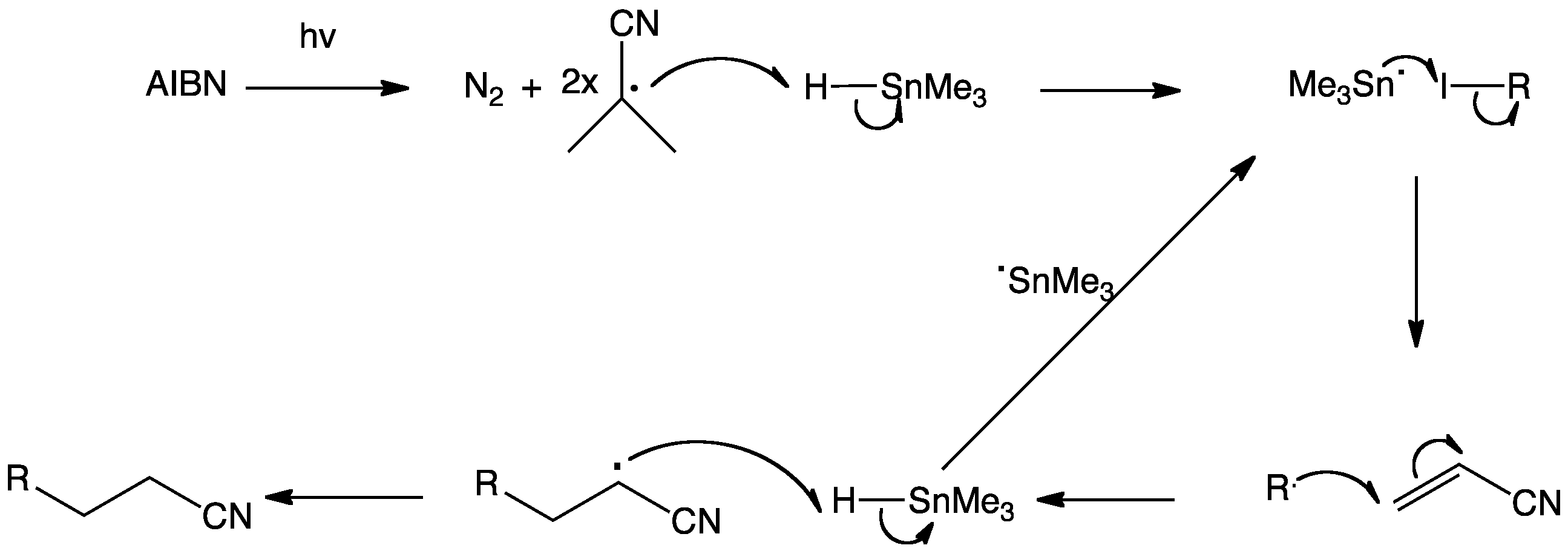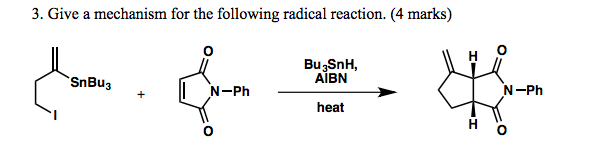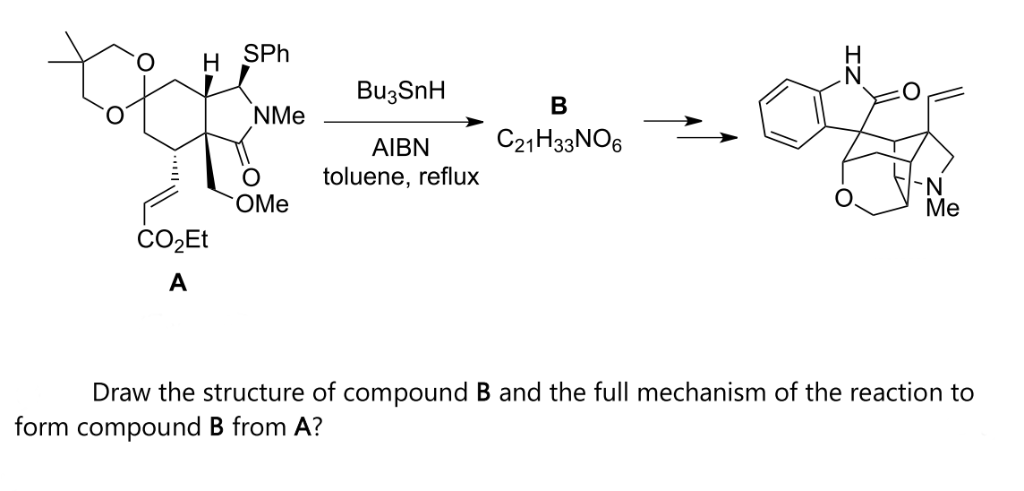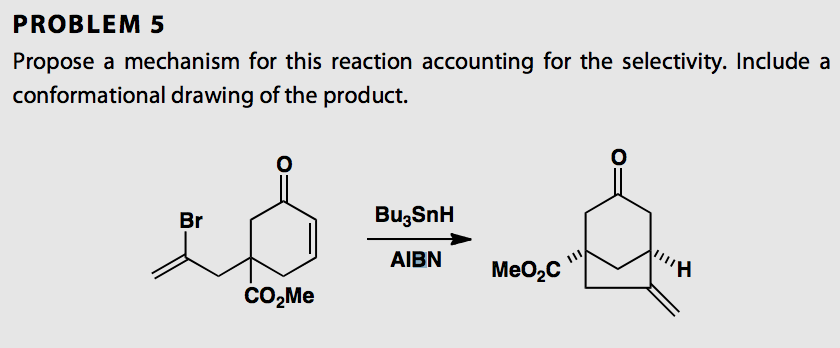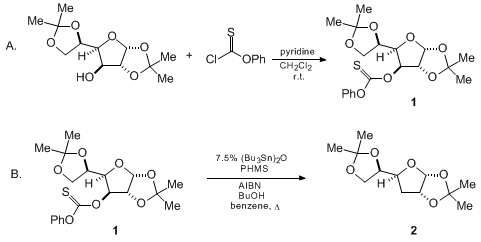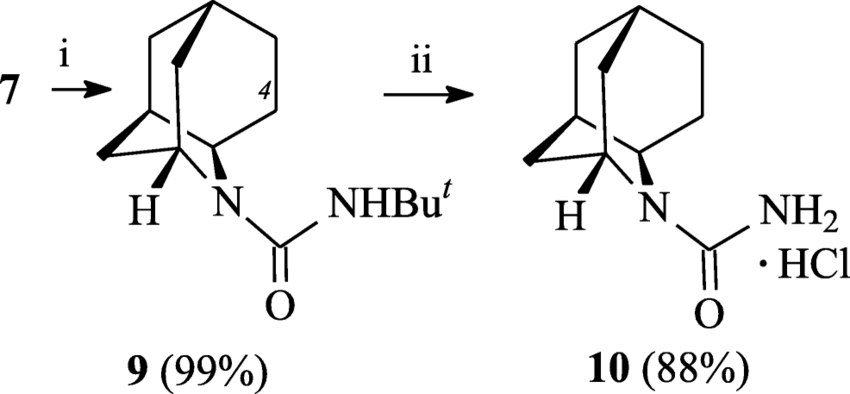
Reagents and conditions: (i) n-Bu3SnH, AIBN, toluene, 100 °C, 6 h; (ii)... | Download Scientific Diagram
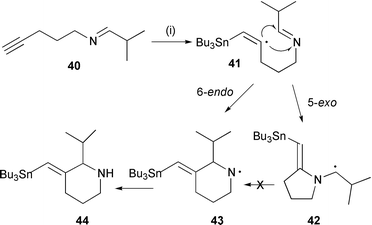
