
PDF) Diethylamine Dess-Martin periodinane: An efficient catalyst-oxidant combination in a sequential, one-pot synthesis of difficult to access 2-amino-3,5-dicarbonitrile-6-sulfanylpyridines at ambient temperature
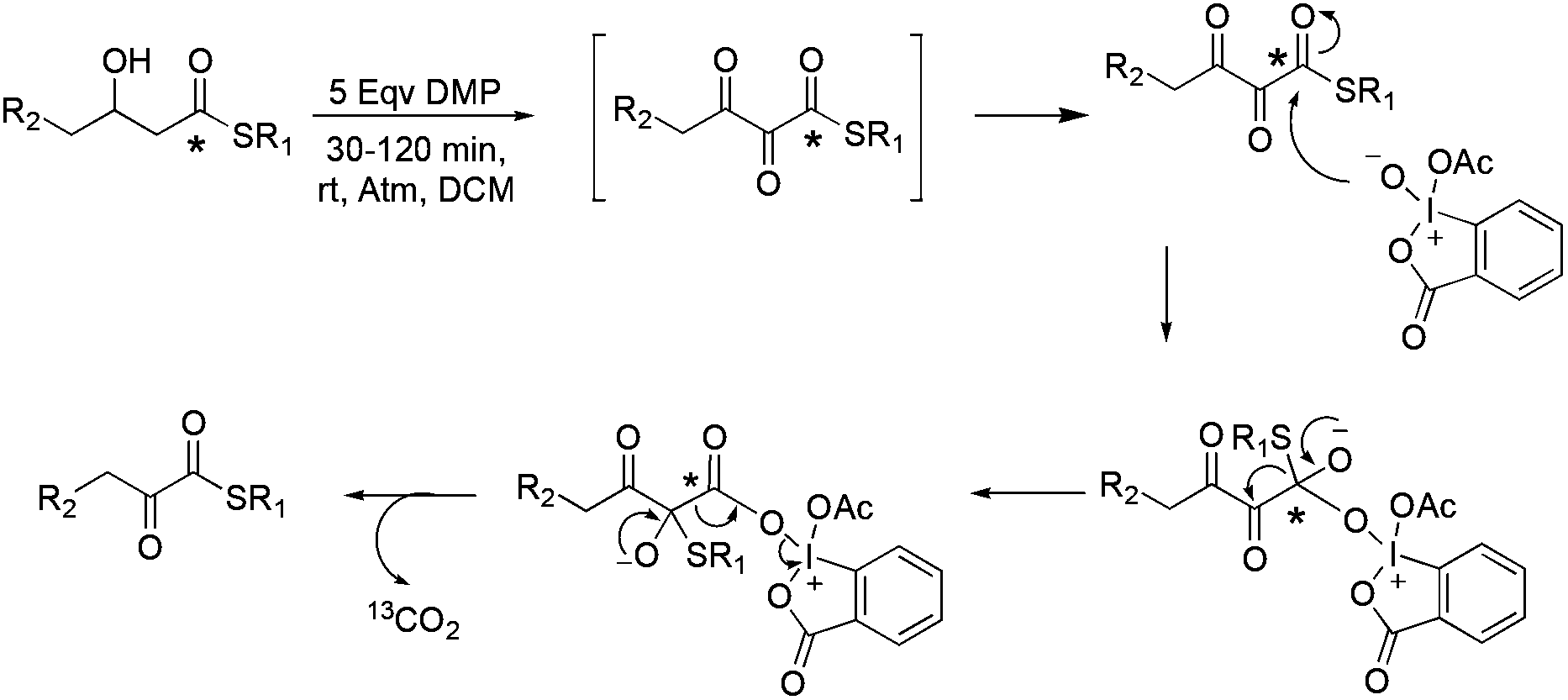
Dess–Martin periodinane oxidative rearrangement for preparation of α-keto thioesters - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB02959D

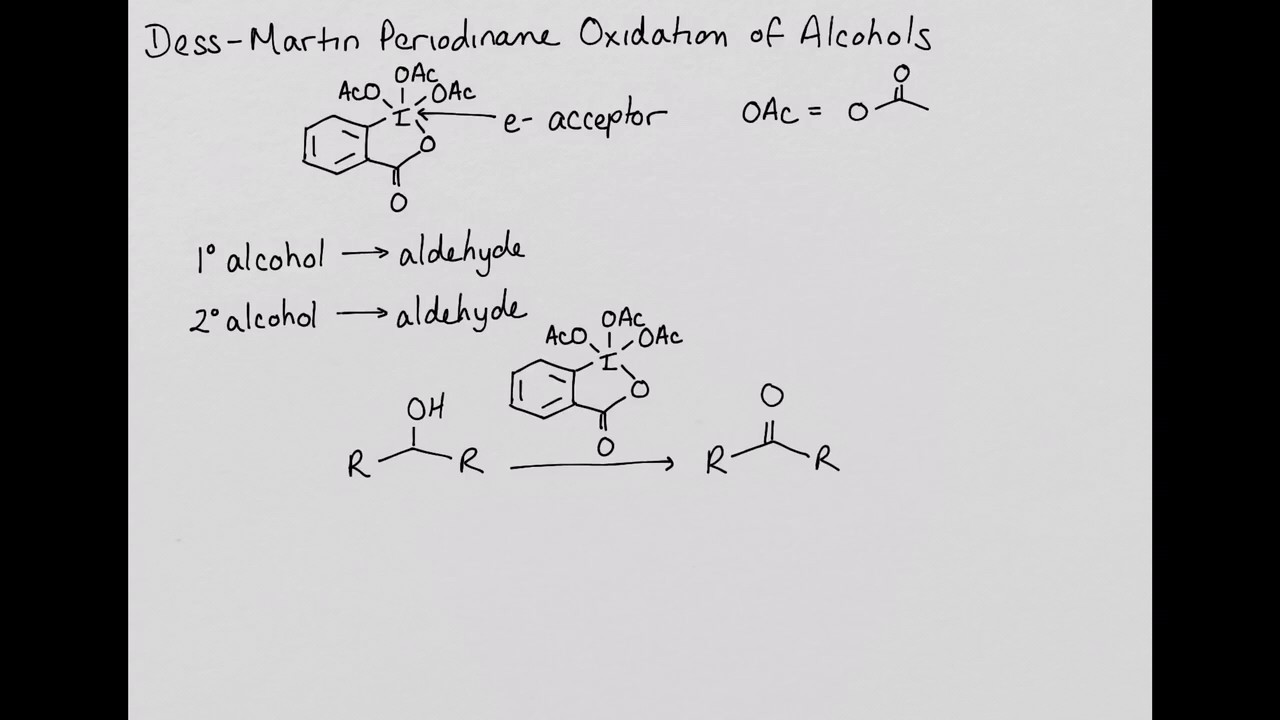
Aldehydes from oxidation of primary alcohols using the Dess- Martin periodinane reagent 14.2 Preparing Aldehydes and Ketones. - ppt download

