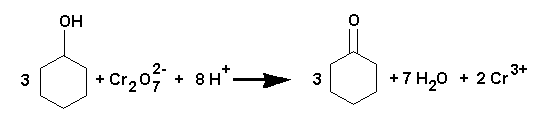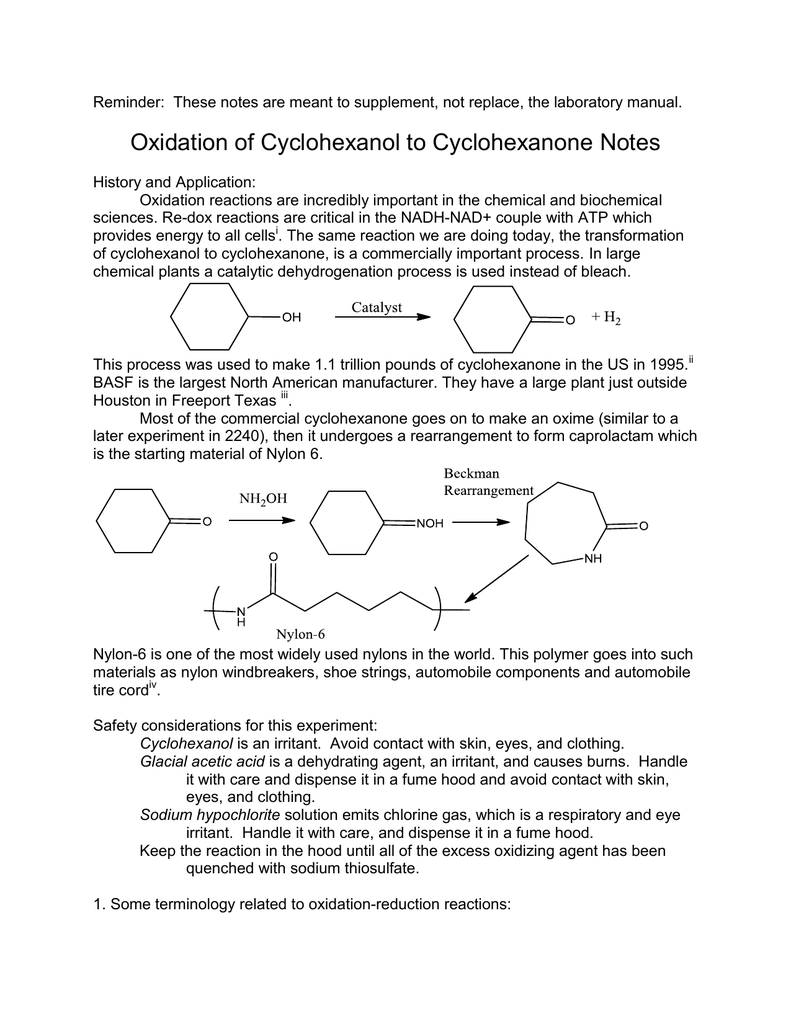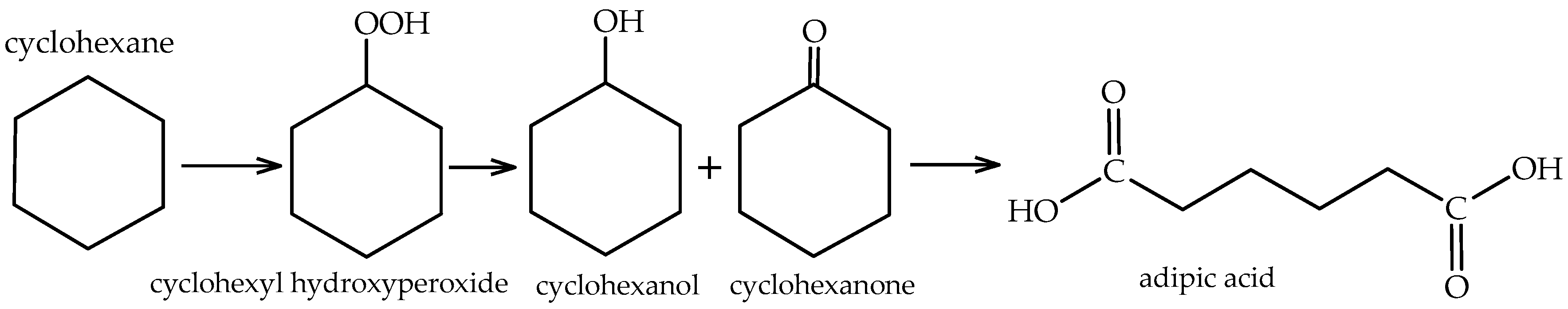
Catalysts | Free Full-Text | Sustainability in Catalytic Cyclohexane Oxidation: The Contribution of Porous Support Materials | HTML
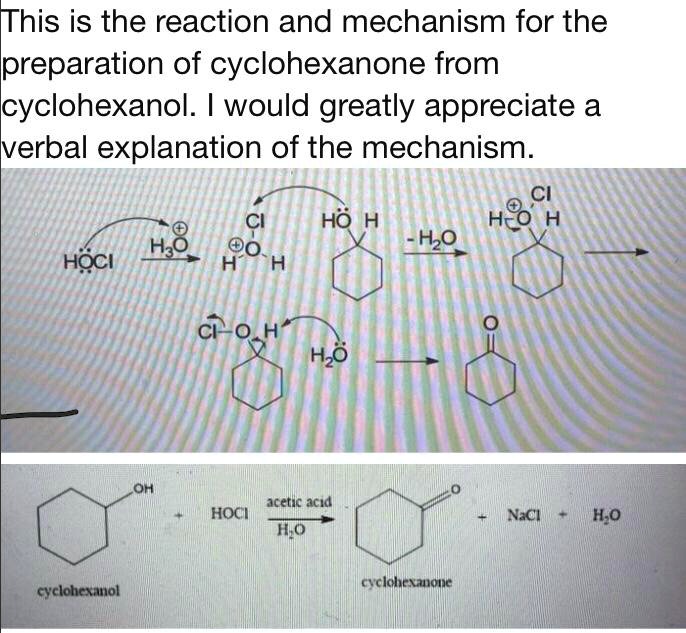
