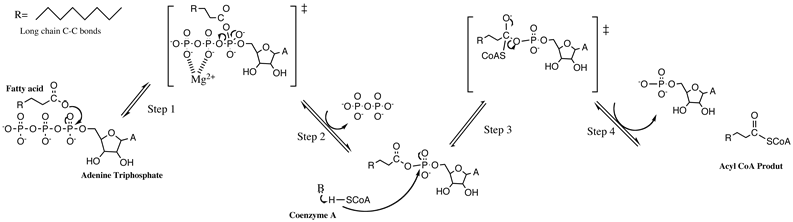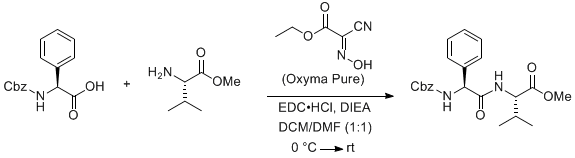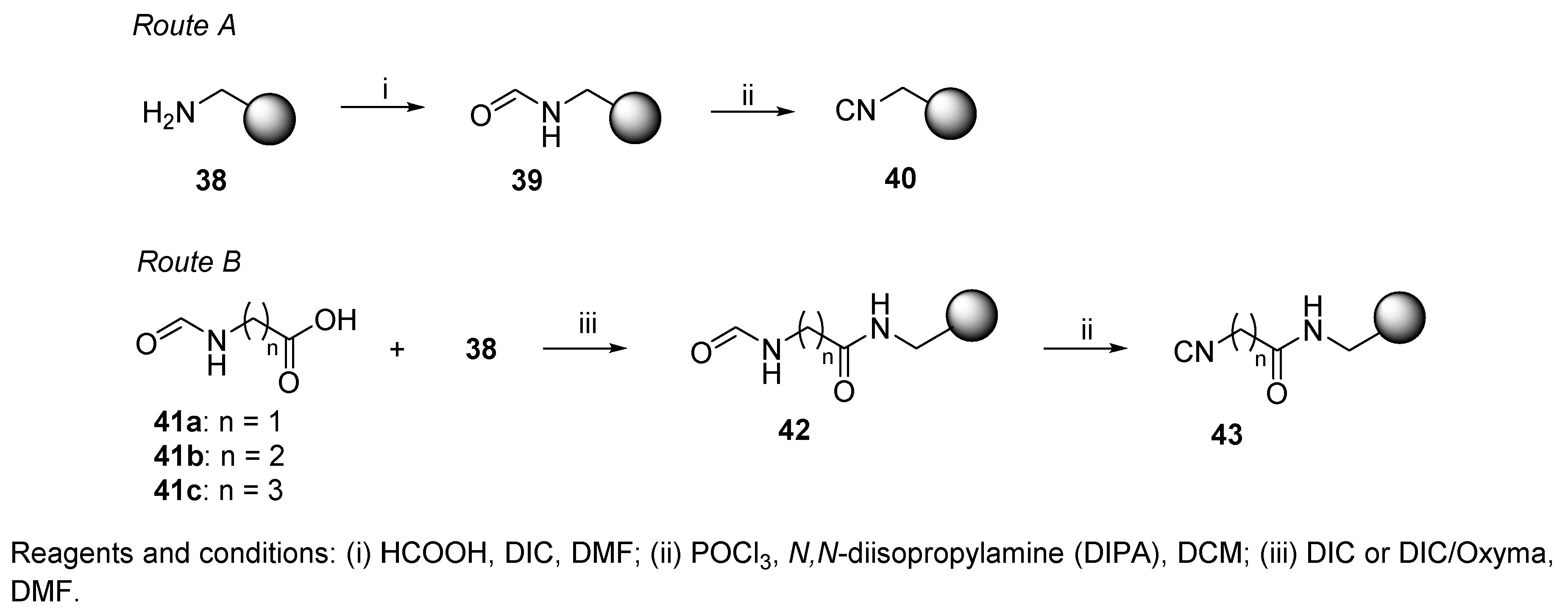
IJMS | Free Full-Text | Isocyanide Multicomponent Reactions on Solid Phase: State of the Art and Future Application | HTML
![PDF] A comparative study of amide-bond forming reagents in aqueous media – Substrate scope and reagent compatibility | Semantic Scholar PDF] A comparative study of amide-bond forming reagents in aqueous media – Substrate scope and reagent compatibility | Semantic Scholar](https://ai2-s2-public.s3.amazonaws.com/figures/2017-08-08/5f8253ff990797847606bac48011d4a849802994/4-Figure2-1.png)
PDF] A comparative study of amide-bond forming reagents in aqueous media – Substrate scope and reagent compatibility | Semantic Scholar

