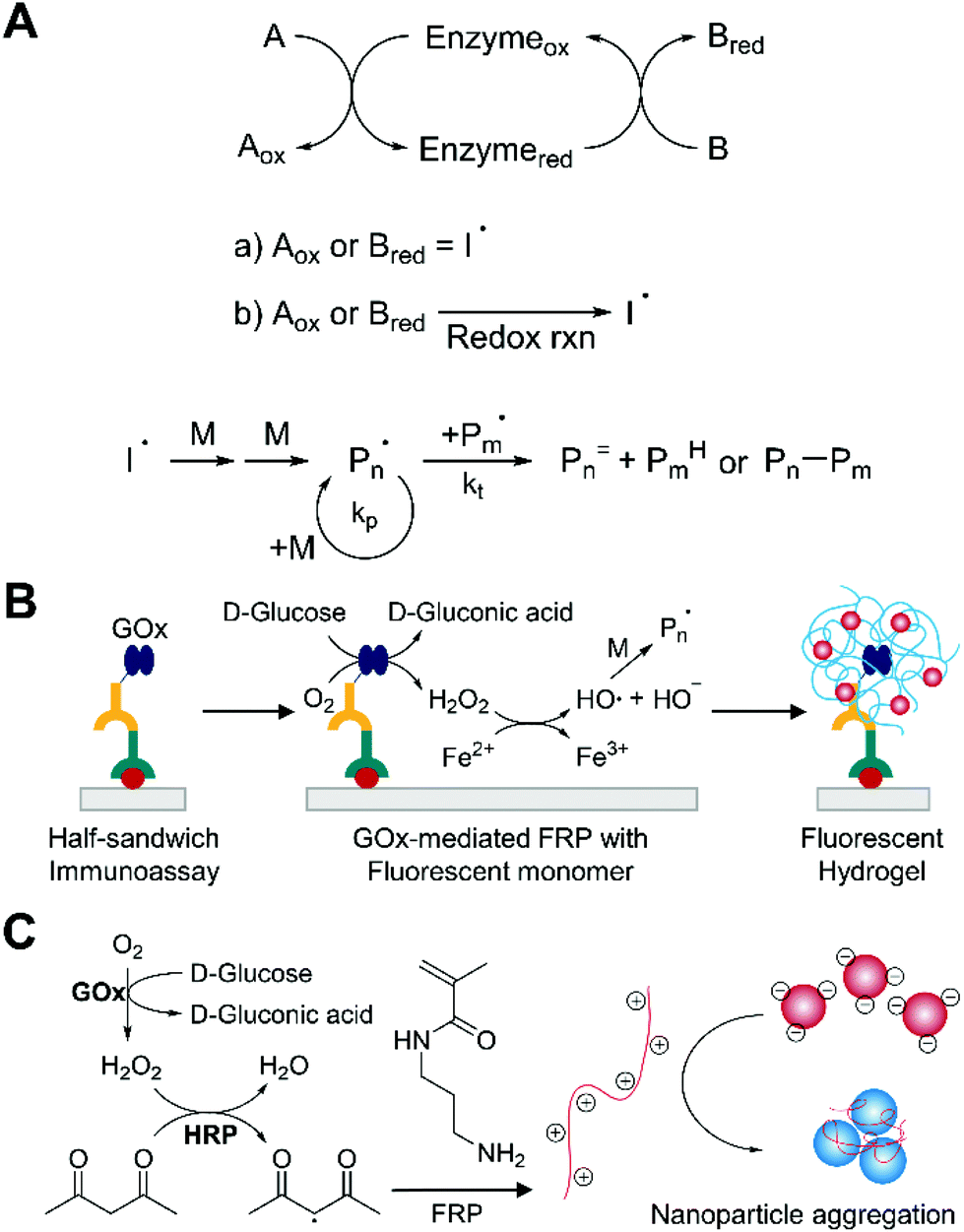
Radical polymerization reactions for amplified biodetection signals - Polymer Chemistry (RSC Publishing) DOI:10.1039/C9PY01801H

