
Spin-Center Shift-Enabled Direct Enantioselective α-Benzylation of Aldehydes with Alcohols | Journal of the American Chemical Society
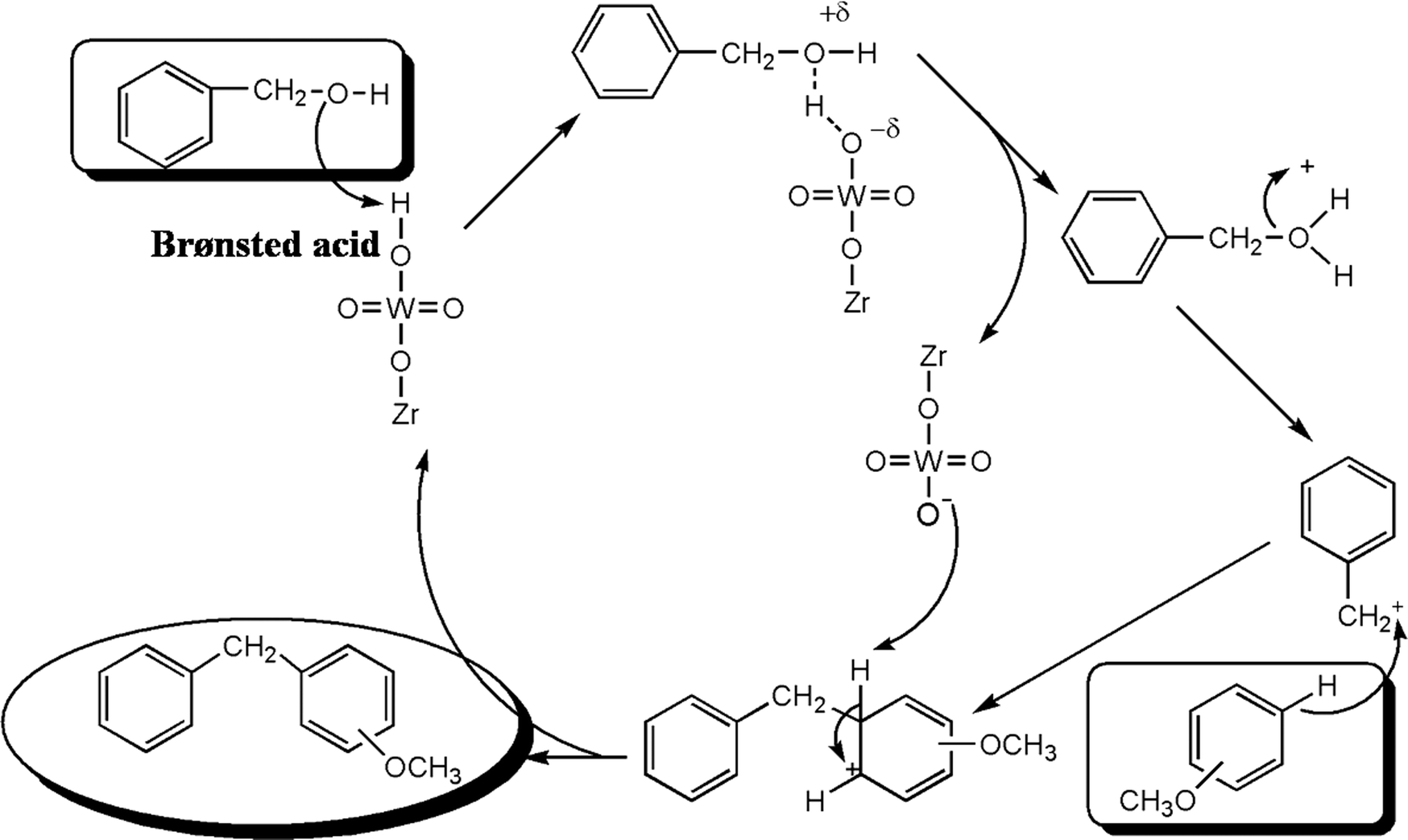
One-pot synthesis of mesoporous ZrPW solid acid catalyst for liquid phase benzylation of anisole - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C3CY00918A