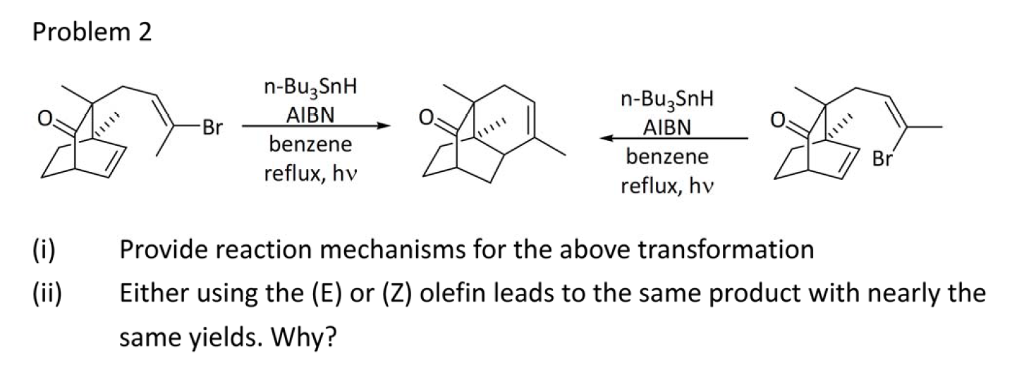
Synthesis of heterocycles by radical cyclisation - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing) DOI:10.1039/A909340K
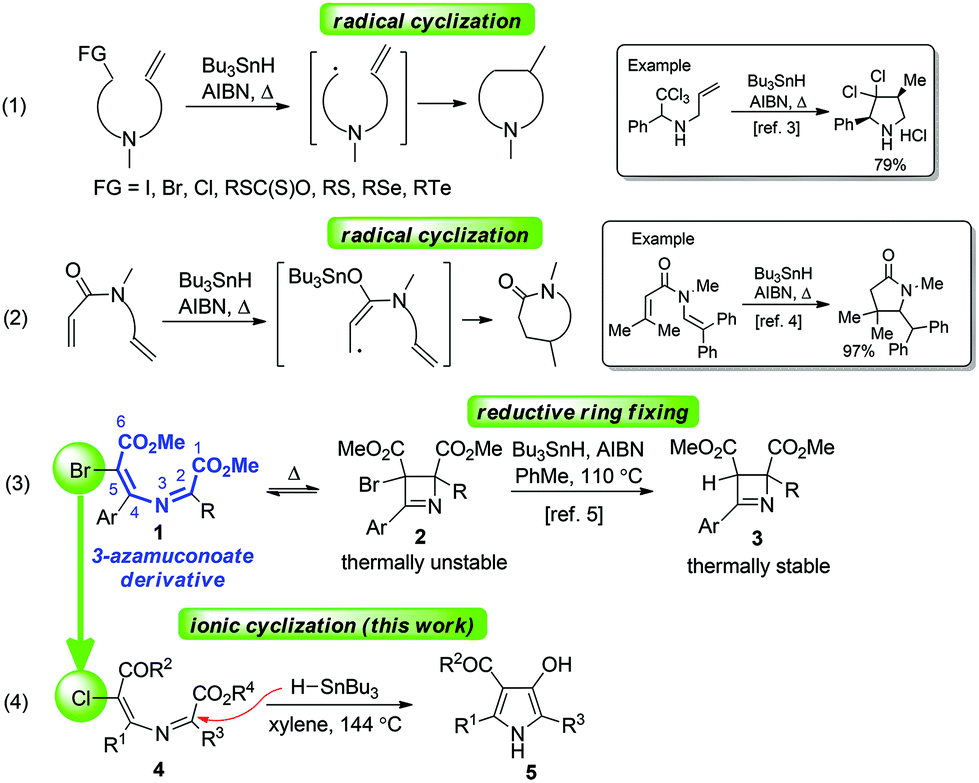
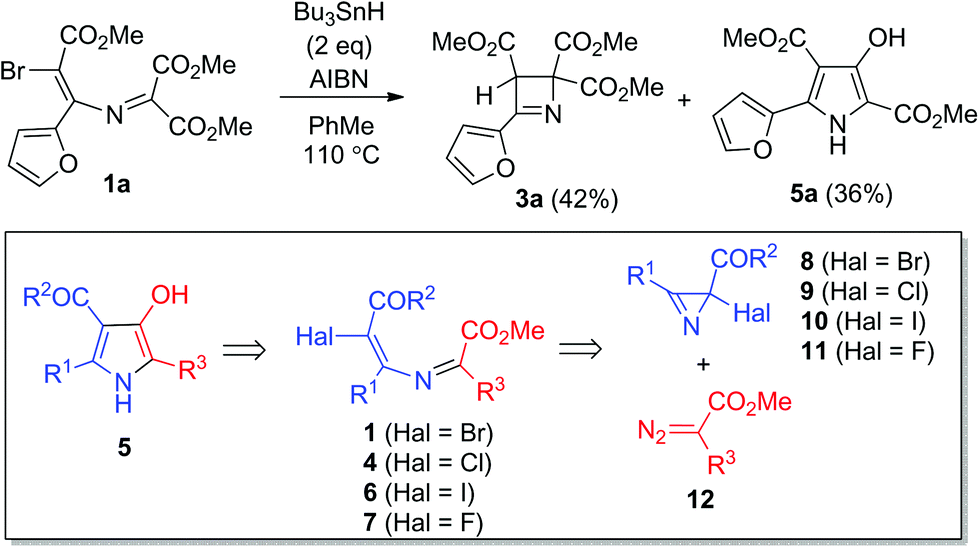
Expedient synthesis of 3-hydroxypyrroles via Bu 3 SnH-triggered ionic 5- exo-trig -cyclization