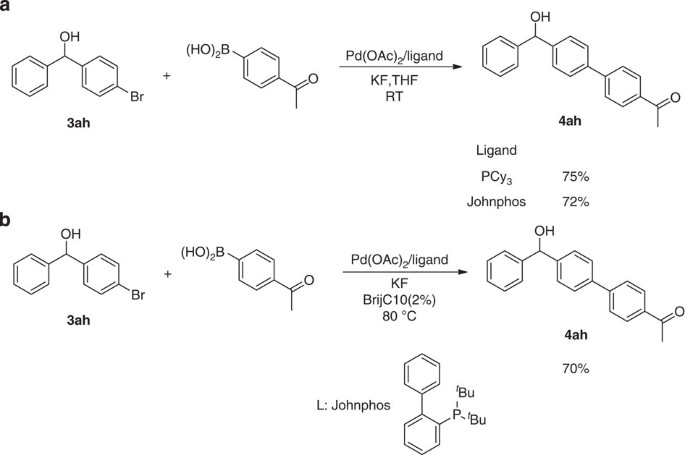
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
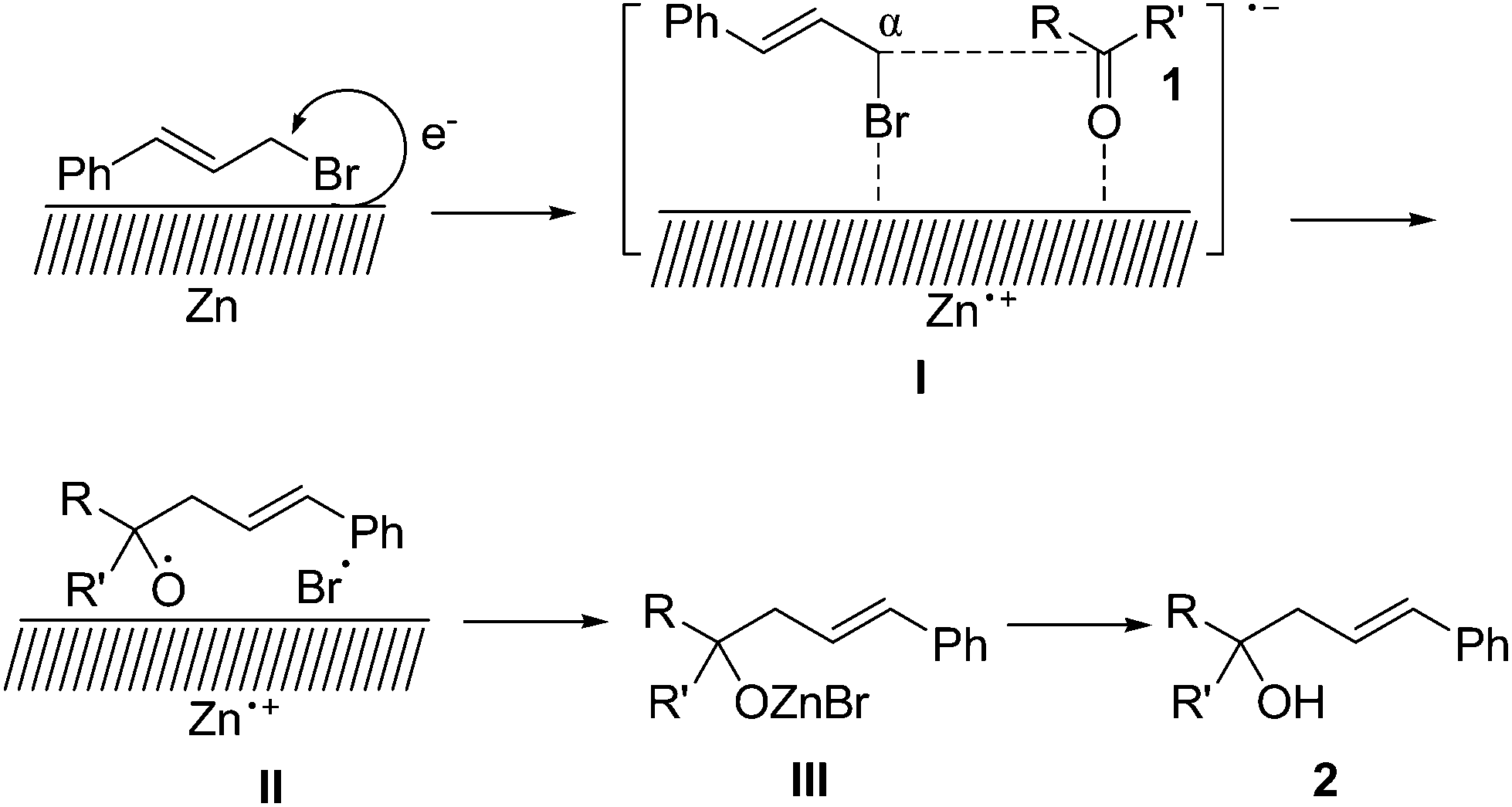
Zinc-mediated α-regioselective Barbier-type cinnamylation reactions of aldehydes, ketones and esters - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB00723J

Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society
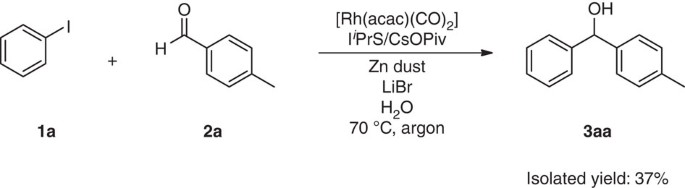
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications

The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
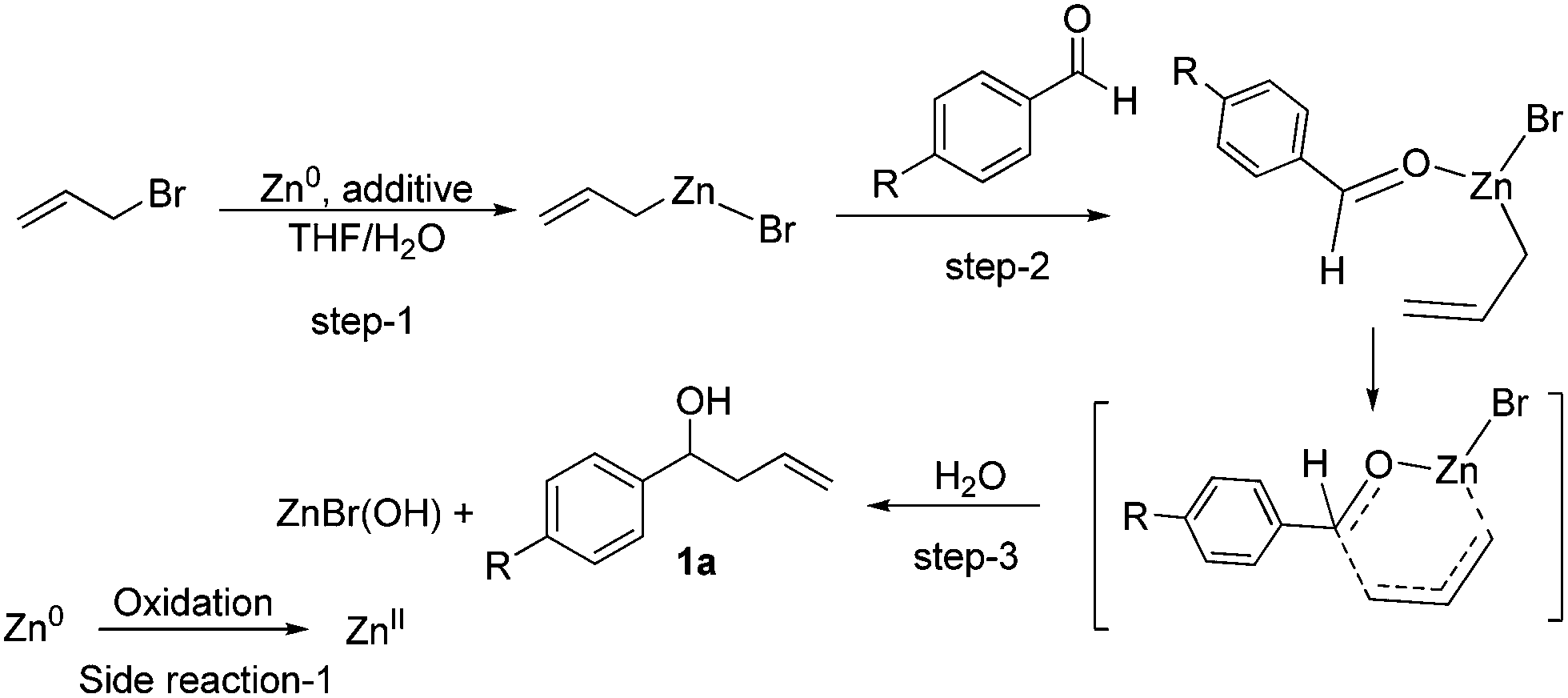
Waste management in zinc promoted allylation of aldehyde - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C5NJ03569D