2 Proposed mechanism of the decarboxylative benzylation. Reproduced... | Download Scientific Diagram

Atom economical benzylation of phenol with benzyl alcohol using 20 % (w/w)Cs2.5H0.5PW12O40 supported on mesocellular foam silica (MCF) and its kinetics - ScienceDirect

Substrate scope for the photoenolization/b-benzylation sequence.a )The... | Download Scientific Diagram

Benzylation Reactions in DMF Lead to an Impurity Which Acts as an Organocatalyst Poison in Thiourea-Catalyzed Glycosylations | The Journal of Organic Chemistry

Rhodium(III)‐Catalyzed C−H Benzylation of Indole's C3 Position with Aza‐o‐Quinone Methides - Liu - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

Benzylation of acetic acid to benzyl acetate over highly active and reusable Micro/Meso-HZSM-5 - ScienceDirect

N-Benzylation of primary amines using magnetic Fe3O4 nanoparticles functionalized with hexamethylenetetramine as an efficient and recyclable heterogeneous catalyst - ScienceDirect

Friedel–Crafts Reaction of Benzyl Fluorides: Selective Activation of CF Bonds as Enabled by Hydrogen Bonding - Champagne - 2014 - Angewandte Chemie - Wiley Online Library

Site-Selective C–H Benzylation of Alkanes with N-Triftosylhydrazones Leading to Alkyl Aromatics - ScienceDirect
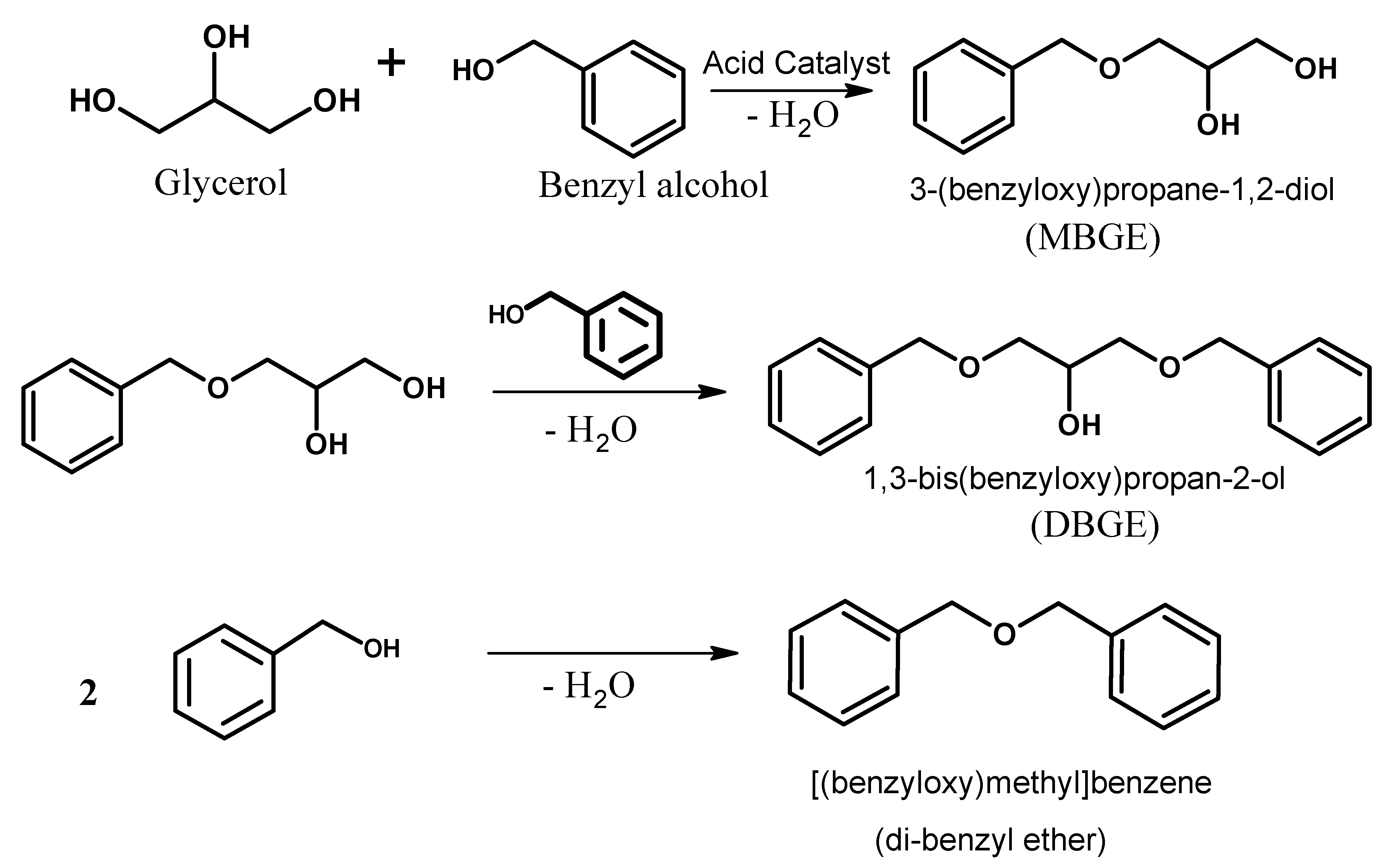
Catalysts | Free Full-Text | Solvent-Free Benzylation of Glycerol by Benzyl Alcohol Using Heteropoly Acid Impregnated on K-10 Clay as Catalyst | HTML

