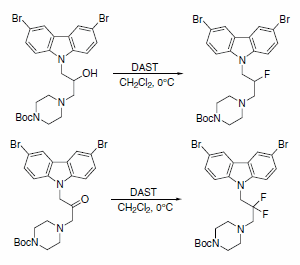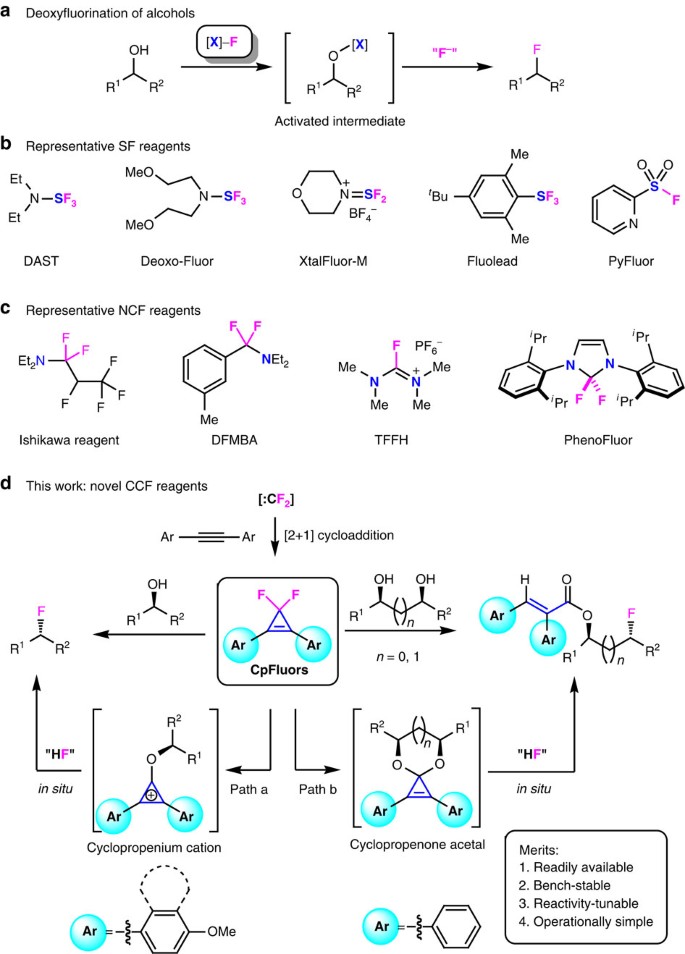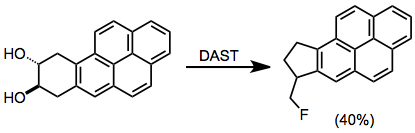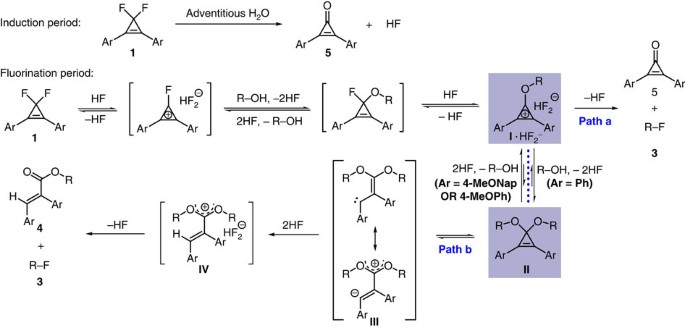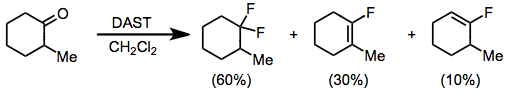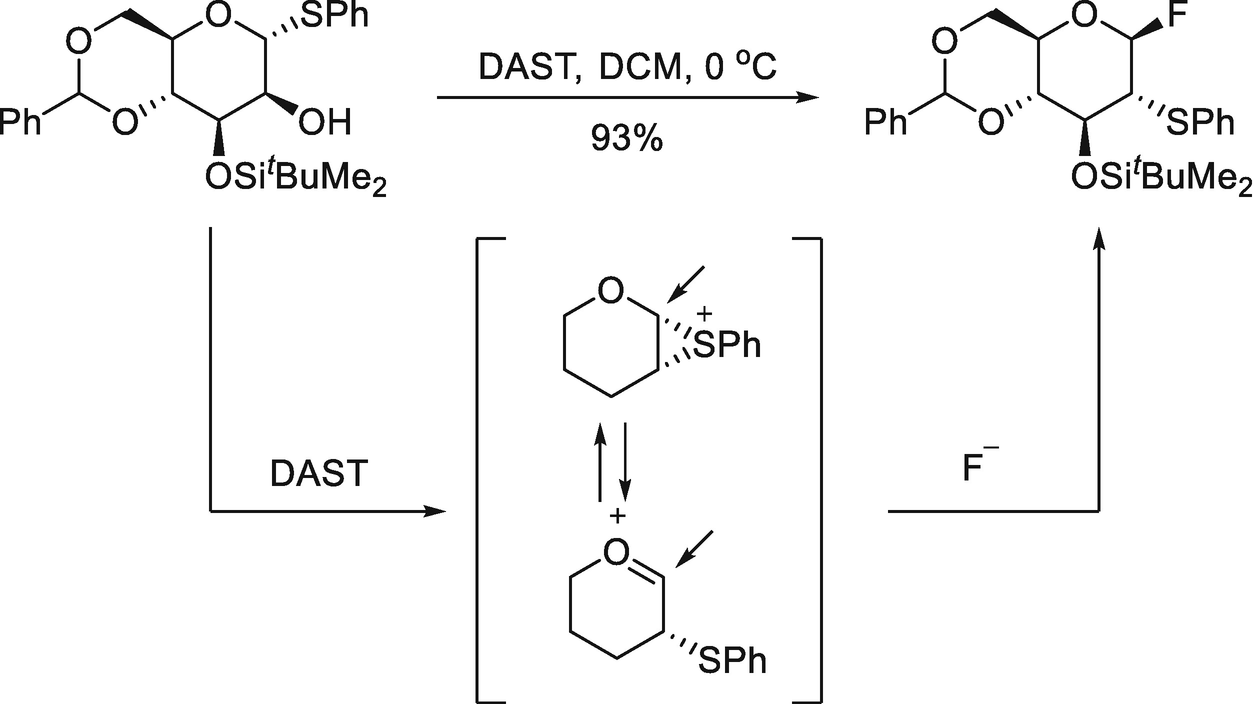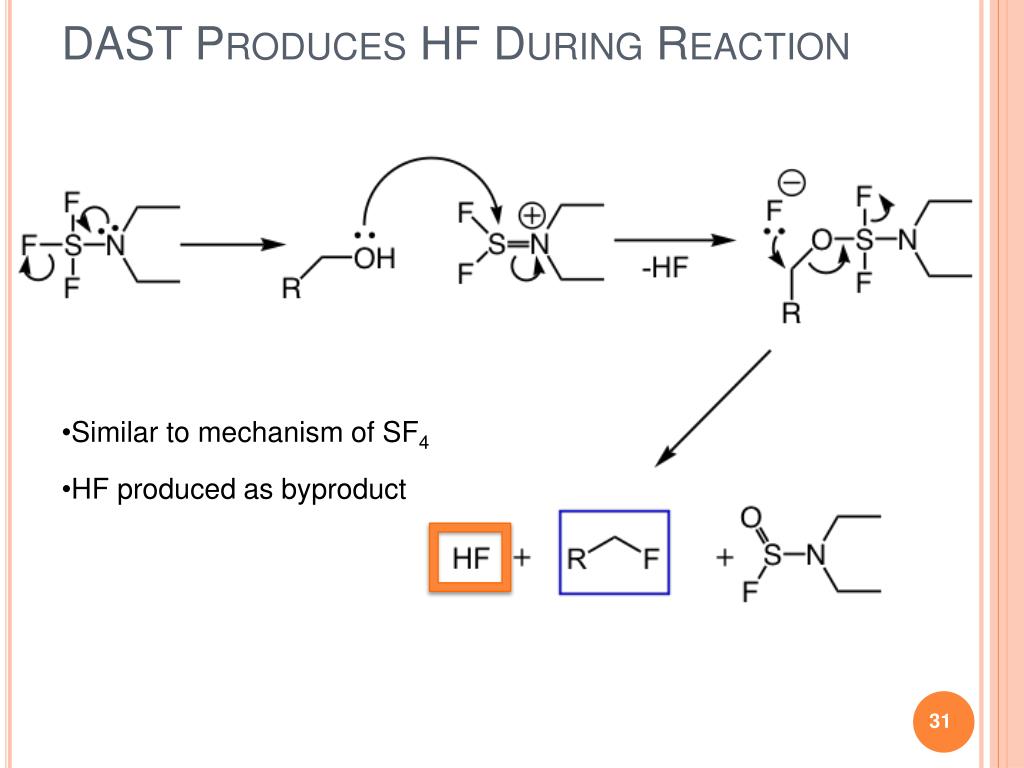
PPT - Design and Development of Safe and Selective Deoxofluorinating Reagents PowerPoint Presentation - ID:6865556
![PDF] RECENT DEVELOPMENTS IN FLUORINATION CHEMISTRY OF DAST WITH SPECIAL REFERENCE TO ALCOHOLS | Semantic Scholar PDF] RECENT DEVELOPMENTS IN FLUORINATION CHEMISTRY OF DAST WITH SPECIAL REFERENCE TO ALCOHOLS | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b4b752df74d43e44d3deebc422f07ece42518525/5-Figure5-1.png)
PDF] RECENT DEVELOPMENTS IN FLUORINATION CHEMISTRY OF DAST WITH SPECIAL REFERENCE TO ALCOHOLS | Semantic Scholar

Synthesis of Cycloalkanoindoles by an Unusual DAST‐Triggered Rearrangement Reaction - Giannini - 2004 - European Journal of Organic Chemistry - Wiley Online Library

Rearrangement reactions in the fluorination of d-glucopyranoside at the C-4 position by DAST - ScienceDirect

Emerging fluorination methods in organic chemistry relevant for life science application - ScienceDirect

Are carboxylic esters really refractory to DAST? On the fluorination of α-hydroxyesters with DAST - ScienceDirect

Synthesis of Acyl Fluorides via DAST-Mediated Fluorinative C–C Bond Cleavage of Activated Ketones | Organic Letters

A modified fluoro-Pummerer reaction with DAST and NIS for synthesis of β-amino-α-fluoro-sulfides from corresponding β-amino-sulfides - ScienceDirect

Successive C-C bond cleavage, fluorination, trifluoromethylthio- and pentafluorophenylthiolation under metal-free conditions to provide compounds with dual fluoro-functionalization. - Abstract - Europe PMC