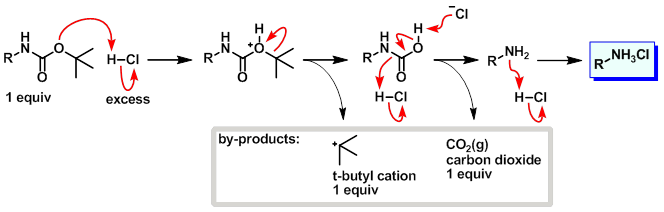
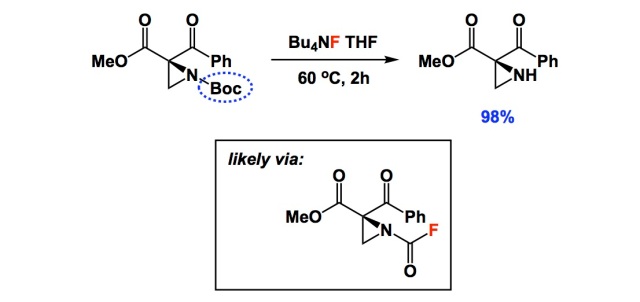
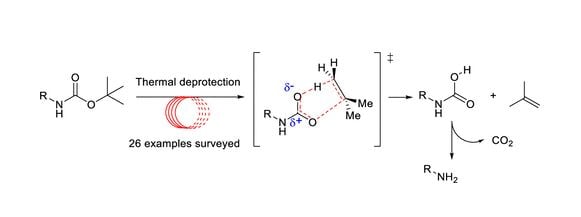
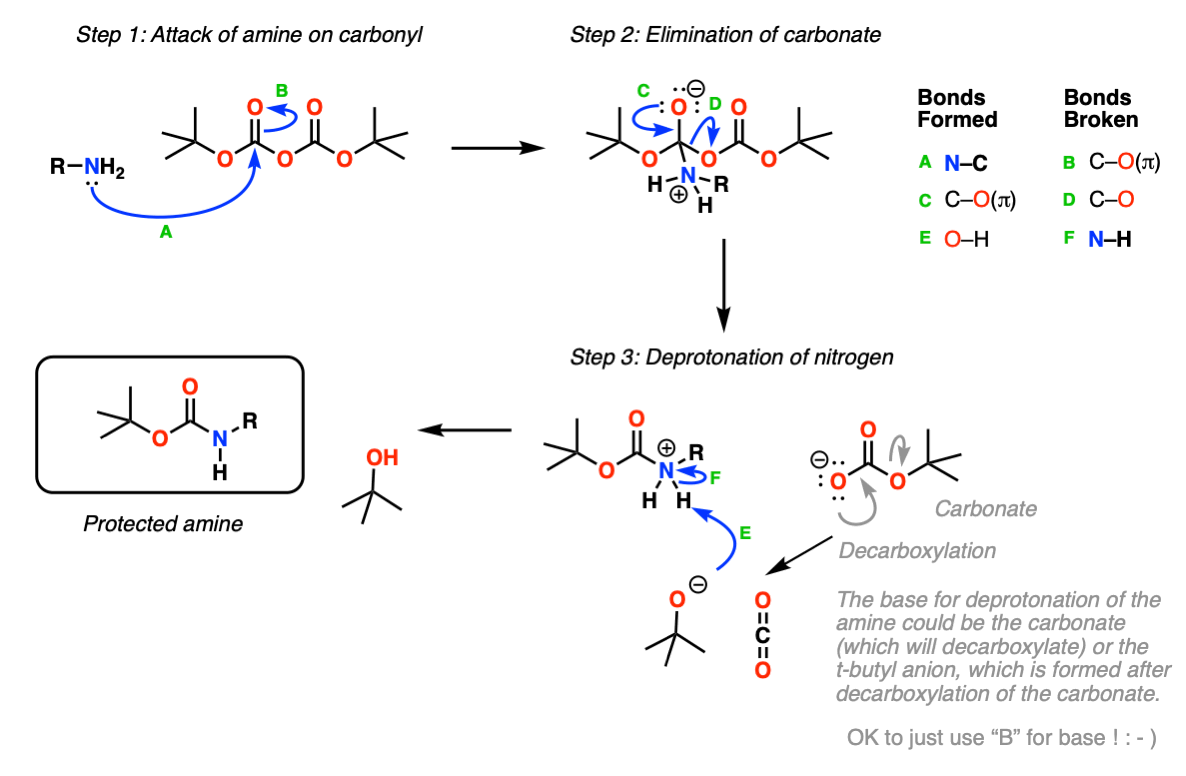
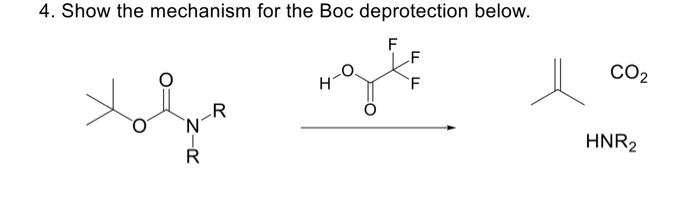
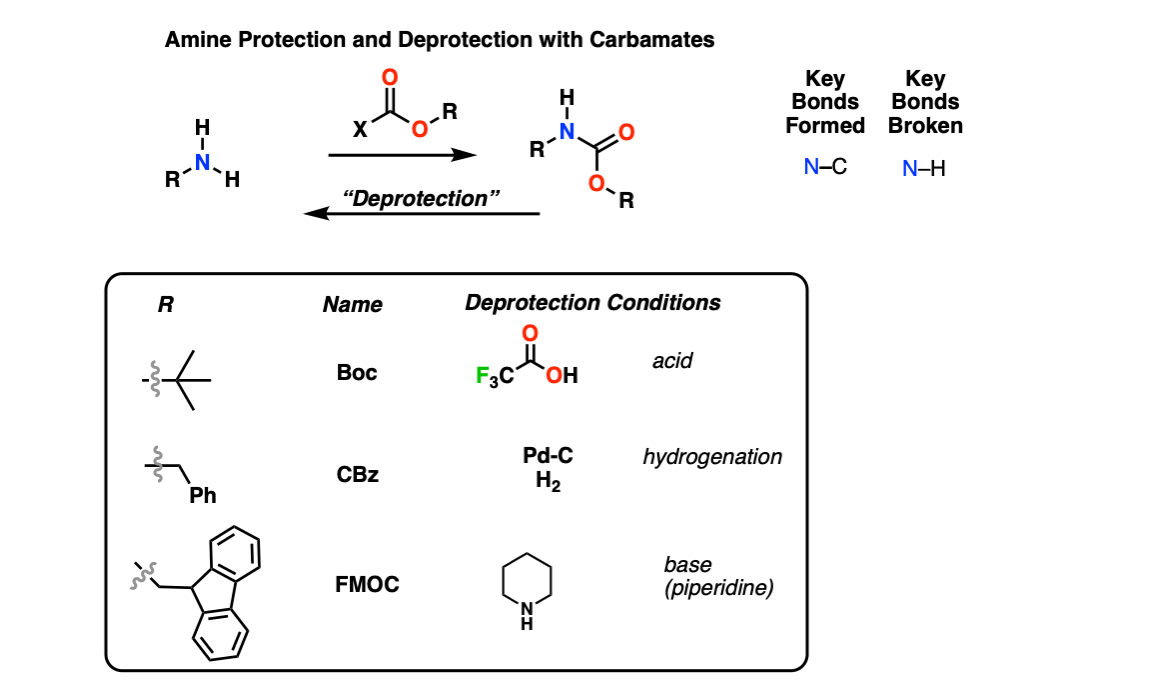
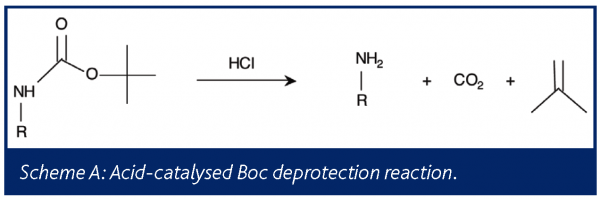
Deprotection of N‐tert‐Butoxycarbonyl (Boc) Protected