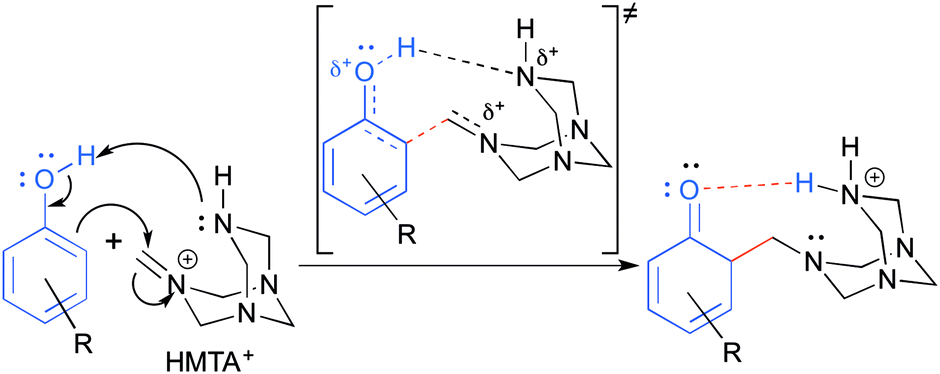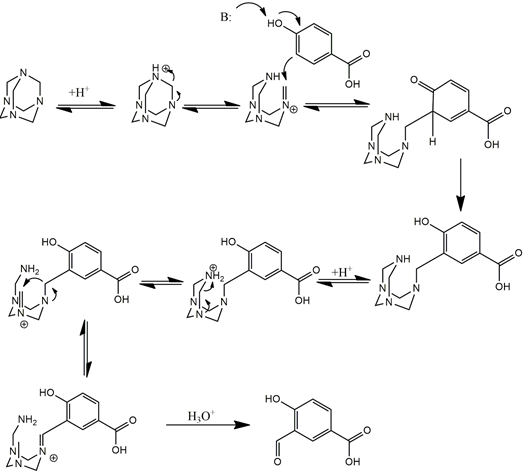
A theoretical study of the Duff reaction: insights into its selectivity - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB01887D
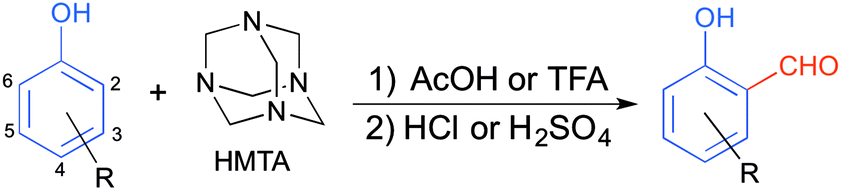
A theoretical study of the Duff reaction: insights into its selectivity - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB01887D
