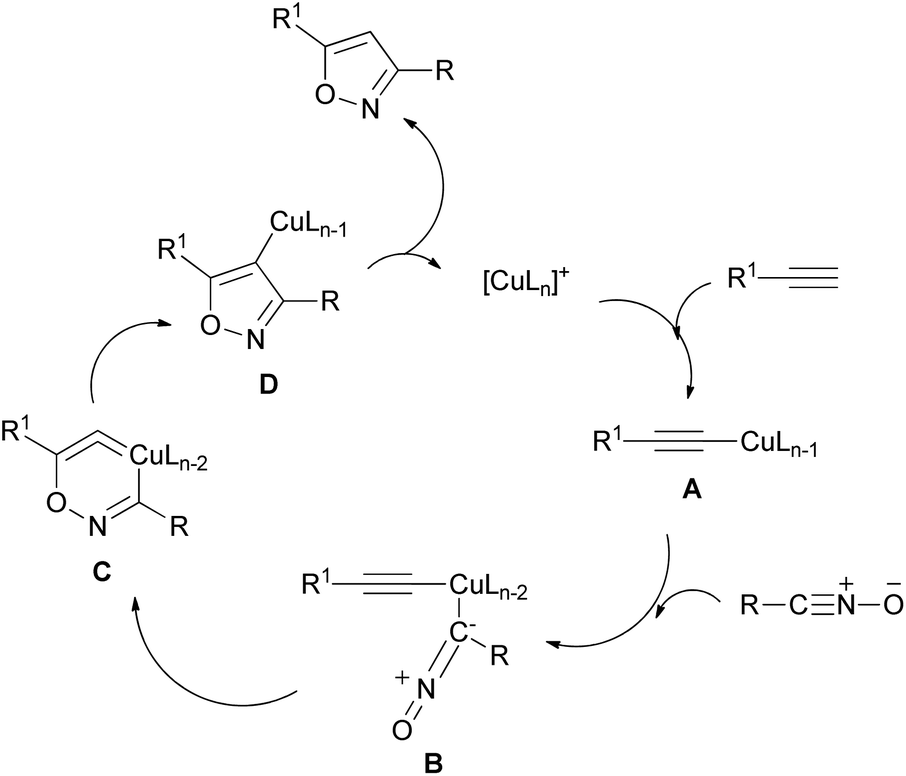
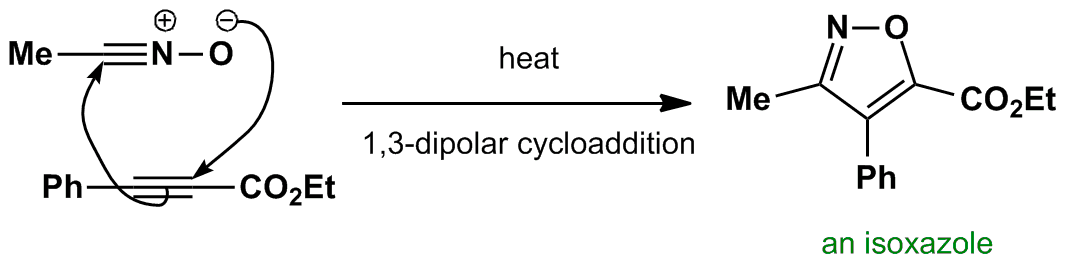
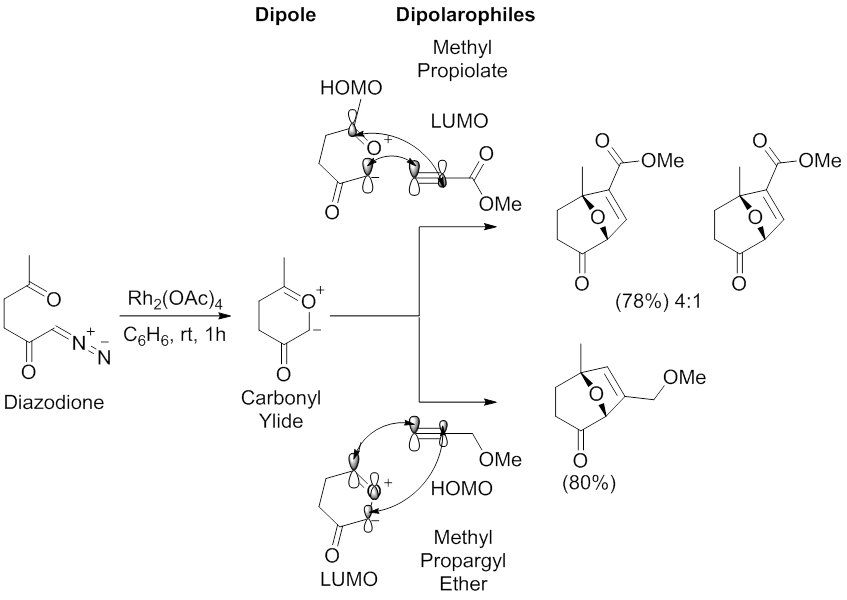
Regio- and stereoselective 1,3-dipolar cycloaddition reactions of C-aryl (or hetaryl)-N-phenylnitrones to monosubstituted ylidene malononitriles and 4-benzylidene-2-phenyloxazol-5(4H)-one
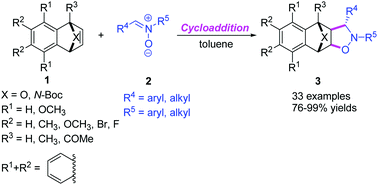
1,3-Dipolar cycloaddition of nitrones to oxa(aza)bicyclic alkenes - Organic Chemistry Frontiers (RSC Publishing)