Metal Free Benzylation and Alkylation of Quinoxalin‐2(1H)‐ones with Alkenes Triggered by Sulfonyl Radical Generated from Sulfinic Acids - Sekhar Dutta - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library
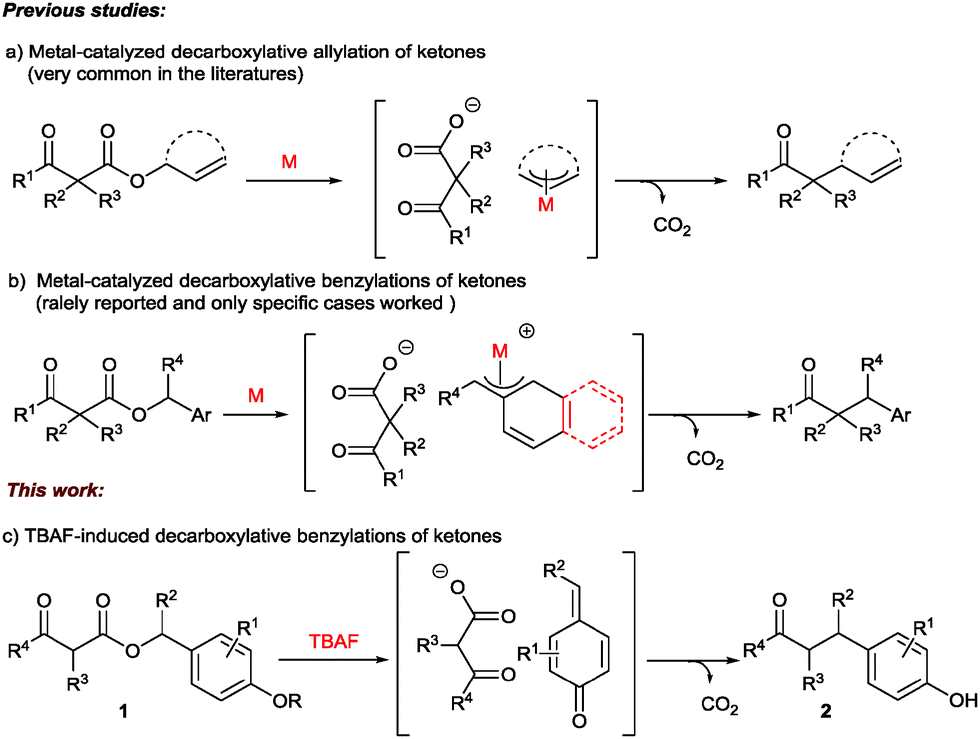
Decarboxylative C sp3 –C sp3 coupling for benzylation of unstable ketone enolates: synthesis of p -(acylethyl)phenols - Chemical Communications (RSC Publishing) DOI:10.1039/C6CC03835B

Environmentally benign process for benzylation of toluene to mono-benzylated toluene over highly active and stable hierarchical zeolite catalyst | SpringerLink