Generation of Hydrogen Cyanide from the Reaction of Oxyma (Ethyl Cyano(hydroxyimino)acetate) and DIC (Diisopropylcarbodiimide),Organic Process Research & Development - X-MOL
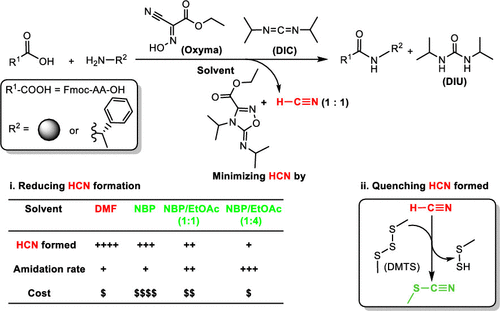
Minimizing HCN in DIC/Oxyma-Mediated Amide Bond-Forming Reactions,Organic Process Research & Development - X-MOL
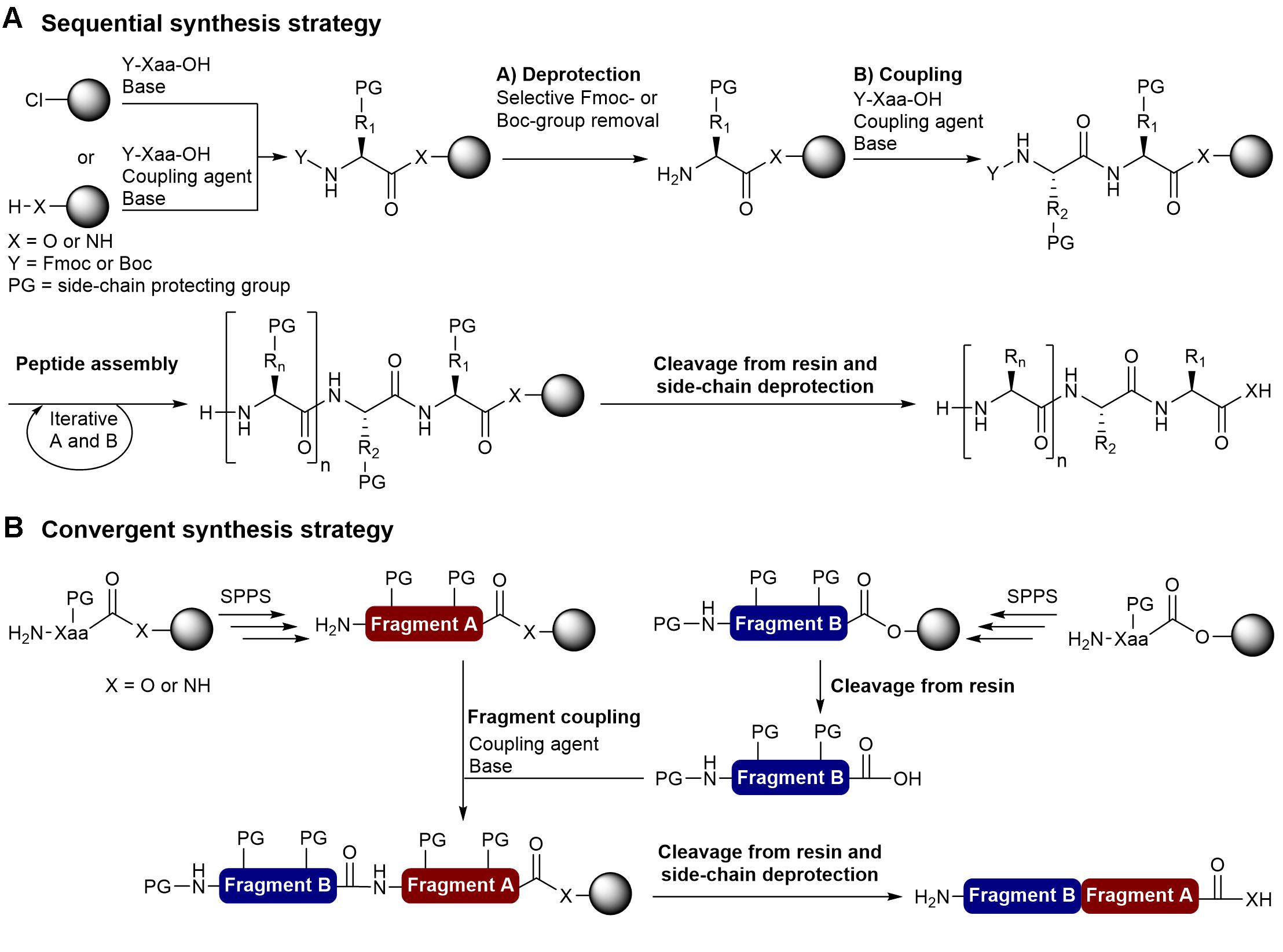
Frontiers | Recent Progress in the Chemical Synthesis of Class II and S-Glycosylated Bacteriocins | Microbiology

