
Development of 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methyl-morpholinium chloride cross-linked carboxymethyl cellulose films - ScienceDirect
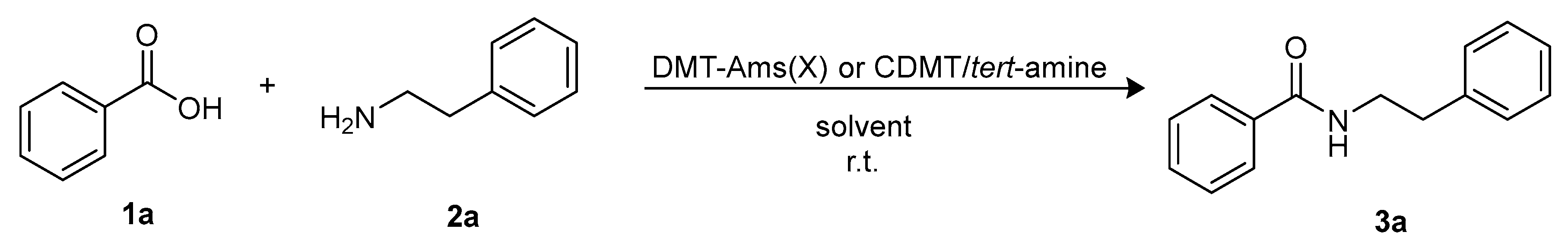
Molecules | Free Full-Text | Sustainable Triazine-Based Dehydro-Condensation Agents for Amide Synthesis | HTML
![Metabolism of a 5HT6 Antagonist, 2-Methyl-1-(Phenylsulfonyl)-4-(Piperazin-1-yl)-1H-Benzo[d]imidazole (SAM-760): Impact of Sulfonamide Metabolism on Diminution of a Ketoconazole-Mediated Clinical Drug-Drug Interaction | Drug Metabolism & Disposition Metabolism of a 5HT6 Antagonist, 2-Methyl-1-(Phenylsulfonyl)-4-(Piperazin-1-yl)-1H-Benzo[d]imidazole (SAM-760): Impact of Sulfonamide Metabolism on Diminution of a Ketoconazole-Mediated Clinical Drug-Drug Interaction | Drug Metabolism & Disposition](https://dmd.aspetjournals.org/content/dmd/46/7/934/F2.large.jpg)
Metabolism of a 5HT6 Antagonist, 2-Methyl-1-(Phenylsulfonyl)-4-(Piperazin-1-yl)-1H-Benzo[d]imidazole (SAM-760): Impact of Sulfonamide Metabolism on Diminution of a Ketoconazole-Mediated Clinical Drug-Drug Interaction | Drug Metabolism & Disposition

Chemical cross-linking/mass spectrometry targeting acidic residues in proteins and protein complexes | PNAS
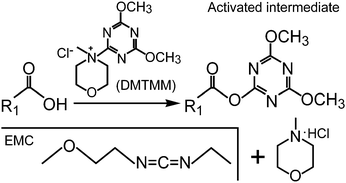
Efficient functionalization of aqueous CdSe/ZnS nanocrystals using small-molecule chemical activators - Chemical Communications (RSC Publishing) DOI:10.1039/C0CC04549G