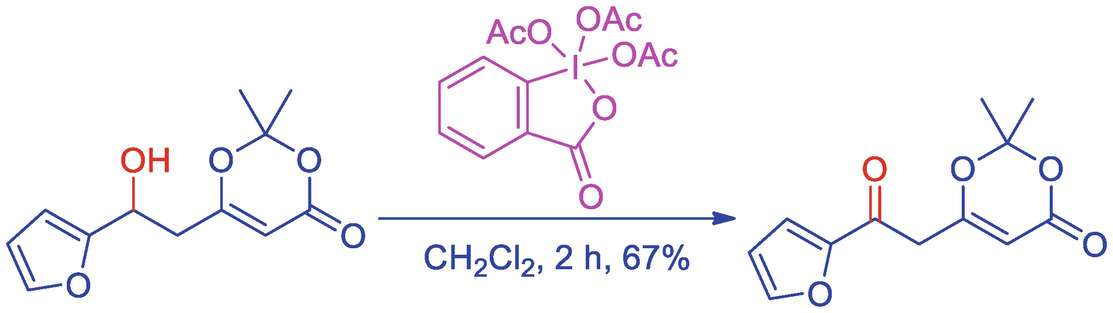
Table 2 from Iodine(V) reagents in organic synthesis. Part 1. Synthesis of polycyclic heterocycles via Dess-Martin periodinane-mediated cascade cyclization: generality, scope, and mechanism of the reaction. | Semantic Scholar




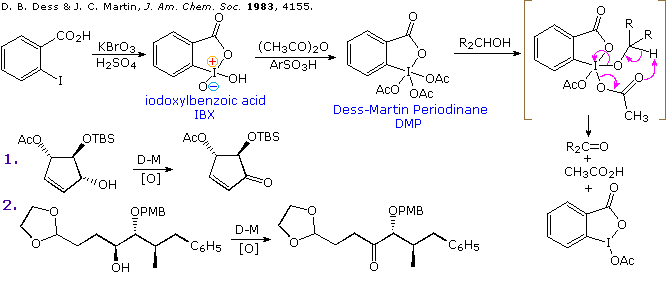








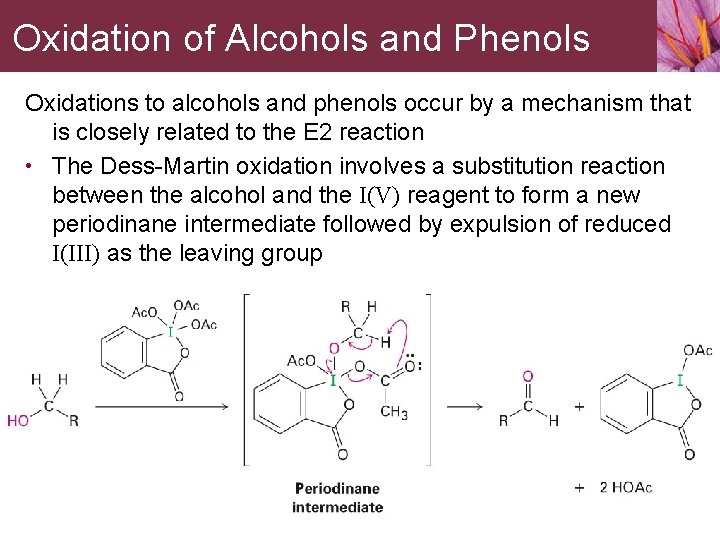
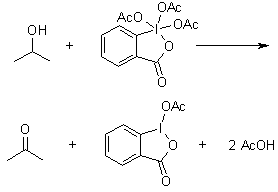
![16] Dess-Martin Oxidation 1983 | Organic chemistry reactions, Organic chemistry, Teaching chemistry 16] Dess-Martin Oxidation 1983 | Organic chemistry reactions, Organic chemistry, Teaching chemistry](https://i.pinimg.com/474x/ef/52/f4/ef52f4b8736085645975a3b6bb57d040.jpg)
![16] Dess-Martin Oxidation 1983 – ChemInfoGraphic 16] Dess-Martin Oxidation 1983 – ChemInfoGraphic](https://cheminfographic.files.wordpress.com/2017/08/16_dess_martin_oxidation.jpg?w=717)
