Intramolecular donor–acceptor cyclopropane ring-opening cyclizations - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C3CS60238A

Nucleophilic ring opening of cyclopropane hemimalonates using internal Brønsted acid activation. | Semantic Scholar

Ring-opening hydroarylation of monosubstituted cyclopropanes enabled by hexafluoroisopropanol. - Abstract - Europe PMC

Stress relief: Exercising Lewis acid catalysis for donor-acceptor cyclopropane ring-opening annulations, a basis for new reaction methodologies | Semantic Scholar

Recent Advances in the Chemistry of Doubly Activated Cyclopropanes: Synthesis and Reactivity | Bentham Science
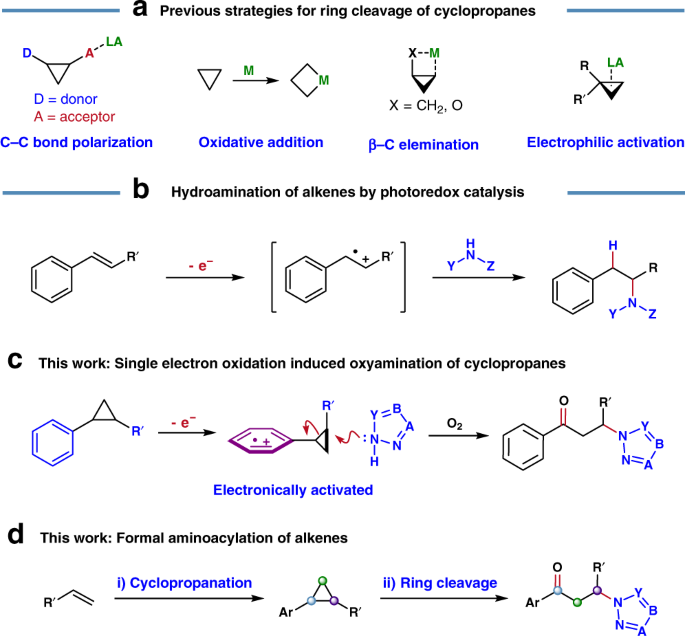
A unique Pd-catalysed Heck arylation as a remote trigger for cyclopropane selective ring-opening | Nature Communications
![Metal‐Free Ring Opening Cyclization of Cyclopropane Carbaldehydes and N‐Benzyl Anilines: An Eco‐Friendly Access to Functionalized Benzo[b]azepine Derivatives - Dey - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library Metal‐Free Ring Opening Cyclization of Cyclopropane Carbaldehydes and N‐Benzyl Anilines: An Eco‐Friendly Access to Functionalized Benzo[b]azepine Derivatives - Dey - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/7227d3d3-a7db-42d5-983b-efcf73109026/adsc201801714-fig-5007-m.jpg)
Metal‐Free Ring Opening Cyclization of Cyclopropane Carbaldehydes and N‐Benzyl Anilines: An Eco‐Friendly Access to Functionalized Benzo[b]azepine Derivatives - Dey - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library