Spin-Center Shift-Enabled Direct Enantioselective α-Benzylation of Aldehydes with Alcohols. - Abstract - Europe PMC

Scheme 2. Benzylation and fluorination reactions leading to compounds... | Download Scientific Diagram
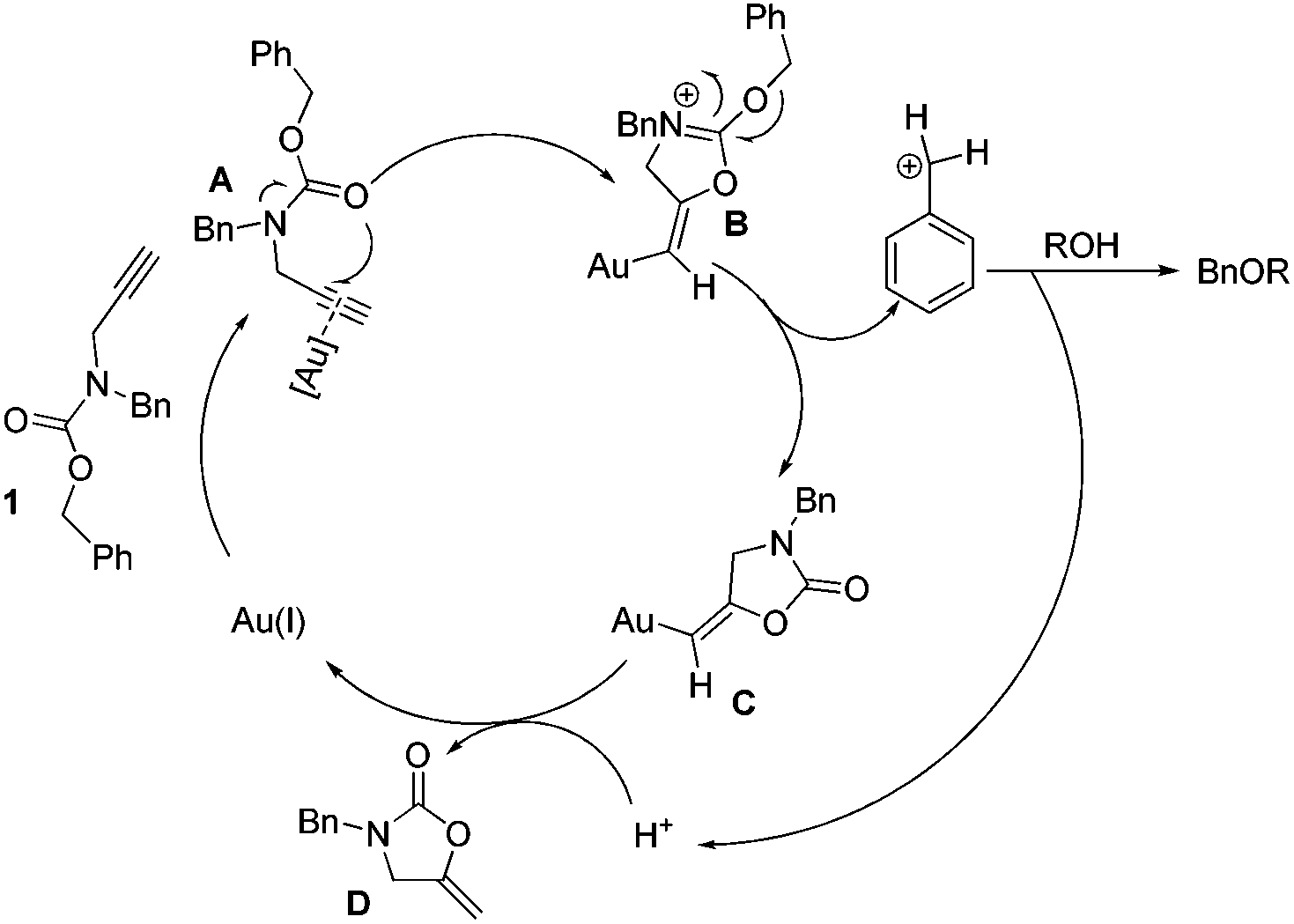
Gold-catalyzed highly efficient benzylation of alcohols with N -Cbz- N -benzyl-propargylamine - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C4QO00255E





