He literally just nutted in me and started shopping for fine pentaerythritol, 70% AR grade nitric acid,98% Sulfuric acid, acetone, filter paper, 300 ml beakers, a thermometer, distilled water, and baking soda. - )

CHEMICAL NAME acetic acid aluminum oxide aluminum potassium sulfate aluminum sulfate ammonium hydroxide carbon carbonate calcium hypochloride calcium oxide calcium sulphate carbonic acid carbon tetrachloride ethylene dichloride ferric oxide glucose ...
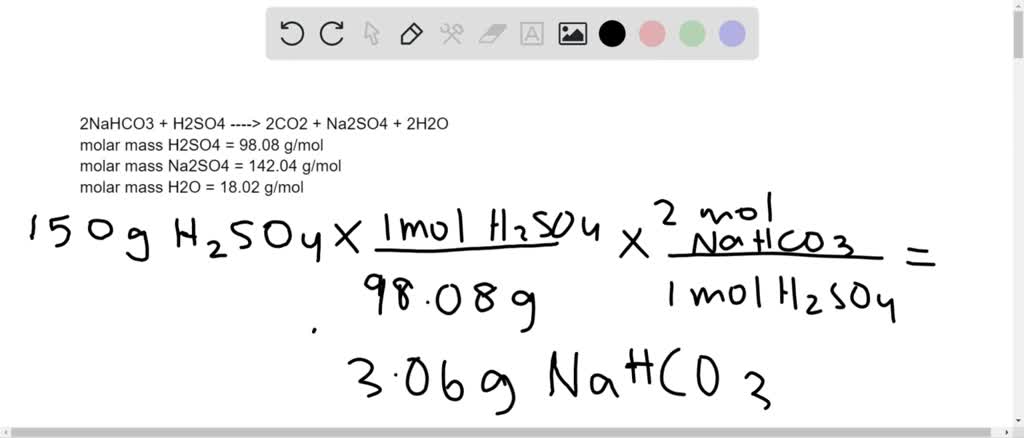
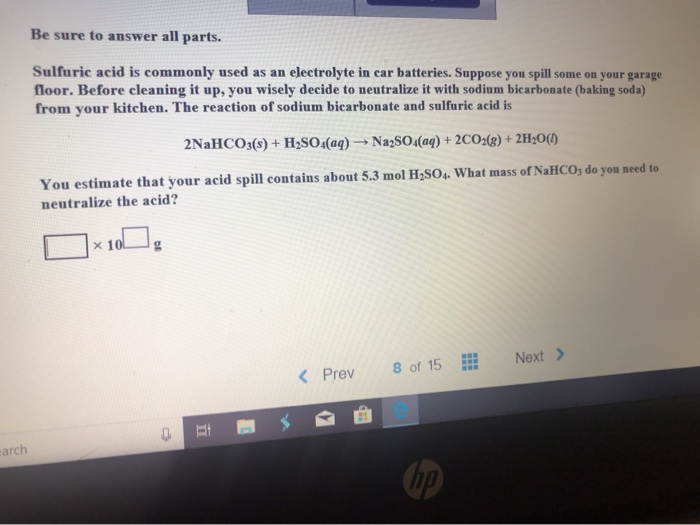
SOLVED:In a soda-acid fire extinguisher, concentrated sulfuric acid reacts with sodium hydrogen carbonate to produce carbon dioxide, sodium sulfate, and water. a. How many moles of sodium hydrogen carbonate would be needed
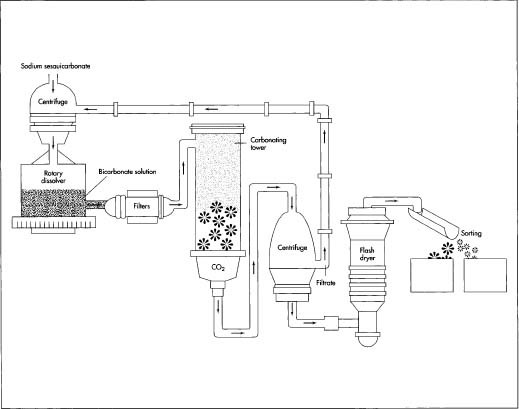
How baking soda is made - material, manufacture, making, history, used, processing, structure, product