Pharmacological targeting of a PWWP domain demonstrates cooperative control of NSD2 localization | bioRxiv
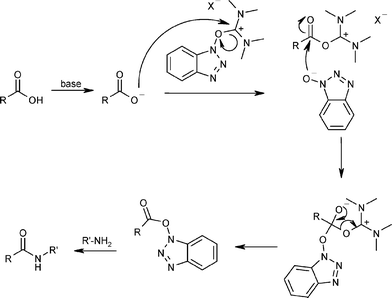
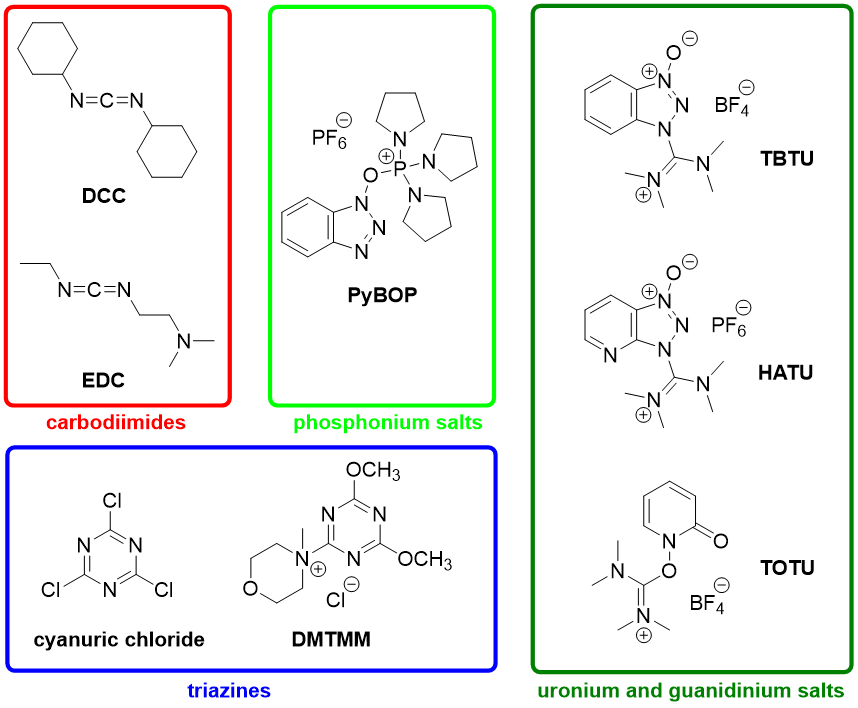
Amide bond formation: beyond the myth of coupling reagents - Chemical Society Reviews (RSC Publishing) DOI:10.1039/B701677H

Mg(OCH3)2-mediated one-pot synthesis of α-aminophosphonate derivatives of cytosine under mild conditions
![Frontiers | Membrane-Active Antibacterial Agents Based on Calix[4]arene Derivatives: Synthesis and Biological Evaluation | Chemistry Frontiers | Membrane-Active Antibacterial Agents Based on Calix[4]arene Derivatives: Synthesis and Biological Evaluation | Chemistry](https://www.frontiersin.org/files/Articles/816741/fchem-10-816741-HTML/image_m/fchem-10-816741-g008.jpg)
Frontiers | Membrane-Active Antibacterial Agents Based on Calix[4]arene Derivatives: Synthesis and Biological Evaluation | Chemistry
![Synthesis of new compounds containing the pyrazolo[3,4‐b]pyridine‐3‐one subunit - Fadel - 2009 - Journal of Heterocyclic Chemistry - Wiley Online Library Synthesis of new compounds containing the pyrazolo[3,4‐b]pyridine‐3‐one subunit - Fadel - 2009 - Journal of Heterocyclic Chemistry - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/a6208aa0-8319-4568-9f31-919659e68057/mgra001.jpg)
Synthesis of new compounds containing the pyrazolo[3,4‐b]pyridine‐3‐one subunit - Fadel - 2009 - Journal of Heterocyclic Chemistry - Wiley Online Library