CoI‐Catalyzed Barbier Reactions of Aromatic Halides with Aromatic Aldehydes and Imines - Presset - 2019 - Chemistry – A European Journal - Wiley Online Library

Zinc‐Catalyzed Organic Synthesis: CC, CN, CO Bond Formation Reactions - Wu - 2012 - Advanced Synthesis & Catalysis - Wiley Online Library
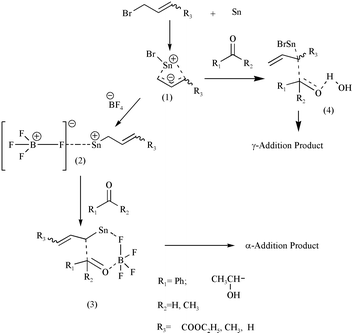
High regio- and stereoselective Barbier reaction of carbonyl compounds mediated by NaBF 4 /Zn (Sn) in water - New Journal of Chemistry (RSC Publishing) DOI:10.1039/B303187J
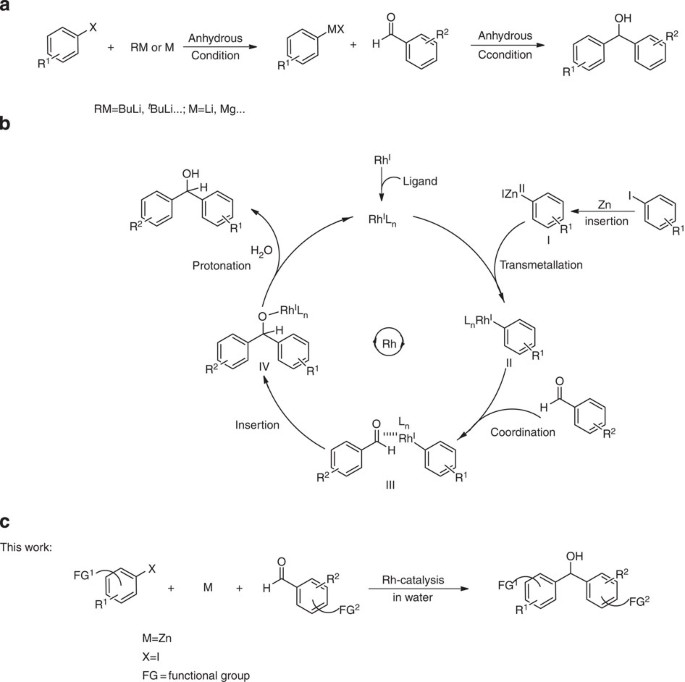





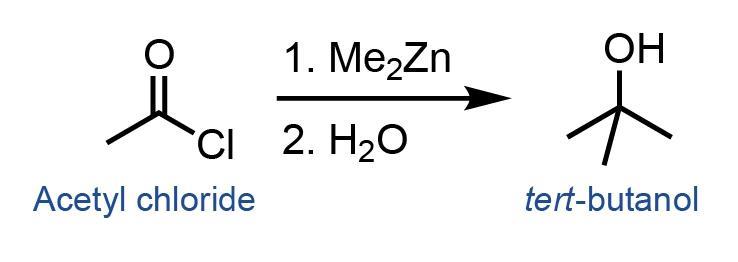



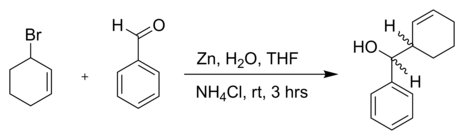


![PDF] The introduction of the Barbier reaction into polymer chemistry | Semantic Scholar PDF] The introduction of the Barbier reaction into polymer chemistry | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bd0646bc8904536c212910b65498158459b8e21a/2-Figure1-1.png)



