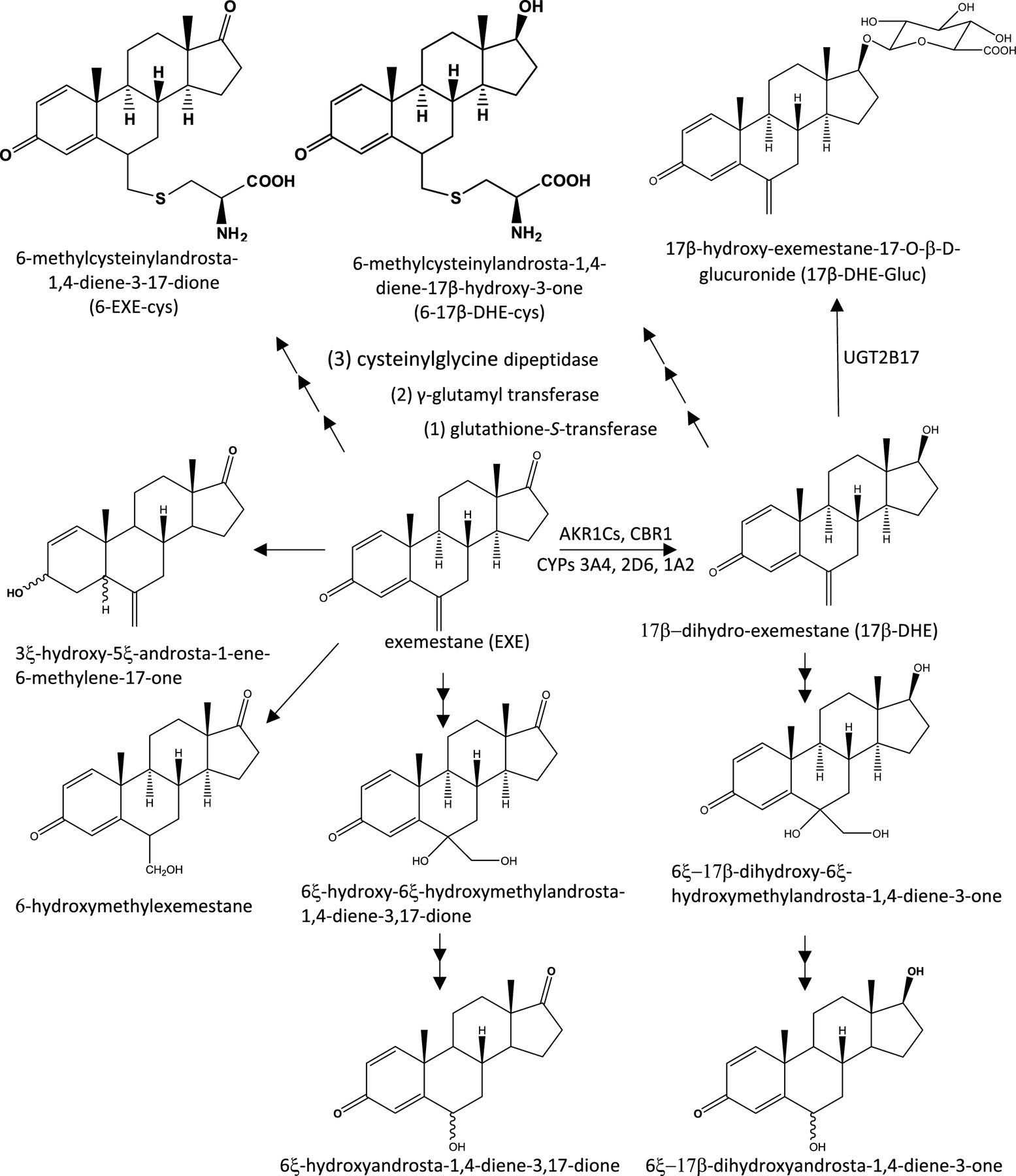
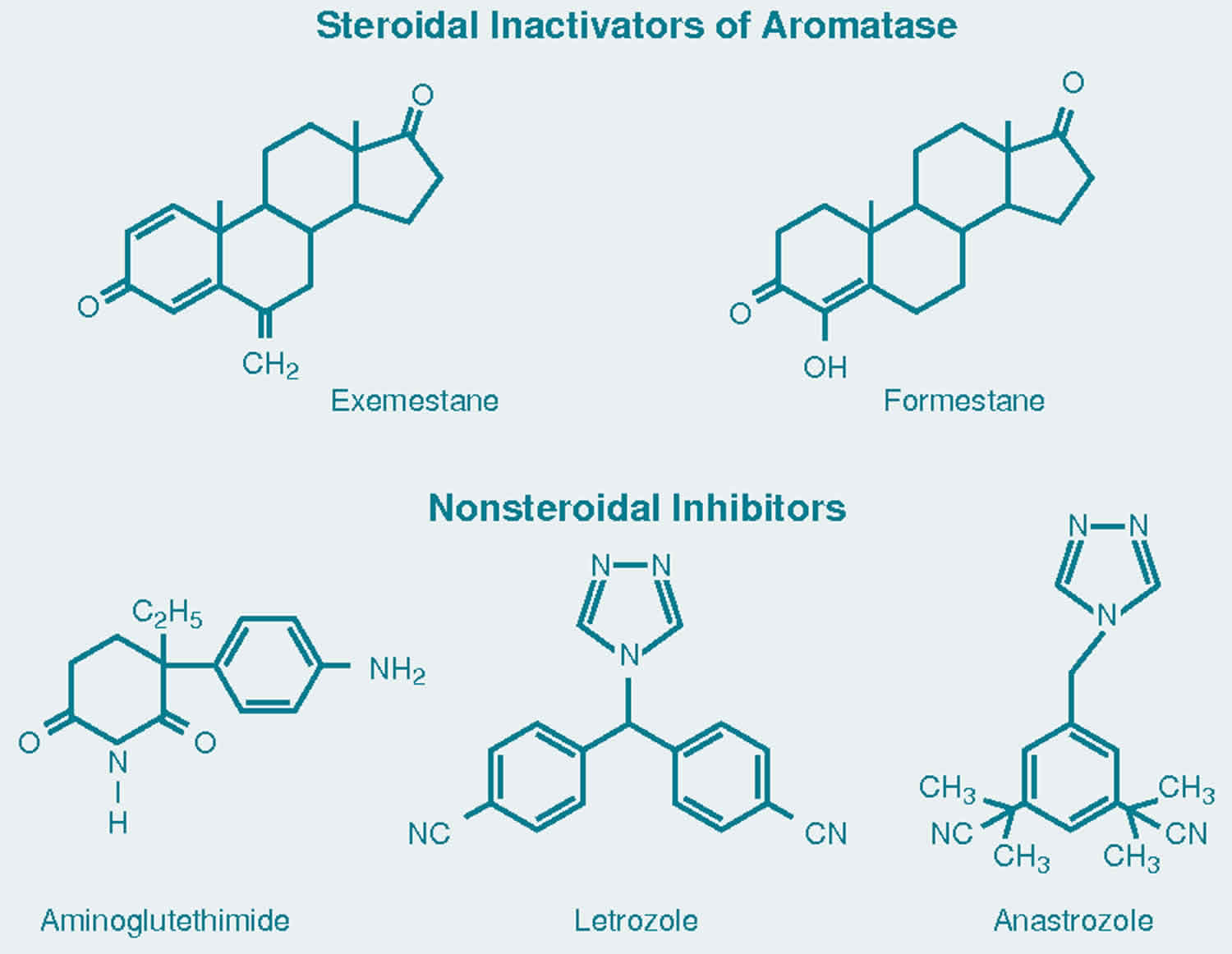
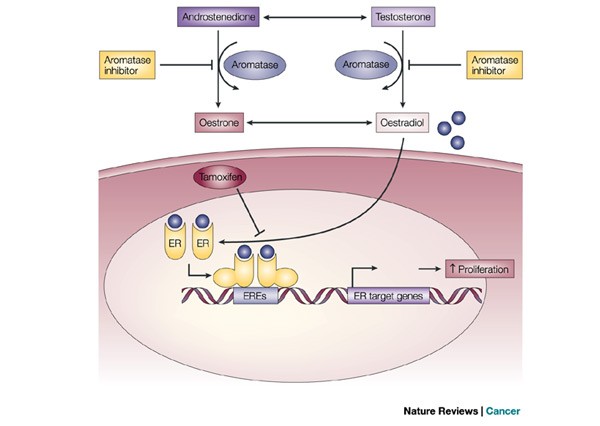
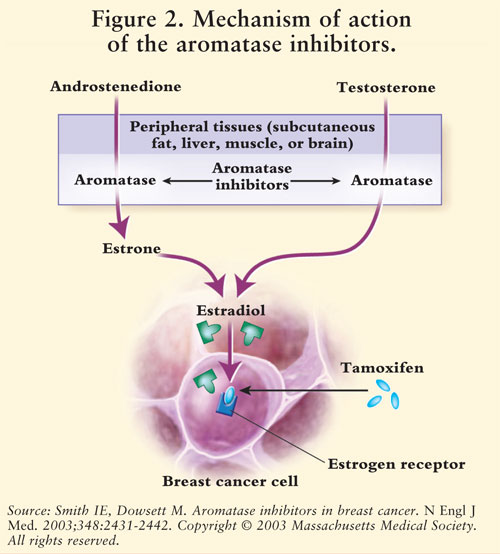
JCI Insight - Pharmacogenomics of aromatase inhibitors in postmenopausal breast cancer and additional mechanisms of anastrozole action

Hypothesized mechanism of action of exemestane on MPM cells. The GPCR... | Download Scientific Diagram

Metabolism and Toxicological Implications of Commonly Used Chemopreventive Drugs Against Breast Cancer/Carcinogenesis | Bentham Science

Tucidinostat plus exemestane for postmenopausal patients with advanced, hormone receptor-positive breast cancer (ACE): a randomised, double-blind, placebo-controlled, phase 3 trial - The Lancet Oncology

Changes in serum estrogenic activity during neoadjuvant therapy with letrozole and exemestane - ScienceDirect

Figure 1 from In vivo inhibition of aromatization by exemestane, a novel irreversible aromatase inhibitor, in postmenopausal breast cancer patients. | Semantic Scholar

Breast Cancer Basics: Reducing Estrogen Receptor Stimulation (Without Drugs) – IOE Program by Dr. Brian Lawenda