An Efficient and Facile Synthesis of 5‐Amino‐3‐methyl‐7‐aryl‐1,3‐dihydroisobenzofuran‐4,6‐dicarbonitrile Derivatives Under Mild Conditions - Yin - 2017 - Journal of Heterocyclic Chemistry - Wiley Online Library
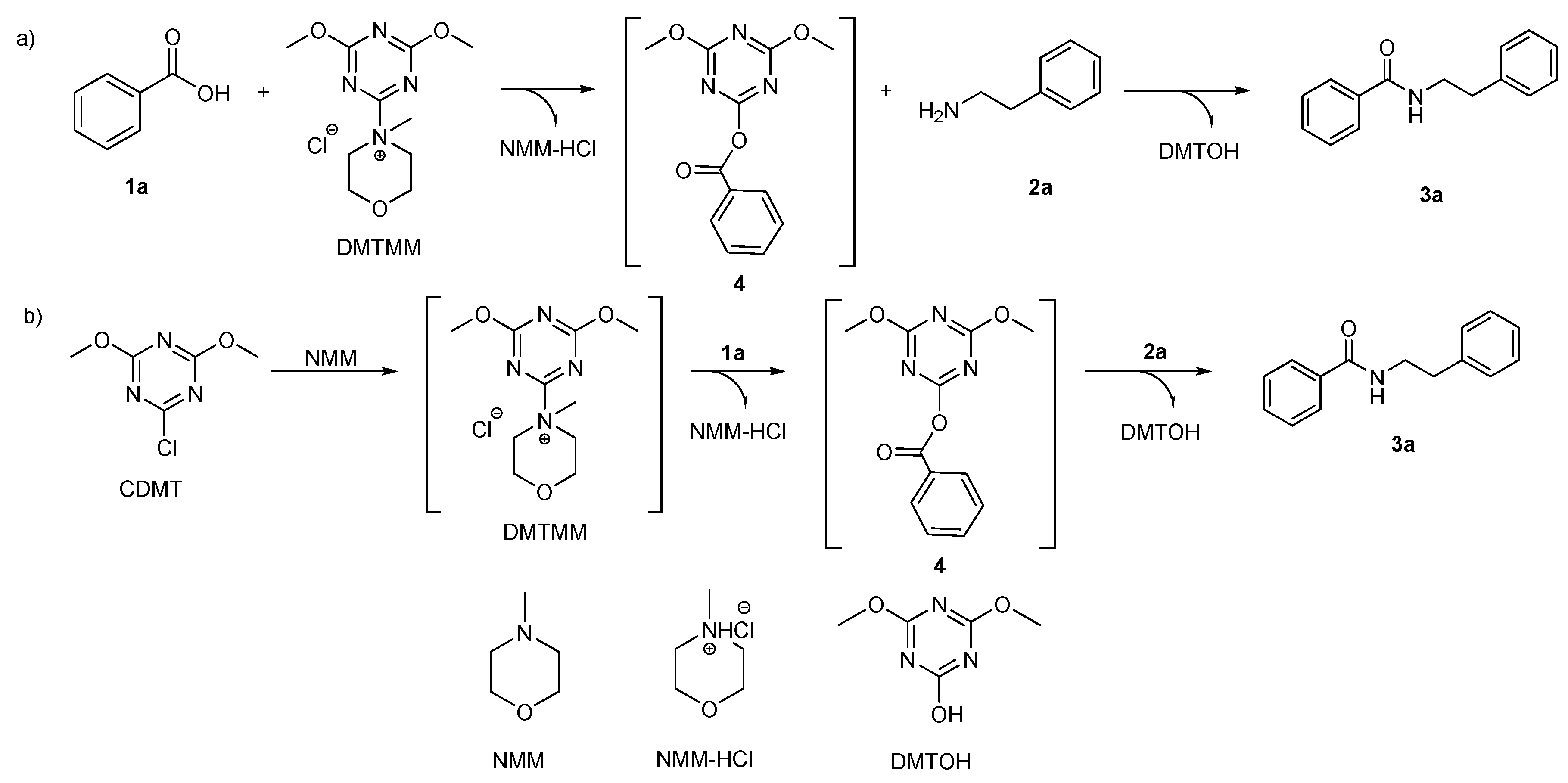
Molecules | Free Full-Text | Sustainable Triazine-Based Dehydro-Condensation Agents for Amide Synthesis | HTML







![PDF] Chemical Modifications of Hyaluronan using DMTMM-Activated Amidation | Semantic Scholar PDF] Chemical Modifications of Hyaluronan using DMTMM-Activated Amidation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e796f209ce28de0f6c0653e99a2f4ed3a40b7825/11-Figure11-1.png)

![PDF] Chemical Modifications of Hyaluronan using DMTMM-Activated Amidation | Semantic Scholar PDF] Chemical Modifications of Hyaluronan using DMTMM-Activated Amidation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e796f209ce28de0f6c0653e99a2f4ed3a40b7825/26-Figure25-1.png)