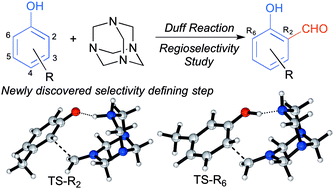
A theoretical study of the Duff reaction: insights into its selectivity - Organic & Biomolecular Chemistry (RSC Publishing)

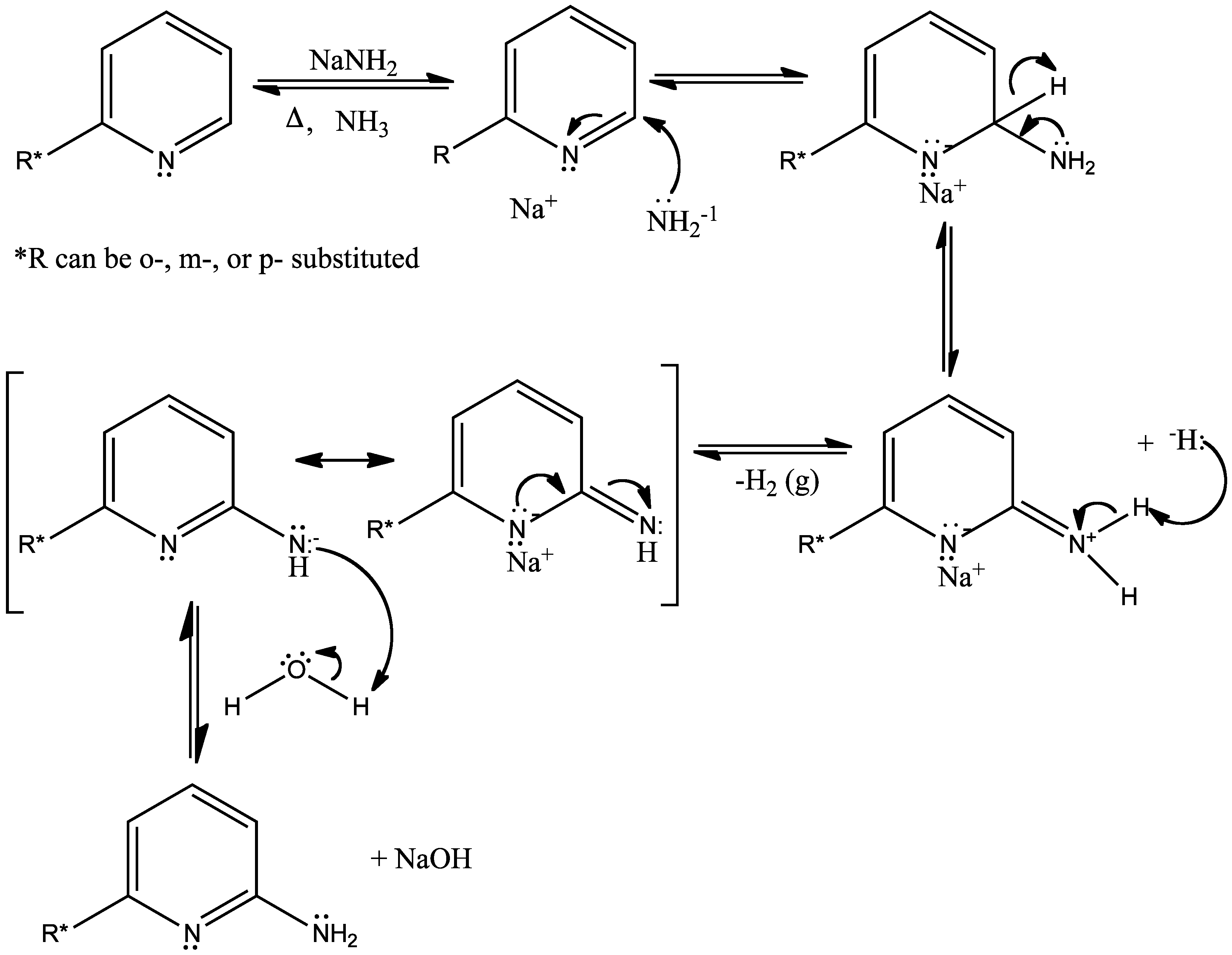
Cross‐Coupling–Elimination Reactions Mediated or Catalyzed by Zirconium Complexes: A Valuable Tool in Organic Synthesis - Fañanás - 2008 - European Journal of Organic Chemistry - Wiley Online Library
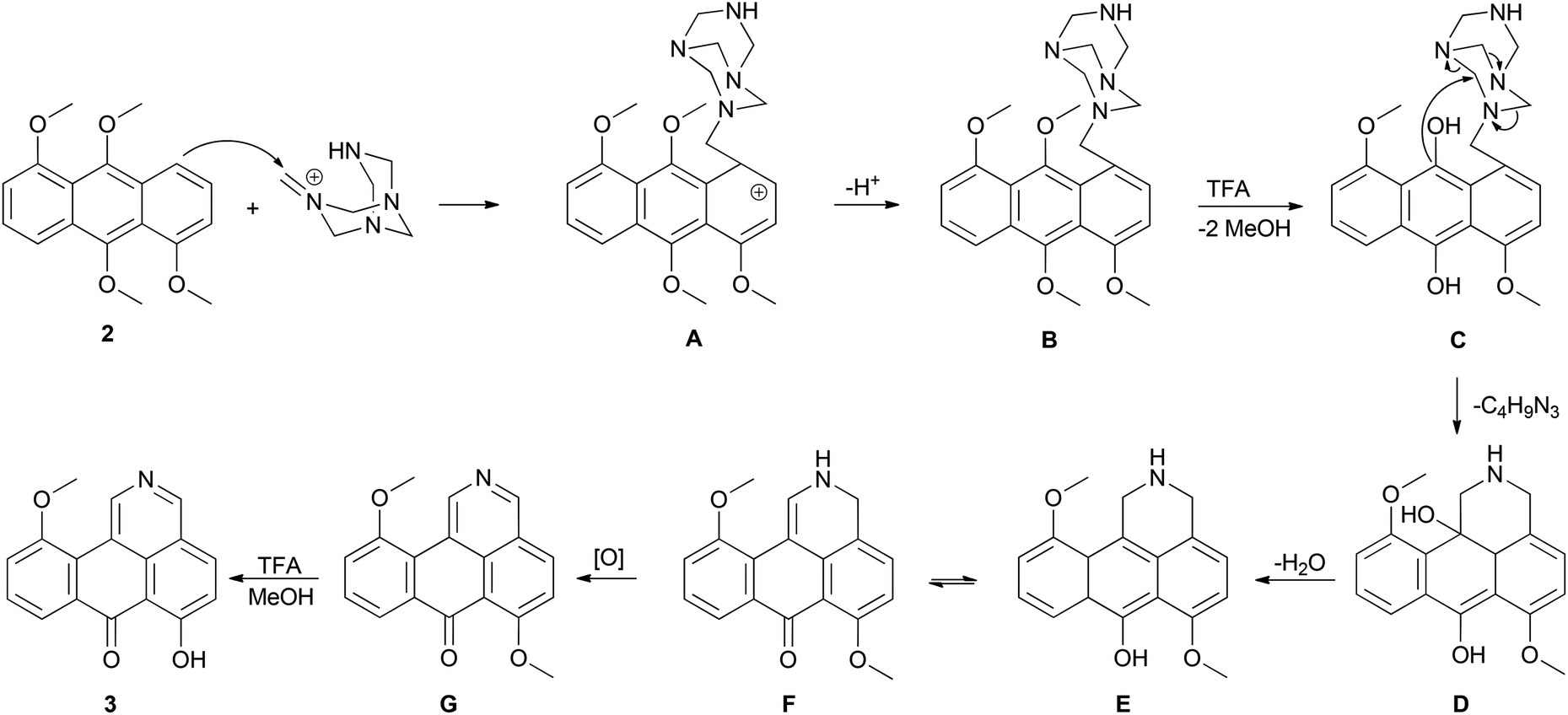
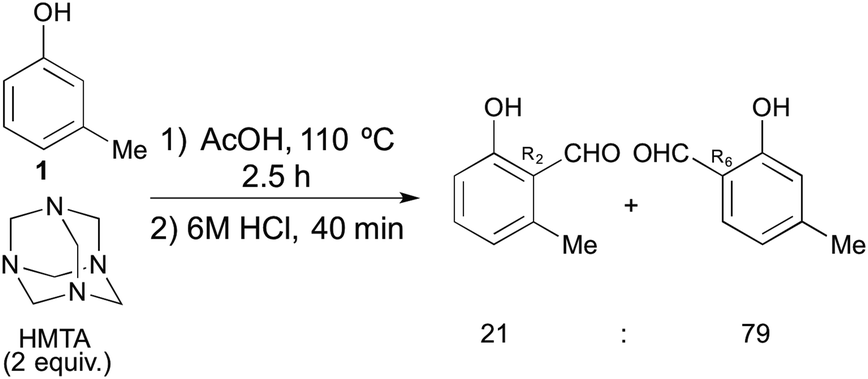
Unexpected formation of π-expanded isoquinoline from anthracene possessing four electron-donating groups via the Duff reaction - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB00981F
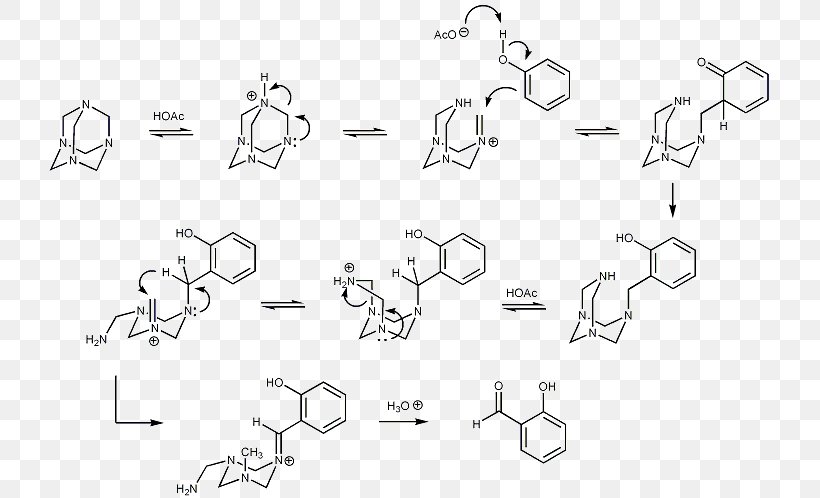