Ynamides as Racemization-Free Coupling Reagents for Amide and Peptide Synthesis | Journal of the American Chemical Society

Use of Oxyma as pH modulatory agent to be used in the prevention of base‐driven side reactions and its effect on 2‐chlorotrityl chloride resin - Subirós‐Funosas - 2012 - Peptide Science -
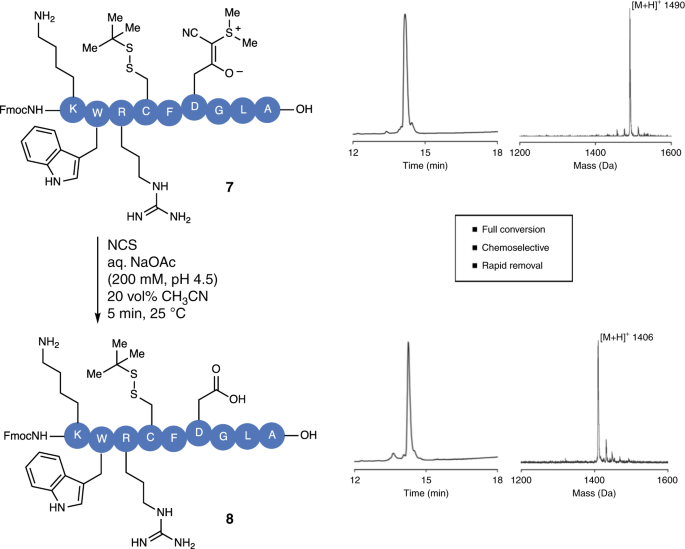
Prevention of aspartimide formation during peptide synthesis using cyanosulfurylides as carboxylic acid-protecting groups | Nature Communications

Oxyma-B, an excellent racemization suppressor for peptide synthesis - Organic & Biomolecular Chemistry (RSC Publishing)
![Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/0dbaed69-836e-41f8-8db8-526b3a6e9531/mcontent.jpg)
Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library

Generation of Hydrogen Cyanide from the Reaction of Oxyma (Ethyl Cyano(hydroxyimino)acetate) and DIC (Diisopropylcarbodiimide) | Organic Process Research & Development
![Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/f25812e6-46f3-48f3-8b7c-f247189f2dfd/msch002.jpg)