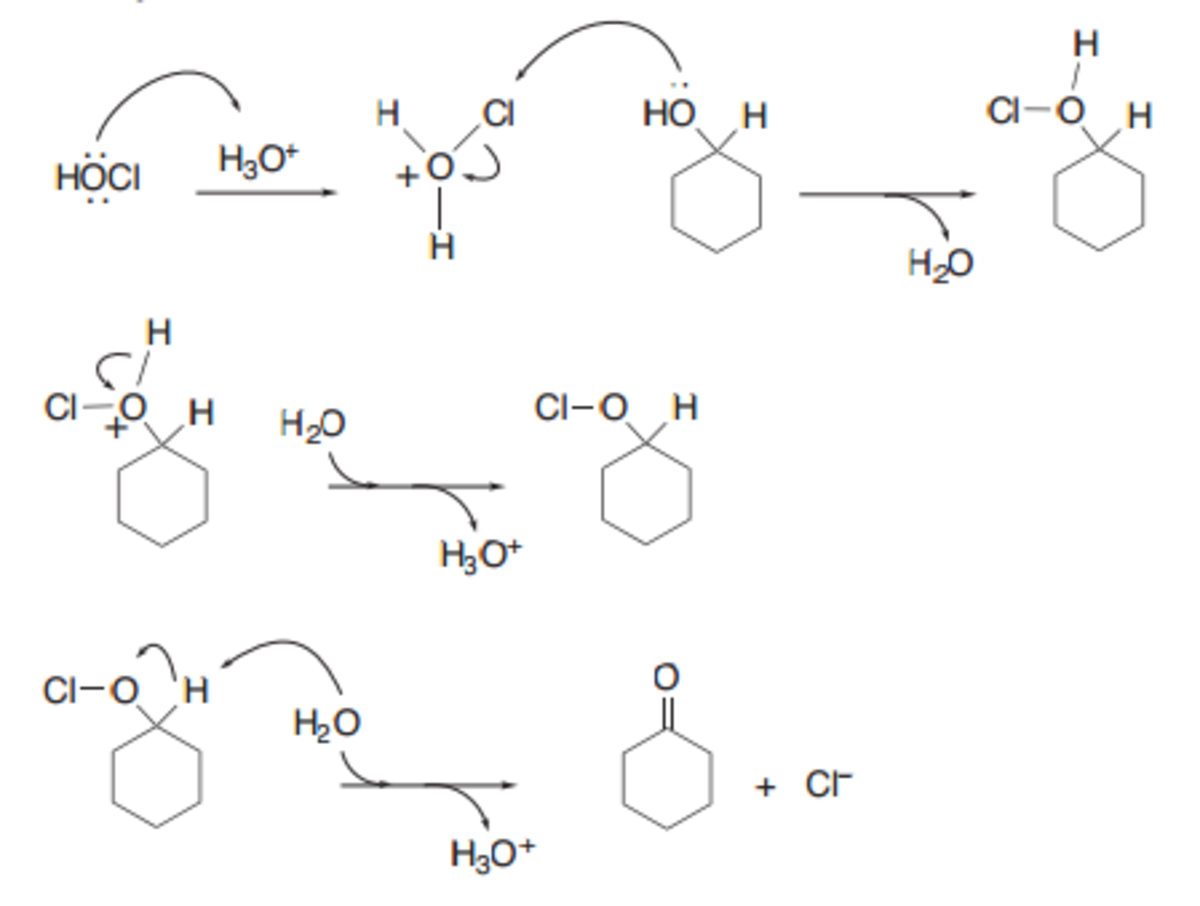
SOLVED:TThis is the reaction and mechanism for the preparation of cyclohexanone from cyclohexanol. would greatly appreciate a verbal explanation of the mechanism: CI Cl HO H H@ H Hzo @0 HzO HoCI

Highly selective hydrothermal production of cyclohexanol from biomass-derived cyclohexanone over Cu powder - ScienceDirect