SciELO - Brasil - The 1,3-Dipolar Cycloaddition of Nitrile Oxide to Vinylacetic Acid: Computational Study of Transition States Selectivity, Solvent Effects, and Bicyclo Formation The 1,3-Dipolar Cycloaddition of Nitrile Oxide to Vinylacetic

The Huisgen Reaction: Milestones of the 1,3‐Dipolar Cycloaddition - Breugst - 2020 - Angewandte Chemie International Edition - Wiley Online Library

Highly Enantioselective Intramolecular 1,3‐Dipolar Cycloaddition: A Route to Piperidino‐Pyrrolizidines - Vidadala - 2015 - Angewandte Chemie International Edition - Wiley Online Library
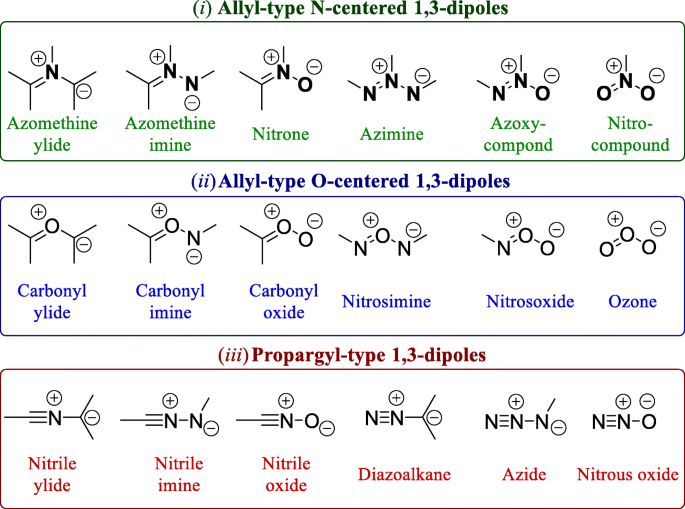
Diradical-singlet character of 1,3-dipoles affects reactivity of 1,3-dipolar cycloaddition reactions and intramolecular cyclization | SpringerLink

The Huisgen Reaction: Milestones of the 1,3‐Dipolar Cycloaddition - Breugst - 2020 - Angewandte Chemie International Edition - Wiley Online Library
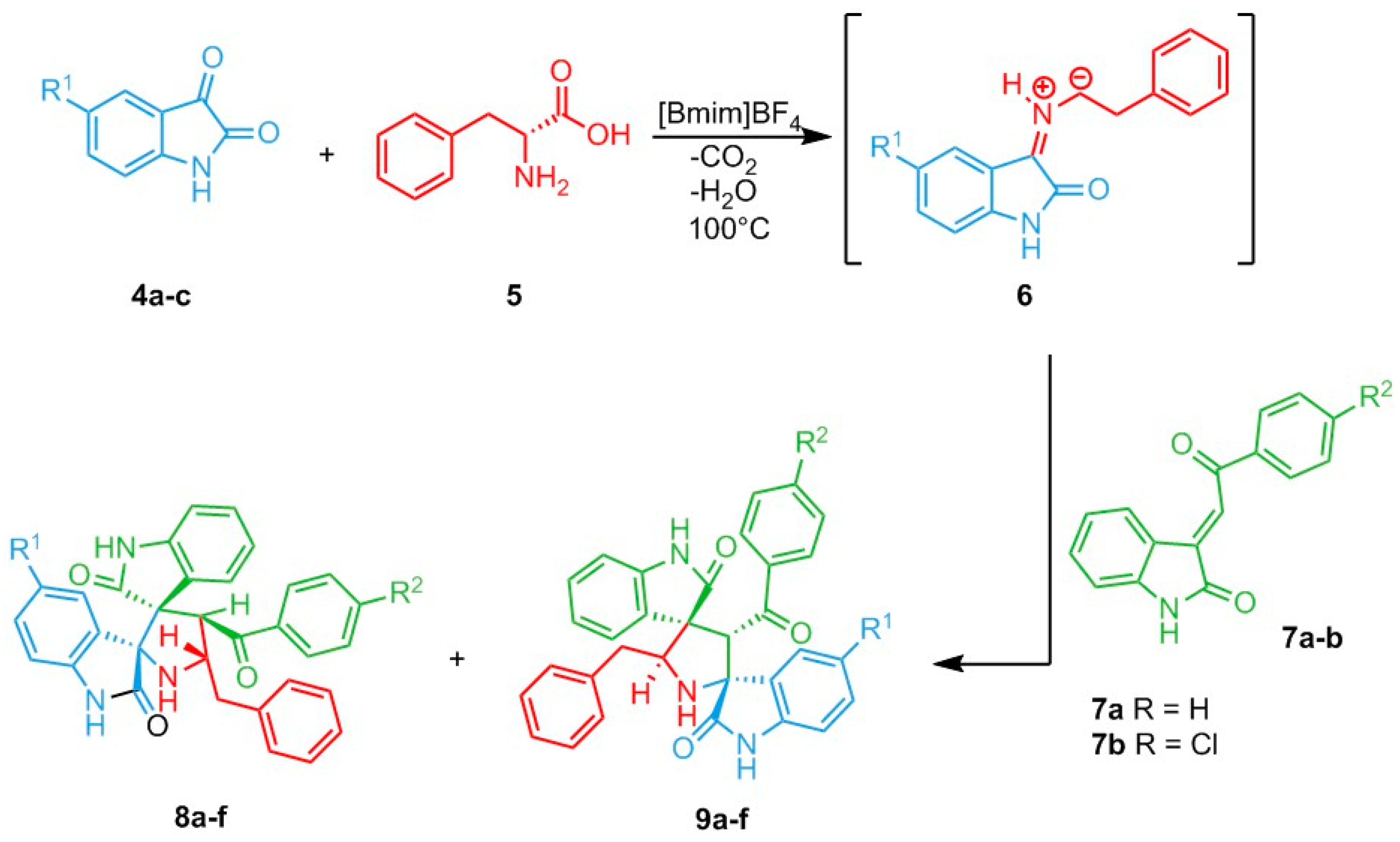
Catalysts | Free Full-Text | Recent Developments on 1,3-Dipolar Cycloaddition Reactions by Catalysis in Green Solvents | HTML