IJMS | Free Full-Text | Computational Design of Radical Recognition Assay with the Possible Application of Cyclopropyl Vinyl Sulfides as Tunable Sensors | HTML
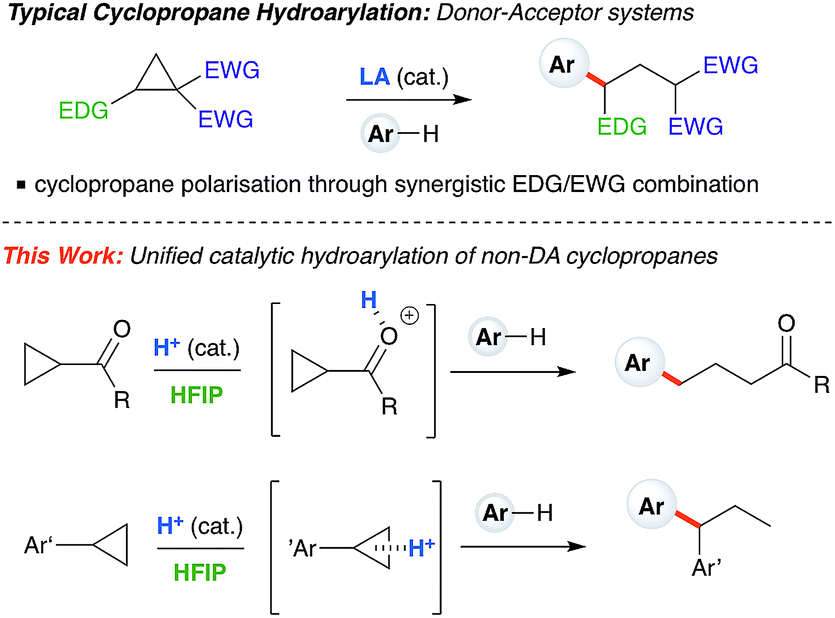
Ring-opening hydroarylation of monosubstituted cyclopropanes enabled by hexafluoroisopropanol - Chemical Science (RSC Publishing) DOI:10.1039/C8SC02126K
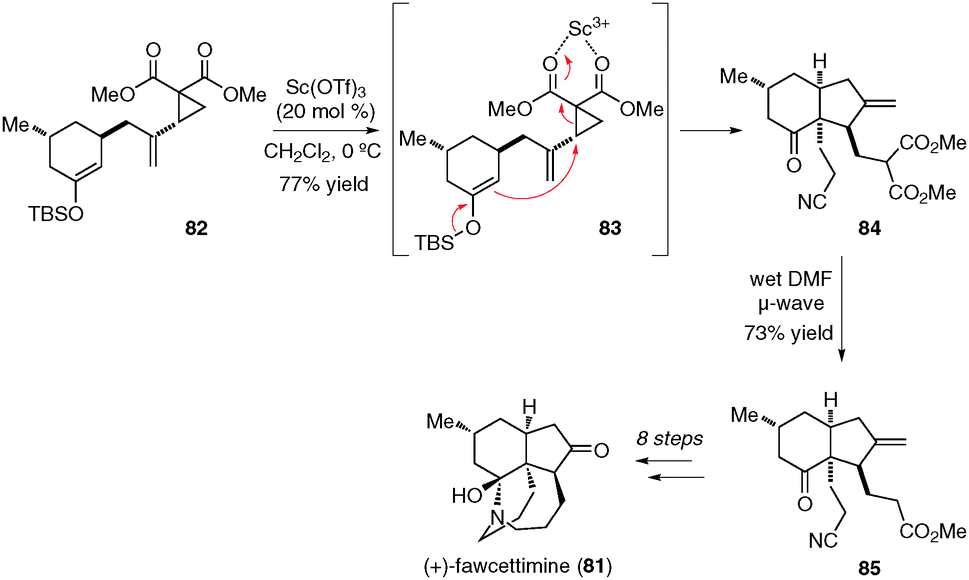
Intramolecular donor–acceptor cyclopropane ring-opening cyclizations - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C3CS60238A

Ring-opening hydroarylation of monosubstituted cyclopropanes enabled by hexafluoroisopropanol. - Abstract - Europe PMC

Asymmetric Catalytic Reactions of Donor–Acceptor Cyclopropanes - Xia - 2021 - Angewandte Chemie - Wiley Online Library
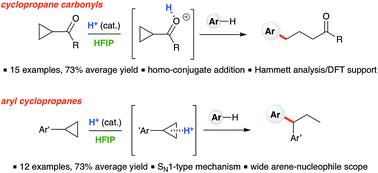
Ring-opening hydroarylation of monosubstituted cyclopropanes enabled by hexafluoroisopropanol - Chemical Science (RSC Publishing)
organic chemistry - Mechanism of acid-catalyzed ring opening of a cyclopropane ring - Chemistry Stack Exchange
![Palladium-catalyzed ring-opening of cyclopropyl benzamides: synthesis of benzo[c]azepine-1-ones via C(sp3)–H functionalization - ScienceDirect Palladium-catalyzed ring-opening of cyclopropyl benzamides: synthesis of benzo[c]azepine-1-ones via C(sp3)–H functionalization - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040402013003062-sc1.jpg)