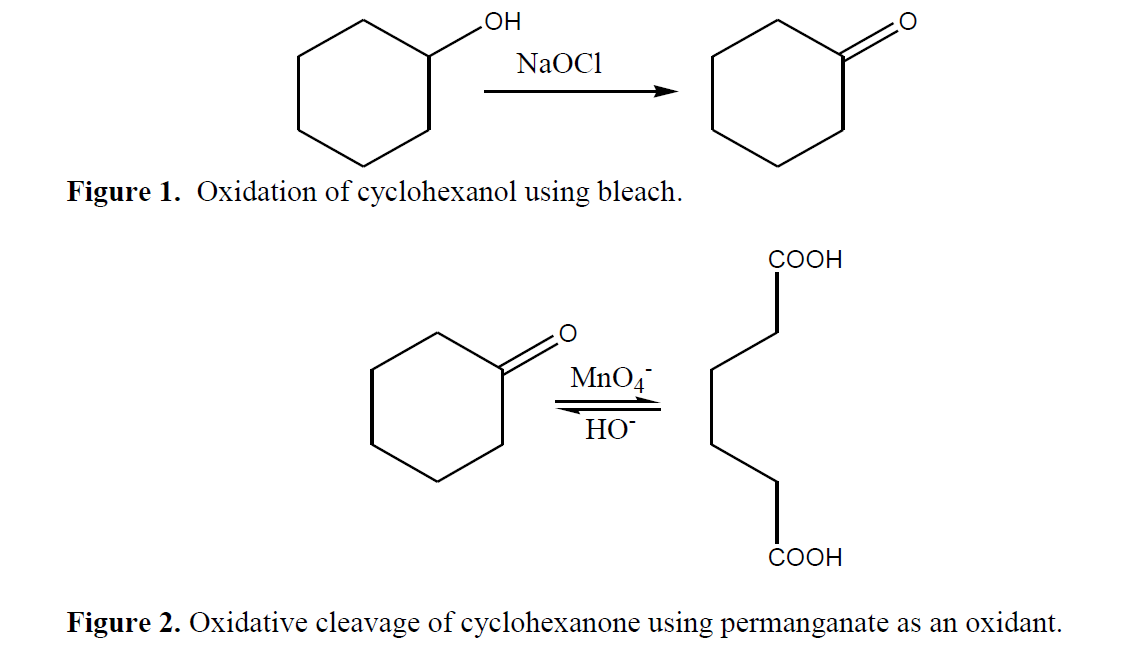
Solved] In the hypochlorite oxidation of cyclohexanol to cyclohexanone, what purpose does the acetic acid serve? | Course Hero

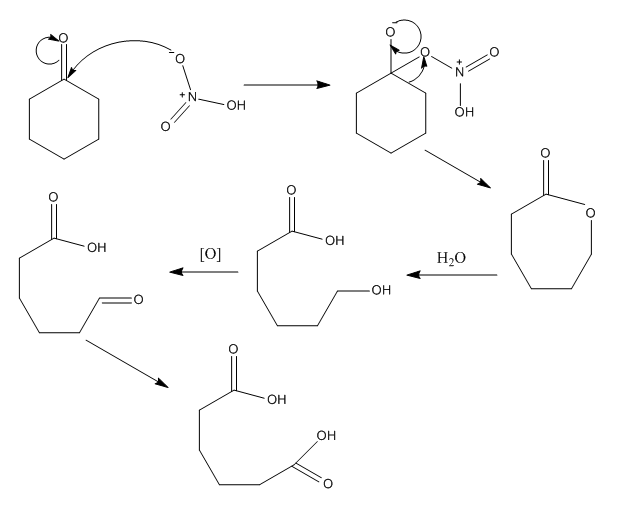
Using cyclohexanone as the starting material, show how each