of 5-chloro-3-azamuconoate derivatives - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C8QO00982A
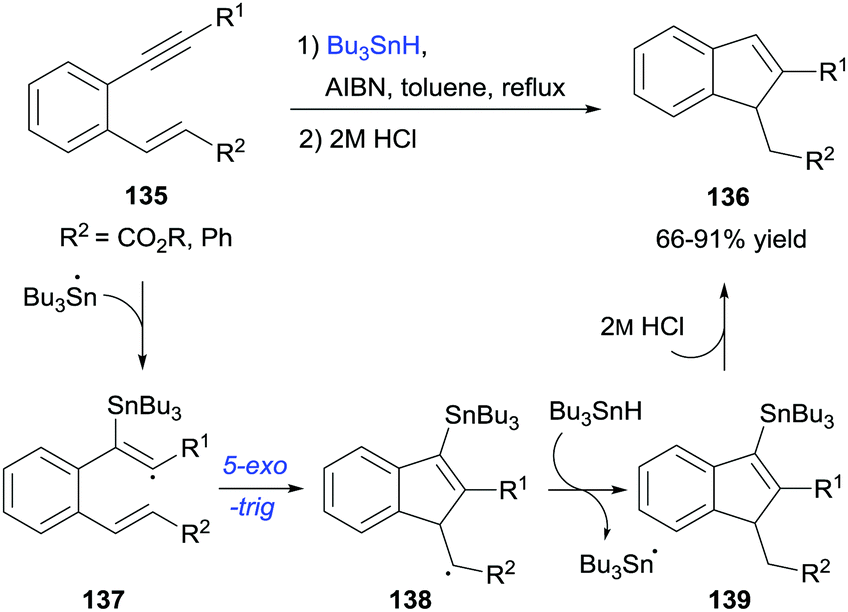
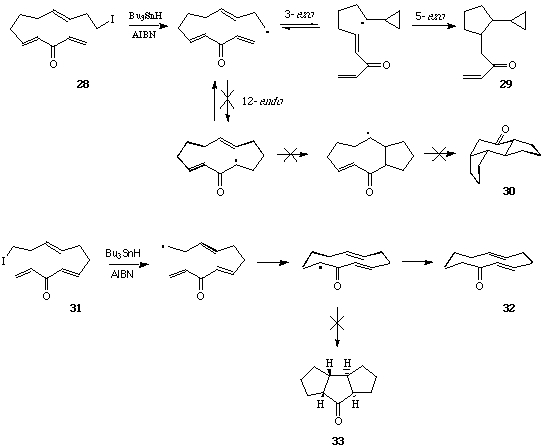
Radical cascade cyclization of 1, n -enynes and diynes for the synthesis of carbocycles and heterocycles - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C6CS00912C

The O‐Directed Free Radical Hydrostannation of Propargyloxy Dialkyl Acetylenes with Ph3SnH/cat. Et3B. A Refutal of the Stannylvinyl Cation Mechanism - Hale - 2019 - The Chemical Record - Wiley Online Library
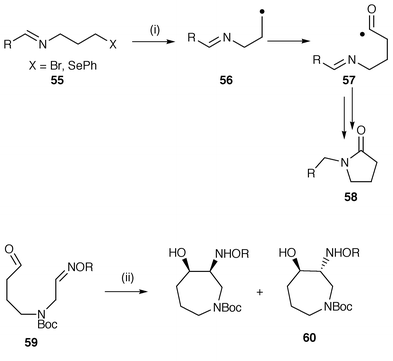
Synthesis of heterocycles by radical cyclisation - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing) DOI:10.1039/A808141G