Functionalized Heteroarenes via Addition–Elimination with 3‐Methoxypropylamine - Gulledge - 2020 - European Journal of Organic Chemistry - Wiley Online Library
organic chemistry - Deprotection of Boc using TAF to obtained free amine group - Chemistry Stack Exchange
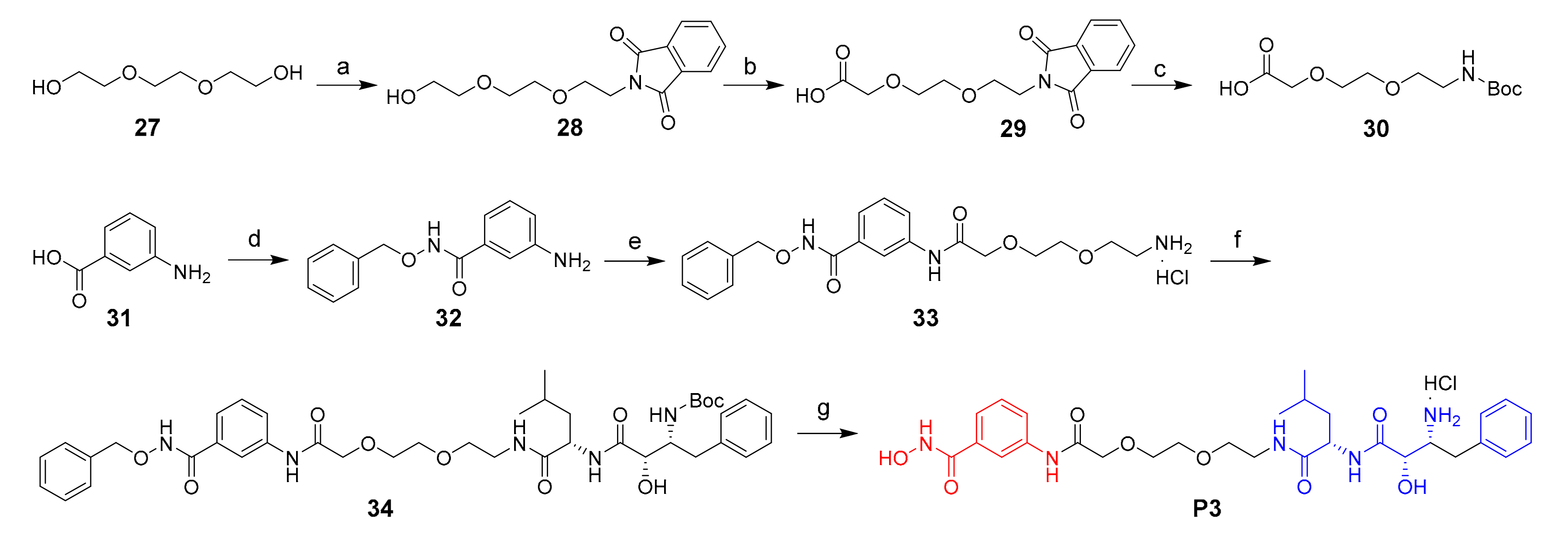
Molecules | Free Full-Text | Development of a Bestatin-SAHA Hybrid with Dual Inhibitory Activity against APN and HDAC | HTML
ChemSpider SyntheticPages | BOC deprotection of an aminophenylethyl methanesulfonate using hydrochloric acid

Mild deprotection of the <i>N</i>-<i>tert</i>-butyloxycarbonyl (<i>N</i>-Boc) group using oxalyl chloride. - Abstract - Europe PMC