of the following compounds could be synthesized. Give all the reagents needed. No mechanism needed. | Study.com
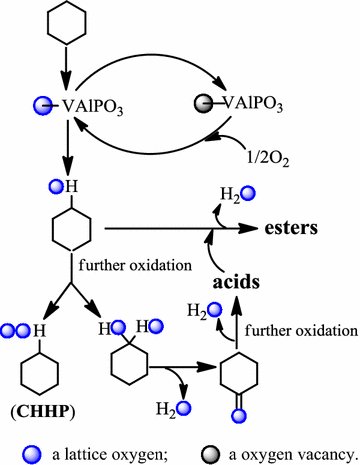
The highly selective oxidation of cyclohexane to cyclohexanone and cyclohexanol over VAlPO4 berlinite by oxygen under atmospheric pressure | BMC Chemistry | Full Text
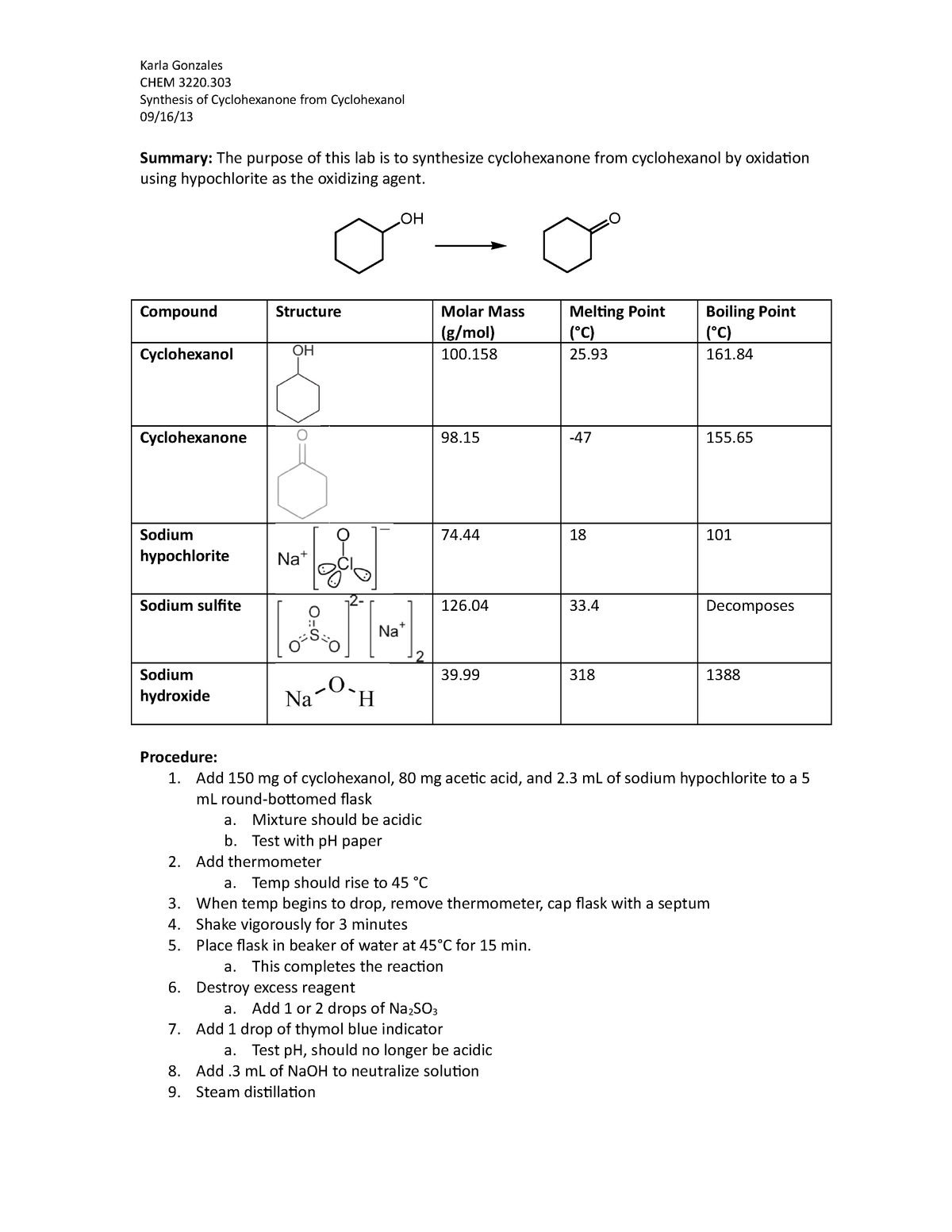
Cyclohexanol from Cyclohexanone Lab Report - Karla Gonzales CHEM 3220 Synthesis of Cyclohexanone - StuDocu

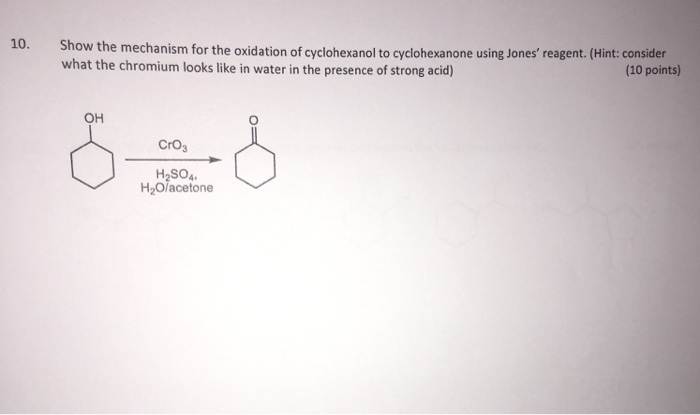
Cyclohexanol to cyclohexanone by the oxidizing agent sodium dichromate - Homework Help - Science Forums