Duff Reaction Formylation Reaction Methenamine Chemical Reaction Organic Chemistry, PNG, 723x498px, Formylation Reaction, Acylation, Aldehyde, Area,
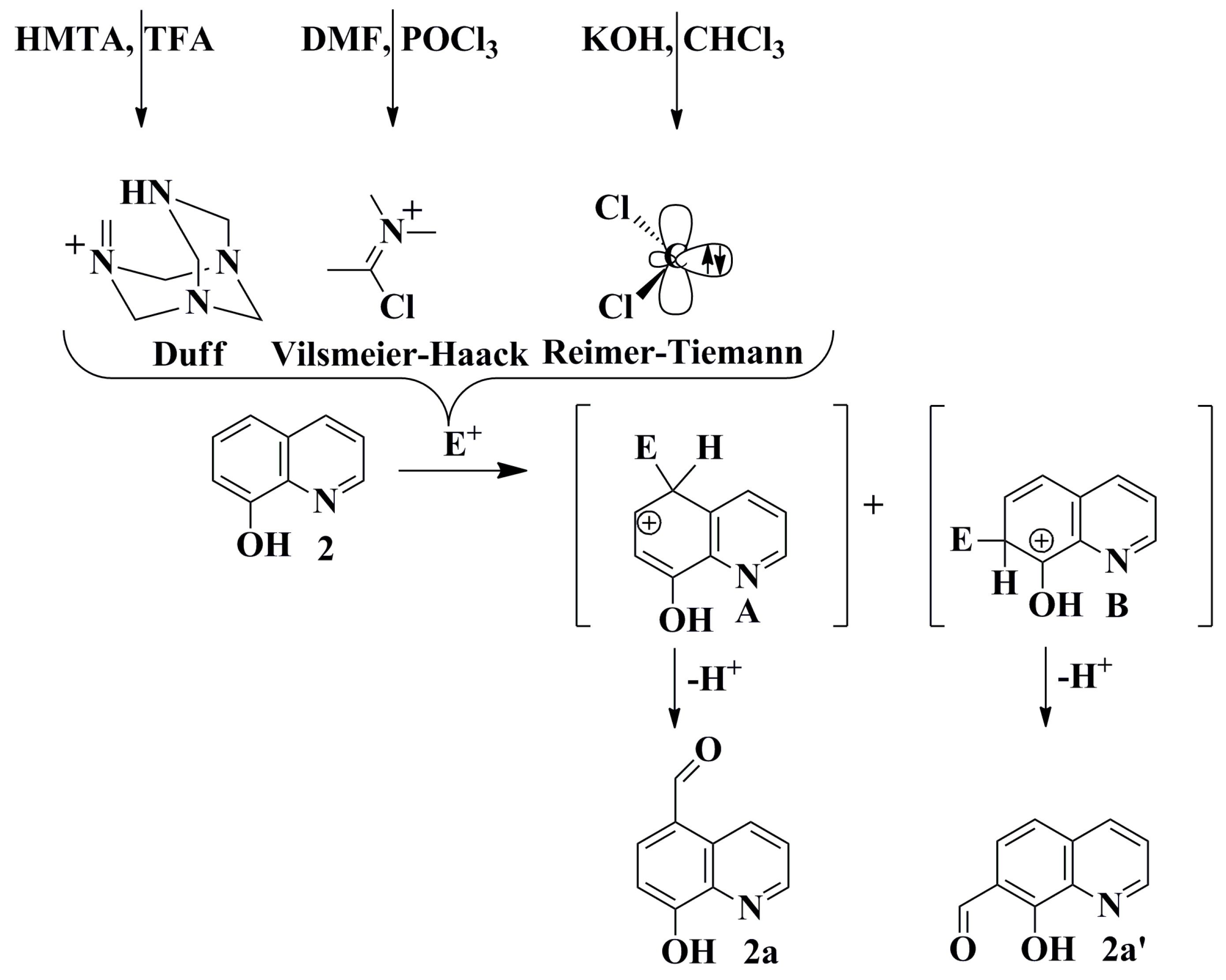
Molecules | Free Full-Text | Synthesis, Electrochemical and Spectroscopic Characterization of Selected Quinolinecarbaldehydes and Their Schiff Base Derivatives | HTML