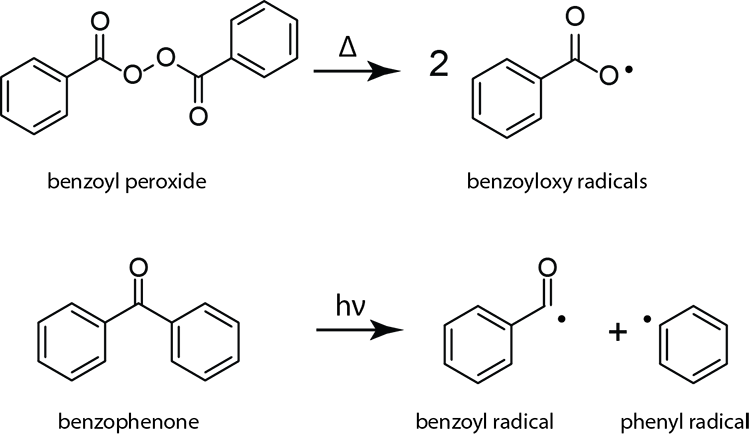
Free radical initiation mechanism based on the combination of alcohol initiators and borane activators - Ohkura - 2014 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

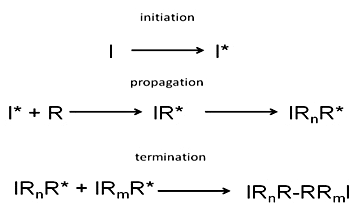
free radical mechanism steps for polymerisation of ethene to poly(ethene) polythene reagents reaction conditions organic synthesis

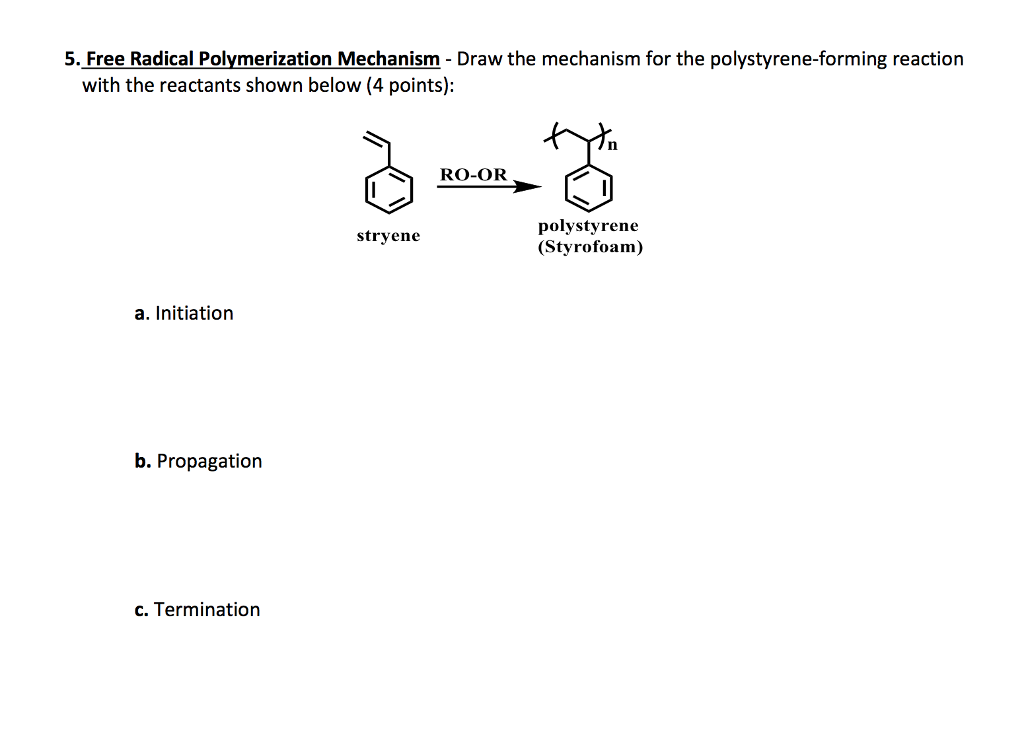
What is an addition polymerization? Explain the free radical addition polymerization mechanism by taking an example.


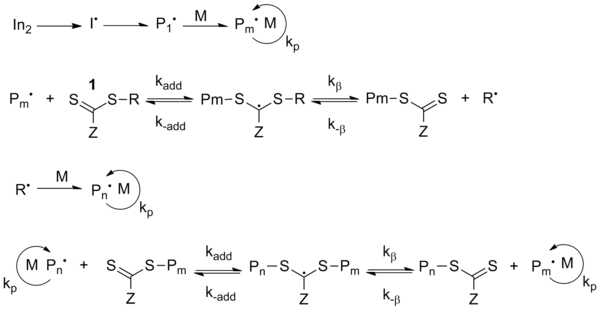

![PDF] Free-Radical Chain-Growth Polymerization | Semantic Scholar PDF] Free-Radical Chain-Growth Polymerization | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ab8d8dd2582c0cfa5e6a8dd8c2237311317953b9/8-Table3.1-2.png)